EP1918974A2 - Mass spectrometer assembly and method for ambient liquid mass spectrometry - Google Patents
Mass spectrometer assembly and method for ambient liquid mass spectrometry Download PDFInfo
- Publication number
- EP1918974A2 EP1918974A2 EP07119671A EP07119671A EP1918974A2 EP 1918974 A2 EP1918974 A2 EP 1918974A2 EP 07119671 A EP07119671 A EP 07119671A EP 07119671 A EP07119671 A EP 07119671A EP 1918974 A2 EP1918974 A2 EP 1918974A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- analytes
- liquid
- liquid sample
- laser
- nozzle
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01J—ELECTRIC DISCHARGE TUBES OR DISCHARGE LAMPS
- H01J49/00—Particle spectrometers or separator tubes
- H01J49/02—Details
- H01J49/10—Ion sources; Ion guns
- H01J49/14—Ion sources; Ion guns using particle bombardment, e.g. ionisation chambers
- H01J49/145—Ion sources; Ion guns using particle bombardment, e.g. ionisation chambers using chemical ionisation
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01J—ELECTRIC DISCHARGE TUBES OR DISCHARGE LAMPS
- H01J49/00—Particle spectrometers or separator tubes
- H01J49/02—Details
- H01J49/04—Arrangements for introducing or extracting samples to be analysed, e.g. vacuum locks; Arrangements for external adjustment of electron- or ion-optical components
- H01J49/0431—Arrangements for introducing or extracting samples to be analysed, e.g. vacuum locks; Arrangements for external adjustment of electron- or ion-optical components for liquid samples
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01J—ELECTRIC DISCHARGE TUBES OR DISCHARGE LAMPS
- H01J49/00—Particle spectrometers or separator tubes
- H01J49/02—Details
- H01J49/04—Arrangements for introducing or extracting samples to be analysed, e.g. vacuum locks; Arrangements for external adjustment of electron- or ion-optical components
- H01J49/0459—Arrangements for introducing or extracting samples to be analysed, e.g. vacuum locks; Arrangements for external adjustment of electron- or ion-optical components for solid samples
- H01J49/0463—Desorption by laser or particle beam, followed by ionisation as a separate step
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01J—ELECTRIC DISCHARGE TUBES OR DISCHARGE LAMPS
- H01J49/00—Particle spectrometers or separator tubes
- H01J49/02—Details
- H01J49/10—Ion sources; Ion guns
- H01J49/16—Ion sources; Ion guns using surface ionisation, e.g. field-, thermionic- or photo-emission
- H01J49/165—Electrospray ionisation
Abstract
Description
- The invention relates to a method for mass spectrometry, more particularly to a method for ambient liquid mass spectrometry that is capable of conducting direct analysis of mass spectrometry on a liquid sample under atmospheric pressure. The present invention also relates to a mass spectrometer assembly for conducting the method of ambient liquid mass spectrometry.
- A method for mass spectrometry is called electrospray ionization mass spectrometry (ESI-MS), which involves ionizing proteins contained in a liquid sample, followed by a protein analysis. As illustrated in FIG.1, an electrospray ionization mass spectrometer (ESI-MS) 1 includes an
electrospray ionization device 11. - The
electrospray ionization device 11 performs an electrospray ionization procedure to ionize the proteins in the liquid sample. An electric field is established between anopen end 111 of a capillary 112 and anentrance side 121 of amass analyzer 12. Subsequently, the liquid sample is pushed through thecapillary 112 toward theopen end 111. As the electric field force overcomes the surface tension of the liquid sample at theopen end 111 of thecapillary 112, aerosol droplets containing multivalent electric charges and protein molecules are formed, and are pushed into themass analyzer 12 through theentrance side 121. The multivalent electrons are attached to the protein molecules to form ionized protein molecules with relatively lower mass-to-charge ratio (m/z) values as the charged droplets dwindle in size when traveling through the air from theopen end 111 of thecapillary 112 toward theentrance side 121 of themass analyzer 12. However, body fluids or other biochemical solutions normally contain a high concentration of various salts. A "desalination" pre-process, such as dialysis, is required to prevent the protein molecules from being ionized by acquiring charges from the salts that are present in the body fluids/biochemical solutions, to thereby result in a simpler ion peak configuration in the mass spectrum obtained from ESI-MS. However, professional personnel are required to execute the "desalination" pre-process, which is a tedious, time consuming and inconvenient process. - Other methods for mass spectrometry require converting an originally liquid-state sample into a solid-state sample prior to conducting the analysis. One of these methods is called the matrix-assisted laser desorption ionization mass spectrometry (MALDI-MS). In MALDI-MS, a water soluble organic acidic matrix of highly laser light absorbing small organic molecules is mixed with a liquid sample containing protein molecules before the mixture is dehydrated to form a crystal. A laser beam is irradiated on the surface of the crystal, causing ionization and desorption of the protein molecules. Under an electric field, the ionized protein molecules are introduced into a mass analyzer for mass spectrometric analysis. However, the desorption process of MALDI-MS needs to be conducted in vacuum. Furthermore, it is inconvenient and time consuming to transform liquid samples into solid samples in order to perform MALDI-MS. In addition, the matrix used in MALDI-MS is generally an organic acid, which affects the analyte (e.g., proteins) chemically, causing the structure of the analyte to change.
- A special type of MALDI-MS called "surface-assisted laser desorption/ionization" (SALDI) mass spectrometry (SALDI-MS) is capable of conducting mass spectrometric analysis on a liquid sample, but a vacuum environment is still required. Moreover, a highly viscous solute, such as glycerin, is required for preparing the liquid sample, keeping the cost of instrumentation high and preparation of the sample tedious.
- It can be seen from the above than conducting protein analysis directly on a liquid sample using mass spectrometry techniques presents a variety of difficulties and inconveniences. Since spatial analytic information on proteins of organs or tissues is extremely important in the medical and biotechnological fields, there exists a great need for a method of mass spectrometry that is capable of conducting rapid, convenient, and accurate protein analysis on a liquid sample under atmospheric pressure.
- Therefore, the object of the present invention is to provide a laser desorption device, a mass spectrometer assembly, and a method for mass spectrometry that is capable of conducting mass analysis directly on a liquid sample under atmospheric pressure.
- According to one aspect of the present invention, there is provided a method for mass spectrometry, which is named "ambient liquid mass spectrometry" (ALMS), and which includes the steps of:
- placing, on a sample stage, a liquid sample including a solution that contains a plurality of analytes and a material serving as a matrix for absorbing laser energy so as to assist in desorption of at least one of the analytes;
- providing an electrospray unit that includes a nozzle configured to sequentially form liquid drops of an electrospray medium thereat;
- providing a receiving unit that is disposed to admit therein ionized analytes that are derived from the liquid sample, and that are to be analyzed by a mass analyzer disposed downstream of the receiving unit, the receiving unit being spaced apart from the nozzle of the electrospray unit in a longitudinal direction so as to define a traveling path;
- establishing a potential difference between the nozzle of the electrospray unit and the receiving unit, the potential difference being of an intensity such that the liquid drops are laden with a plurality of charges, and such that the liquid drops are forced to leave the nozzle as multiple-charged ones for heading toward the receiving unit along the traveling path; and
- irradiating the liquid sample with a laser beam such that, upon irradiation, laser energy is passed on to at least one of the analytes contained in the solution of the liquid sample via the matrix so that said at least one of the analytes contained in the liquid sample is desorbed to fly along a flying path which intersects the traveling path so as to enable said at least one of the analytes to be occluded in the multiple-charged liquid drops, and such that as a result of dwindling in size of the multiple-charged liquid drops when approaching the receiving unit along the traveling path, charges of the liquid drops will pass on to said at least one of the analytes occluded therein to form a corresponding one of the ionized analytes.
- According to another aspect of the present invention, there is provided a laser desorption device for use in a mass spectrometer assembly.
- The mass spectrometer assembly includes a receiving unit, an electrospray unit, and a voltage supplying member. The laser desorption device includes a sample stage and a laser transmission mechanism. The sample stage and the laser transmission mechanism are arranged with the receiving unit, the electrospray unit, and the voltage supplying member in a manner such that all the steps of the abovementioned method can be duly carried out. The laser transmission mechanism can be one of an ultraviolet (UV) laser, an infrared (IR) laser, a nitrogen laser, an argon ion laser, a helium-neon laser, a carbon dioxide (CO2) laser, and a garnet (Nd:YAG) laser.
- Other features and advantages of the present invention will become apparent in the following detailed description of the preferred embodiments with reference to the accompanying drawings, of which:
- FIG.1 is a schematic diagram of various components included in an electrospray ionization mass spectrometer (ESI-MS) of the prior art to illustrate relative positions of the components and operational method involved in the ESI-MS;
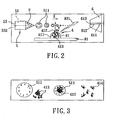
- FIG. 2 is a schematic diagram of a laser desorption device and an electrospray unit for the first preferred embodiment of a mass spectrometer assembly implementing the method of ambient liquid mass spectrometry (ALMS) according to the present invention, illustrating desorption of analytes contained in a liquid sample so as to fly along a flying path that intersects a traveling path of multiple-charged liquid drops;
- FIG.3 is a schematic diagram, illustrating occlusion of the analytes in the multiple-charged liquid drops, and formation of ionized analytes as a result of dwindling in size of the multiple-charged liquid drops having the analytes occluded therein;
- FIG. 4 is a schematic side view of the first and fifth preferred embodiments of a mass spectrometer assembly implementing the method of ALMS according to the present invention;
- FIG.5 is a fragmentary enlarged view of the second preferred embodiment of a mass spectrometer assembly implementing the method of ALMS according to the present invention, illustrating relative positions of an airstream supplying mechanism and a nozzle;
- FIG.6 is a fragmentary sectional view of the third preferred embodiment of a mass spectrometer assembly implementing the method of ALMS according to the present invention, illustrating relative positions of a micro-tube, a nozzle, and a pump;
- FIG. 7 is a schematic side view of the fourth and sixth preferred embodiments of a mass spectrometer assembly implementing the method of ALMS according to the present invention;
- FIG.8(a) to 8(c) illustrate mass spectra obtained as experiment results of comparative example 1 and
exemplary methods - FIG. 9(a) to 9(f) illustrate mass spectra obtained as experiment results of
exemplary methods 3 to 8. - Before the present invention is described in greater detail, it should be noted herein that like elements are denoted by the same reference numerals throughout the disclosure. It is also noted herein that in the accompanying drawings, sizes of constituting elements and relative distances among the elements are not drawn to scale.
- In this invention, a suitable matrix is added to a liquid sample prior to conducting a mass spectrometric analysis. Particularly, as shown in FIGS.2 to 4, while an electrospray ionization process is implemented to form sequentially multiple-charged
liquid drops 511 of aliquid electrospray medium 51, alaser beam 821 is irradiated onto aliquid sample 4, which includes asolution 41 that containsanalytes 412 and amaterial 413 serving as a matrix for absorbing laser energy, and which is disposed in the passage way of a receivingunit 6 adapted to admit therein ionizedanalytes 414 that are derived from theliquid sample 4 for mass spectrometric analysis. The obtained mass spectrometric analysis results established that this novel technique, referred to as "ambient liquid mass spectrometry" (ALMS), is practicable directly on liquid samples under atmospheric pressure. - As the
liquid sample 4 is irradiated by thelaser beam 821, laser energy which is absorbed by thematrix material 413 is presumably passed on to at least one of theanalytes 412 via thematrix material 413 so that said at least one of theanalytes 412 is desorbed, and is occluded in the multiple-chargedliquid drops 511 formed during the electrospray ionization process. As a result of dwindling in size of the multiple-chargedliquid drops 511 when approaching the receivingunit 6, charges of theliquid drops 511 will pass on to said at least one of theanalytes 412 occluded therein to form a correspondingionized analyte 414. Theionized analyte 414 is received by thereceiving unit 6 for mass spectrometric analysis thereby. - Moreover, since water molecules are highly absorbent to infrared (IR) light, the water molecules contained in an aqueous solution serve as the matrix for absorbing laser energy and transferring the laser energy to the analytes. Therefore, with procedures similar to those disclosed hereinabove, an infrared laser beam is employed to irradiate directly on an aqueous solution to obtain accurate mass spectrometric analysis results.
- The abovedescribed novel method of mass spectrometry, named "Ambient Liquid Mass Spectrometry" (ALMS), apparently opens up a new era for mass spectrometric analysis of liquid samples, especially on aqueous solutions containing proteins, under atmospheric pressure. Operation procedure of ALMS is relatively simple and rapid, and resolution thereof is higher than that of ESI-MS. ALMS is capable of accurately detecting molecular weights of analytes, even when the analytes are macromolecules, such as proteins, thereby showing an outstanding ability in protein identification. These advantages allow ALMS to quickly analyze biochemical and medical liquid samples so as to obtain reliable results, which is extremely favorable in relevant applications, such as immediate diagnosis of diseases.
- As shown in FIGS. 2 to 4, the method of ALMS according to the present invention can be implemented by performing the following steps:
- Place a
liquid sample 4, on asample stage 81, that includes asolution 41 containing a plurality ofanalytes 412 and amatrix material 413 for absorbing laser energy so as to assist in desorption of theanalytes 412. In particular, thesolution 41 includes a solvent 411 that contains theanalytes 412 and thematrix material 413 therein. - Provide an
electrospray unit 5 that includes areservoir 52 for accommodating aliquid electrospray medium 51, and anozzle 53 which is disposed downstream of thereservoir 52, and which is configured to sequentially form liquid drops 511 of theelectrospray medium 51 thereat. - Provide a receiving
unit 6 that is spaced apart from thenozzle 53 for receiving and analyzingionized analytes 414 derived from theliquid sample 4. - Provide a
detector 7 for detecting signals generated as a result of analyzing theionized analytes 414 by the receivingunit 6, and for generating a mass spectrum of theliquid sample 4 from the signals. - Establish between the
nozzle 53 and the mass analyzer 61 a potential difference which is of an intensity such that the liquid drops 511 are laden with a plurality of electric charges, and such that the liquid drops 511 are forced to leave thenozzle 53 as multiple-charged ones for heading toward the receivingunit 6 along a traveling path (X). - Irradiate the
liquid sample 4 with alaser beam 821 such that at least one of theanalytes 412 is desorbed to fly along a flying path (Y) which intersects the traveling path (X) so as to enable said at least one of theanalytes 412 to be occluded in the multiple-charged liquid drops 511, and such that as a result of dwindling in size of the multiple-charged liquid drops 511 when approaching themass analyzer 61 along the traveling path (X), charges of the liquid drops 511 will pass on to said at least one of theanalytes 412 to form a corresponding one of the ionizedanalytes 414. - The electrospray medium forming the liquid drops is a solution normally used in electrospray methods. An example of the electrospray medium is a solution containing a volatile liquid such that the liquid portion in the liquid drops can vaporize prior to the receipt of the ionized analytes by the mass analyzer. Further, in order to help dissolve protein molecules and avoid interference due to an addition of salt in the volatile liquid, and to simplify the resultant mass spectrum, the volatile liquid is preferably one with a low polarity, such as isoacetonitrile, acetone, alcohol, etc. If the analyte in the liquid sample is a protein, and it is desired to investigate the un-denatured state of the protein, the electrospray medium is preferably a solution that contains a volatile liquid and that does not contain an acid, such as methanol aqueous solution.
- Based on different requirements, "an aqueous solution containing methanol and acetic acid" and a "methanol aqueous solution" are used as the electrospray medium in the embodiments of the present invention, respectively. In addition, it is assumed that the ion portion of the obtained analytes is multivalent with each charge being contributed by a proton (H+) .
- One of the main objects that the method of ALMS aims at is the detection of analytes from a liquid sample including a solution that contains the analytes and a matrix material. Therefore, no limitation is imposed on the types of solutions and the kinds of analytes detectable for the implementations of the present invention. Whether the solution is an aqueous solution, contains an organic solvent, or is a body fluid secreted by an organism and having a complicated composition, and whether the analytes are macroscopic molecules such as proteins, or are microscopic molecules such as ordinary compounds, mass spectrometric analysis results can be obtained through implementing the method of ALMS according to the present invention.
- Therefore, the liquid sample under study can include various solutions, including organism's body fluids, chemical solutions, environment sampling solutions, or various eluates from liquid chromatography, etc. Preferably, the organism's body fluid can be selected from the group consisting of blood, tear, milk, perspiration, intestinal juice, brains fluid, spinal fluid, lymph, pus, blood serum, saliva, nasal mucus, urine, and excrement. When the liquid sample under study includes a chemical solution, the chemical solution can be an organic solution.
- Preferably, the matrix material ismade fromamaterial that is non-transmissible by laser, such as gold, carbon, cobalt, iron, 2,5-dihydroxybenzoic acid (2,5-DHB), 3,5-dimethoxy-4-hydroxycinnamic: acid (sinapinic acid, (SA)), α-cyano-4-hydroxycinnamic acid (α-CHC), and a combination thereof. Better results are obtained when the material serving as the matrix has a particle diameter ranging from 50 nm to 50 µm.
- When the solution included in the liquid sample is an aqueous solution, water molecules contained in the aqueous solution would be the material serving as the matrix, and an infrared laser beam is used for desorption. Incidentally, it is also practicable to add another material purposely into an aqueous solution to serve as the matrix. However, when it is desired to analyze an organic solution, a material serving as a matrix is added into the solution to form the liquid sample under study prior to implementing the method of ALMS. Descriptions related to detailed operational practices and mechanisms for the method of ALMS will be described in subsequent embodiments.
- The present invention is described in greater detail hereinbelow with respect to the preferred embodiments and exemplary applications presented. It should be noted herein that the embodiments and exemplary applications are for illustrative purposes only, and should not be considered as limitations imposed on the present invention.
- The preferred embodiments, exemplary methods, and comparison (experiment) example were conducted using the following chemicals and equipments:
- 1. Laser Transmission Mechanism:
- a. Ultraviolet (UV) Laser model no. VSL-3371, manufactured by Laser, Science Inc. of the United States. The laser beams transmitted by the ultraviolet laser have a wavelength of 337nm, a frequency of 10Hz, a pulse duration of 4ns, and a pulse energy of 100µJ.
- b. Infrared (IR) Laser model no. LS-2130SHP, manufactured by LOTIS TII of Russia. The laser beams transmitted by the infrared laser have a wavelength of 1064nm, a frequency of 2Hz, a pulse duration of 0.5ns, and a pulse energy of 50mJ.
- 2. Mass Analyzer (including the Detector): Quadrupole Time-of-Flight Mass Analyzer model no. BioTOF-Q, manufactured by Bruker Dalton company of Germany.
- 3. Electrospray Medium:
- a. Methanol: an HPLC solvent manufactured by Merck company of Germany.
- b. acetic acid: an HPLC solvent manufactured by Mallinckrodt company of Germany.
- 4. Analytes:
- a Protein Standard: insulin (molecular weight of 5733), myoglobin (molecular weight of 17566), lysozyme (molecular weight of 14305), and cytochrome c (molecular weight of 12232), all of which are high purity protein standards with concentrations of above 95% and manufactured by Sigma-Aldrich company of the United States.
- b. Hemin: molecular weight of 652.0, model no. H-2250 manufactured by Aldrich company of the United States.
- c. 18-crown-6-ether: molecular weight of 264.32, model no. C0860 manufactured by Tokyo Chemical Industry Co., Ltd. of Japan.
- d. 1-hexadecylamine: molecular weight of 241.46, model no. H740-8 manufactured by Aldrich company of the United States.
- e. Methyl (triphenyl-phosphoranylidene) acetate: molecular weight of 334, model no. 64941 manufactured by Fluka company.
- f. Cinnamic acid benzyl ester: molecular weight of 260, model no. C0358 manufactured by Tokyo Chemical Industry Co., Ltd. of Japan.
- g. Cetylpyridinium chloride: molecular weight of 339.99 (note: Chemical equation of cetylpyridinium chloride is C21H38N-C1 with an average molecular weight of 339. 99 and a monoisotope molecular weight of 339.27, where C21H38N(304.30) is a cation and Cl(34.97) is an anion. Since a mass spectrometer detects cations, the molecular weight obtained is not 339.99), model no. 145-100G manufactured by AJAX Chemical.
- 5. Solvents:
- a. Methanol (identical to the above)
- b. Tetrahydrofuran (THF): model no. 9440-03 manufactured by J. T. Baker company of the United States.
- c. Ethyl acetate: model no. 9282-03 manufactured by J. T. Baker company of the United States.
- d. Methylene dichloride: a HPLC solvent manufactured by Merck company of Germany.
- e. Tolene: an HPLC solvent manufactured by J. T. Baker company of the United States.
- f. N-hexane: an HPLC solvent manufactured by J. T. Baker company of the United States.
- 6. Other chemicals:
- a. H2O2: concentration of 30%, model no. 31642 manufactured by Riedel-de Haën company.
- b. NaoH: model no. SK371842 manufactured by Nihon Shiyaku Industries Ltd.
- 7. Matrix Material:
- a. Carbon powders: model no. 4206A manufactured by Merck company of Germany; particle diameter of below 50µm.
- b. Gold nano-particles: provided privately; particle diameter of approximately 56nm.
- c. α -cyano-4-hydroxycinnamic acid (α -CHC), an HPLC material manufactured by Sigma-Aldrich company of the United States.
- d. 2,5-dihydroxybenzoic acid (2,5-DHB): model no. D0569 manufactured by Tokyo Chemical Industry Co., Ltd. of Japan.
- e. 3,5-dimethoxy-4-hydroxycinnamic acid (Sinapinic acid (SA)) : model no. D1765 manufactured by Tokyo Chemical Industry Co., Ltd. of Japan.
- 8. Matrix-Assisted Laser Desorption Ionization Mass Spectrometer (MALDI-MS): model no. Autoflex MALDI/TOF, manufactured by Bruker Dalton company of Germany, and suitable for analyzing macromolecules in the linear mode.
- 9. Electrospray Ionization Mass Spectrometer (ESI-MS): including an electrospray unit, a mass analyzer, and a detector; the electrospray unit, the mass analyzer and the detector are identical to those used in the embodiments of the mass spectrometer assembly implementing the method of ALMS according to the present invention.
- 10. Relevant chemicals or equipments for bacterial extraction:
- a. Glass beads: model no. 11079101 manufactured by Biospec Products, Inc.; diameter of 100 µm.
- b. Sonicator: model no. XL2020 manufactured by Heat Systems, Inc.
- c. Centrifuge: modelno. DSC-1524SDT TFA manufactured by Digisystem Laboratory Instruments, Inc.
- d. Trifluroacetic acid: an analysis class acid with model no. 61030 manufactured by Riedel-de Haën company.
- e. Acetonitrile (ACN) : an HPLC material with model no. UN1648 manufactured by Merck company of Germany.
- Referring to FIG.4, the first preferred embodiment of a mass spectrometer assembly implementing the method of ALMS is adapted to conduct mass spectrometric analysis on a
liquid sample 4. With reference back to FIG. 2 and FIG. 3, theliquid sample 4 includes asolution 41 including a solvent that contains a plurality ofanalytes 412 and amaterial 413 serving as a matrix (also referred to as a matrix material 413) for assisting in desorption of at least one of theanalytes 412. The mass spectrometer assembly includes anelectrospray unit 5, a receivingunit 6, avoltage supplying member 3, and alaser desorption device 8. - The
laser desorption device 8 includes asample stage 81 on which theliquid sample 4 is placed, alaser transmission mechanism 82 that is capable of transmitting alaser beam 821 and that is disposed to irradiate theliquid sample 4, alens 83 that is disposed to receive thelaser beam 821 from thelaser transmission mechanism 82 for focusing the energy carried by thelaser beam 821, and areflector 84 that is disposed to change the path of thelaser beam 821. In this embodiment, thelaser transmission mechanism 82 is an ultravioletlaser transmission mechanism 82a that is capable of transmitting thelaser beam 821. In principle, thelaser desorption device 8 is designed as long as thelaser desorption device 8 is capable of irradiating theliquid sample 4 such that, upon irradiation, at least one of theanalytes 412 contained in thesolution 41 of theliquid sample 4 is desorbed. Therefore, in practice, thelens 83 and thereflector 84 can be varied in position as required, or can even be completely eliminated according to other embodiments of the present invention. - The
sample stage 81 of thelaser desorption device 8 includes asupport member 811 that is made from a material non-transmissive by laser, and ahoister platform 812 that is provided for mounting of thesupport member 811 thereon, and that is movable. Thesupport member 811 is provided for placement of theliquid sample 4, and has asupport surface 813 for placement of theliquid sample 4 directly thereon. This way, an operator can begin performing the method of ALMS by dripping theliquid sample 4 on thesupport surface 813. - The receiving
unit 6 is disposed to admit therein ionizedanalytes 414 that are derived from theliquid sample 4, and that are to be analyzed for mass spectrometric analysis. The receivingunit 6 includes amass analyzer 61 disposed for analyzing the ionizedanalytes 414. Themass analyzer 61 is formed with aconduit 611 that is in air communication with the environment. Thedetector 7 is disposed to receive signals generated by themass analyzer 61 as a result of analyzing theionized analytes 414 so as to generate a mass spectrometric analysis result, i.e., a mass spectrum. - The
electrospray unit 5 includes areservoir 52 for accommodating aliquid electrospray medium 51, a nozzle 53 (in the embodiments of the present invention, thenozzle 53 is a capillary 53a) which is disposed downstream of thereservoir 52, and which is configured to sequentially form liquid drops 511 of theelectrospraymedium 51 thereat, and apump 54 disposed downstream of thereservoir 52 and upstream of thenozzle 53 for drawing theelectrospray medium 51 into thenozzle 53. Thenozzle 53 is spaced apart from themass analyzer 61 of the receivingunit 6 in a longitudinal direction so as to define a traveling path (X). - The
voltage supplying member 3 is disposed to establish between thenozzle 53 of theelectrospray unit 5 and themass analyzer 61 of the receiving unit 6 a potential difference which is of an intensity such that the liquid drops 511 are laden with a plurality of charges, and such that the liquid drops 511 are forced to leave thenozzle 53 as multiple-charged ones for heading toward themass analyzer 61 along the traveling path (X). - In the first preferred embodiment, the
nozzle 53 is made from a metal material, and a firstcentral axis 532 of thenozzle 53 and a second central axis 612 of theconduit 611 in themass analyzer 61 are substantially parallel to each other. Thesupport member 811 of thesample stage 81 extends in the longitudinal direction such that thesupport surface 813 thereof defines a leveled plane in the longitudinal direction. The distance between projections of anoutlet 531 of thenozzle 53 and anentrance 613 into theconduit 611 of themass analyzer 61 on the leveled plane is approximately 8 mm. In addition, when theliquid sample 4 is placed on thesupport surface 813 of thesupport member 81, the shortest distance between theliquid sample 4 and theoutlet 531 of thenozzle 53 is 1.5 mm. - When the
laser transmission mechanism 82 of thelaser desorption device 8 transmits thelaser beam 821 to irradiate theliquid sample 4, upon irradiation, at least one of theanalytes 412 contained in thesolution 41 of theliquid sample 4 is desorbed to fly along a flying path (Y) which intersects the traveling path (X) so as to enable said at least one of theanalytes 412 to be occluded in the multiple-charged liquid drops 511. As a result of dwindling in size of the multiple-charged liquid drops 511 when approaching themass analyzer 61 of the receivingunit 6 along the traveling path (X), charges of the liquid drops 511 will pass on to said at least one of theanalytes 412 to form a corresponding one of the ionizedanalytes 414. Theionized analytes 414 enter themass analyzer 61 via theentrance 613 into theconduit 611 for subsequent mass spectrometric analysis. - With reference to FIG.5, the second preferred embodiment of a mass spectrometer assembly implementing the method of ALMS according to the present invention is similar to the first preferred embodiment. The only difference between the first and second preferred embodiments is that the electrospray unit 5' of the second preferred embodiment further includes an airstream supplying mechanism 55' for accelerating vaporization of the multiple-charged liquid drops 511 (refer to FIGS.2 to 4) to result in dwindling in size thereof when approaching the mass analyzer 61 (refer to FIG.5) along thetravelingpath (X). The airstream supplying mechanism 55' surrounds the
nozzle 53, and supplies a nitrogen airstream 5.51'. In particular, the temperature of the nitrogen airstream 551' can be controlled by the user between the room temperature and 325°C as is required. - As shown in FIG. 6, the third preferred embodiment of a mass spectrometer assembly implementing the method of ALMS according to the present invention is similar to the first preferred embodiment. The difference between the first and third preferred embodiments is that the
nozzle 53" of theelectrospray unit 5" of the third preferred embodiment is made from a non-metal material, and theelectrospray unit 5" further includes a micro-tube 56". The micro-tube 56" includes a tubular body 561" connected between and disposed in fluid communication with thepump 54 and thenozzle 53", and acenter portion 562" connected to the tubular body 561" and coupled to the voltage supplying member 3 (refer to FIG. 4) such that the potential difference is established between the micro-tube 56" and themass analyzer 61 of the receivingunit 6. - Referring to FIG. 7, the fourth preferred embodiment of a mass spectrometer assembly implementing the method of ALMS according to the present invention is similar to the first preferred embodiment. The difference between the first and fourth preferred embodiments is that the sample stage 81'" of the
laser desorption device 8"' includes amovable track 814"', and a support member set 815'" including a plurality ofsupport members 816"' (only one is visible in FIG. 7) connected in sequence and mounted movably on thetrack 814"'. - To conduct mass spectrometric analysis using the mass spectrometer assembly of the fourth preferred embodiment, a plurality of liquid samples 4 (as shown in FIG.4) are first contained in containers 10 (e.g., test tubes or centrifuge tubes) (only one is visible in FIG. 7), respectively. Subsequently, each of the
containers 10 is disposed on a corresponding one of thesupport members 816"'. Through control of a computer software, thesupport members 816"' move along thetrack 814"', carrying theliquid samples 4 thereon, such that theliquid samples 4 are sequentially disposed at a predefined location set by the operator. When each of theliquid samples 4 is disposed at the predefined location, theliquid sample 4 is irradiated by thelaser beam 821 transmitted by thelaser transmission mechanism 82 of thelaser desorption device 8, and subsequent mass spectrometric analysis is conducted. - It should be noted herein that only one
support member 816"' and onecontainer 10 are visible in FIG.7 due to the direction of observation. - Referring to FIG.4, the fifth preferred embodiment of a mass spectrometer assembly implementing the method of ALMS according to the present invention is similar to the first preferred embodiment. The only difference between the first and the fifth preferred embodiments is that the laser transmission mechanism of the fifth preferred embodiment is an infrared (IR)
laser 82c instead of theultraviolet laser 82a as in the first preferred embodiment. - Referring to FIG. 6, the sixth preferred embodiment of a mass spectrometer assembly implementing the method of ALMS according to the present invention is similar to the fourth preferred embodiment. The only difference between the fourth and the sixth preferred embodiments is that the laser transmission mechanism of the sixth preferred embodiment is the infrared (IR)
laser 82c (as shown in FIG.4) instead of theultraviolet laser 82a. - It should be noted herein that each of the components of the mass spectrometer assembly according to the present invention can be designed to be movable so as to permit adjustments of the positions thereof by the user as are required, such that relative positions or distances among the various components of the mass spectrometer assembly can be determined. Similarly, parameters, such as the energy, frequency, incident angle of the laser beam irradiated by the laser transmission mechanism, and the composition and flow rate of the electrospray medium, etc., can be adjusted according to the objectives aimed, so as to obtain optimal detection results.
- Presented hereinbelow are exemplary methods for the method of ALMS according to the present invention, along with a comparative example. In the exemplary methods and the comparative example, the liquid samples and electrospray medium are prepared following a certain proportion, or are obtained directly, under room temperature and atmospheric pressure. If it is not particularly pointed out, the liquid sample includes an aqueous solution, and the composition of the electrospray medium is [water : methanol : acetic acid = 50 : 50 : 0.1], and the flow rate of the electrospray medium is 150 µL per minute.
- Further, if it is not particularly pointed out, the exemplary methods and the comparative example are conducted according to the third preferred embodiment of the present invention. In addition, the mass analyzer conducts the scans with a 2s/scan scanning rate. For each liquid sample presented, the molecular weight of the solvent is excluded from a scanning range of the mass analyzer.
- In
exemplary methods exemplary methods Table 1 Liquid Sample Mass spectrum Analytes Carbon powder Concentration Comparative example 1 myoglobin (10-5 M), cytochrome c (10-5 M), lysozyme (10-5 M) 0 mg/µL FIG. 8(a) Exemplary Method 10.4 mg/µL FIG. 8(b) Exemplary Method 20.8 mg/µL FIG. 8(c) - Since the electrospray medium used does not contain any acid, the applicant predicted that the mass spectra obtained should present the formation of "un-denatured proteins". In other words, the molecular weight of myoglobin resulted from
exemplary methods - It is clearly shown in FIGS. 8(b) and 8(c) that there are three ion peaks, which are respectively denoted by "■", "▲", "•", and whose molecular weights are calculated by a computer software to be 12232 Da, 14306 Da, and 17567 Da, respectively. The calculated molecular weights almost completely correspond to the molecular weights of myoglobin, cytochrome c, and lysozyme as provided by the manufacturer. In addition, it is obvious that the detected myoglobin is in an un-denatured state. The results confirm that the method of ALMS works effectively, and is capable of conducting direct detection on a liquid sample including a protein so as to obtain accurate and satisfactory quantitative results.
- The reason for this success is that, upon irradiation, laser energy of the ultraviolet laser beam is passed on to at least one of the analytes (proteins) contained in the solution of the liquid sample via the matrix material (carbon powders) so that the analyte is successfully desorbed. On the other hand, the liquid sample used in comparative example 1 does not contain carbon powders or any other materials to serve as a matrix, the analytes could not be effectively desorbed from the liquid sample (or the volume of desorbed analytes was too small). Since no or a minimal number of analytes reached and was detected by the mass analyzer for mass spectrometric analysis, corresponding signals for the analytes could not be generated.
- It should be noted herein that the peak shown in FIG. 8 (a) is an interference signal, and is relatively enlarged due to the absence of analyte signals.
- The composition of the liquid sample used, and the figure number of corresponding mass spectrum for each of the
exemplary methods 3 to 8 are tabulated in Table 2 below.Table 2 Liquid Sample Mass Spectrum Material serving as Matrix Solvent Analytes and of Concentration thereof Exemplary Method 3Carbon powder (0.8 mg/µL) Methanol Hemin (2*10-3M) FIG.9(a) Exemplary Method 4THF 18-crown-6-ether (2*10-2M) FIG.9 (b) Exemplary Method 5EA. 1-hexadecylamine (1*10-3M) FIG. 9 (c) Exemplary Method 6Methylene dichloride Methyl (triphenyl-phosphoranylidene) acetate (1*10-2M) FIG. 9(d) Exemplary Method 7Tolene Cinnamic acid benzyl ester (2*10-2M) FIG.9(e) Exemplary Method 8 n-hexane Cetylpyridinium chloride (1*10-4M) FIG. 9(f) - It is clearly observed in FIGS.9(a) to 9(f) corresponding ion peaks formed by analytes of the liquid samples used in
exemplary methods 3 to 8. In addition, the molecular weights obtained after calculation match with the known facts, confirming the operability of the method of ALMS on liquid samples provided with organic solutions and organic compounds. - In an experiment conducted by the applicant, a linear equation obtained through linear regression analysis demonstrates that glycosylated hemoglobin/ hemoglobin (HbA1/Hb) values of in blood of a diabetes patient obtained using ALMS analysis has a specific relationship with those obtained using ionic chromatography, which is a currently common method used in the medical field for obtaining the quantities of Hb and HbA1. Therefore, the (HbA1/Hb) values obtained using ALMS analysis should have a certain degree of credibility and reference value. In particular, it is reported that it takes approximately an hour, including preparation work on the samples, to conduct analysis using ionic chromatography. On the other hand, instantaneous detection and result can be obtained using ALMS analysis. Therefore, the method of ALMS should have the potential of replacing the method of ionic chromatography in providing the basis for diagnoses of diseases.
- In addition, it is also verified by experiment that a highly credible mass spectrometric analysis result can be obtained when a liquid sample includes an aqueous solution, even if it is a body fluid with complicated composition and containing a large quantity of salts, after a simple diluting step, rapid and convenient analysis can be conducted using the method of ALMS, without adding an additional matrix material, by irradiating an infrared laser beam on the liquid sample. In this case, the "water molecules" contained in the aqueous solution serve as the "matrix" for transferring the laser energy to the analytes such that the analytes are desorbed and enter the mass analyzer for subsequent mass spectrometric analysis.
- With reference to the results described hereinabove with respect to the exemplary methods and the comparative example, it is shown that the present invention is in deed capable of performing rapid and accurate mass spectrometric analysis directly on a liquid sample. In addition, no specific restriction is imposed on the sample to be analyzed, i.e., whether it is a body fluid with a complicated composition, or an organic solution, a protein solution, etc., qualitative information about the contents therein can be obtained through the method of ALMS according to the present invention. Moreover, other than qualitative information, relative quantitative information on various analytes in a liquid sample, such as compositional proportions of the analytes in the liquid sample, can also be reflected through the use of ALMS analysis. It is of special importance that when a liquid sample includes an aqueous solution, by irradiating the liquid sample with infrared laser, satisfactory detection results can be obtained through ALMS analysis.
- In addition, a mass spectrometer assembly implementing the method of ALMS should be capable of being connected in series to other analytic instruments. A high performance liquid chromatograph (HPLC) is taken as an example hereinbelow for illustration. When a biochemical sample (normally including an aqueous solution) is eluted after passing through the HPLC, ALMS analysis can be conducted by irradiating laser on the eluted sample when it is disposed between the electrospray unit and the mass analyzer of the mass spectrometer assembly implementing the method of ALMS.
- In sum, since.the method of ALMS according to the present invention is conducted directly under atmospheric pressure, instead of vacuum, and since operation time needed is extremely short, the cost of instrumentation for implementing the present invention, the technical requirements for manufacturing such instrumentation and for operation of such method have all greatly reduced as compared to matrix-assisted laser desorption ionization mass spectrometry (MALDI-MS) of the prior art. Further, it has been verified that the method of ALMS according to the present invention can be used to analyze various kinds of liquid samples, including protein aqueous solutions, body fluids, and organic solutions containing organic compounds, etc., can all be analyzed directly (with minimal sample preparation), as opposed to making the originally liquid samples into solid samples. In addition, satisfactory results can be obtained both for qualitative analysis (i.e., the determination of the identity of the analytes detected) and relative quantitative analysis (i.e., the quantity of various kinds of analytes contained in the liquid sample).
- Due to the convenience and speed of the method of ALMS according to the present invention, and immediate results obtainable through use of such method, it is evident that the present invention is advantageous in related fields, where qualitative analysis of analytes in a large quantity of liquid samples or determination of relative concentrations of analytes in liquid samples is required, such as in medical fields, environmental examination, criminal judgment, academic research, etc.
- The method of ALMS according to the present invention can also be applied to the analysis of a body fluid secreted by an organism. Through identities and relative concentrations of substances in an organism' s body fluid, the biological condition of the organism can be determined.
- Moreover, the mass spectrometer assembly implementing the method of ALMS according to the present invention can be connected in series to other analytic instruments, such as a high performance liquid chromatograph (HPLC), so that an operator can conduct ALMS analysis so as to obtain information on the substances contained in the sample in sequence with conducting sample purification. This greatly enhances operational convenience and greatly reduces operational time when several analyses need to be conducted on identical samples.
- While the present invention has been described in connection with what are considered the most practical and preferred embodiments, it is understood that this invention is not limited to the disclosed embodiments but is intended to cover various arrangements included within the spirit and scope of the broadest interpretation and equivalent arrangements.
Claims (10)
- A mass spectrometer assembly comprising:a receiving unit disposed to admit therein ionized analytes that are derived from a liquid sample, and including a mass analyzer disposed for analyzing the ionized analytes; andan electrospray unit including a reservoir for accommodating a liquid electrospray medium, and a nozzle which is disposed downstream of said reservoir, and which is configured to sequentially form a liquid drop of said electrospray medium thereat, said nozzle being spaced apart from said receiving unit in a longitudinal direction so as to define a traveling path;a voltage supplying member disposed to establish between said nozzle and said receiving unit a potential difference which is of an intensity such that the liquid drop is laden with a plurality of charges, and such that the liquid drop is forced to leave said nozzle as a multiple-charged one for heading toward said receiving unit along the traveling path; anda laser desorption device includinga sample stage on which the liquid sample is placed, the liquid sample including a solution that contains the analytes and a material serving as a matrix for absorbing laser energy; anda laser transmission mechanism disposed to irradiate the liquid sample such that, upon irradiation, laser energy is passed on to at least one of the analytes contained in the solution of the liquid sample via the matrix so that said at least one of the analytes is desorbed to fly along a flying path which intersects the traveling path of the multiple-charged liquid drops of said electrospray medium so as to enable said at least one of the analytes to be occluded in said multiple-charged liquid drops, and such that as a result of dwindling in size of the multiple-charged liquid drops when approaching said receiving unit from said nozzle of said electrospray unit along the traveling path, charges of the liquid drops will pass on to said at least one of the analytes occluded therein to form a corresponding one of the ionized analytes.
- The mass spectrometer assembly as claimed in Claim 1, wherein the solution of the liquid sample is an aqueous solution, the material serving as the matrix being water molecules contained in the aqueous solution, said laser transmission mechanism being an infrared laser.
- The mass spectrometer assembly as claimed in Claim 1, wherein said sample stage of said laser desorption device includes a movable track, and a support member having the liquid sample disposed thereon, and mounted movably on said track such that the liquid sample moves with said supporting member along said track.
- The mass spectrometer assembly as claimed in Claim 1, wherein said sample stage of said laser desorption device includes a support member that is made from a material non-transmissible by laser, and that has a support surface for placement of the liquid sample directly thereon.
- A method for mass spectrometry, comprising the steps of:placing, on a sample stage, a liquid sample including a solution that contains a plurality of analytes and a material serving as a matrix for absorbing laser energy;providing an electrospray unit that includes a nozzle configured to sequentially form liquid drops of an electrospray medium thereat;providing a receiving unit that is disposed to admit therein ionized analytes that are derived from the liquid sample, and that are to be analyzed by a mass analyzer disposed downstream of the receiving unit, the receiving unit being spaced apart from the nozzle of the electrospray unit in a longitudinal direction so as to define a traveling path;establishing a potential difference between the nozzle of the electrospray unit and the receiving unit, the potential difference being of an intensity such that the liquid drops are laden with a plurality of charges, and such that the liquid drops are forced to leave the nozzle as multiple-charged ones for heading toward the receiving unit along the traveling path; andirradiating the liquid sample with a laser beam such that, upon irradiation, laser energy is passed on to at least one of the analytes contained in the solution of the liquid sample via the matrix so that said at least one of the analytes contained in the liquid sample is desorbed to fly along a flying path which intersects the traveling path so as to enable said at least one of the analytes to be occluded in the multiple-charged liquid drops, and such that as a result of dwindling in size of the multiple-charged liquid drops when approaching the receiving unit along the traveling path, charges of the liquid drops will pass on to said at least one of the analytes occluded therein to form a corresponding one of the ionized analytes.
- The method as claimed in Claim 5, wherein the solution is an aqueous solution, the material serving as the matrix being water molecules contained in the aqueous solution, the laser beam being infrared laser beam.
- The method as claimed in Claim 5, wherein the material serving as the matrix is made from a material that is non-transmissible by laser.
- The method as claimed in Claim 5, wherein the solution included in the liquid sample includes a body fluid secreted by an organism.
- The method as claimed in Claim 11, wherein the body fluid is selected from the group consisting of blood, tear, milk, perspiration, intestinal juice, brains fluid, spinal fluid, lymph, pus, blood serum, saliva, nasal mucus, urine, and excrement.
- The method as claimed in Claim 5, wherein the solution included in the liquid sample is a protein solution.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNA200610142595XA CN101173914A (en) | 2006-10-30 | 2006-10-30 | Atmospheric pressure liquid phase mass spectrometric analysis method and atmospheric pressure liquid phase mass spectrograph |
| US11/746,282 US20080116366A1 (en) | 2006-11-17 | 2007-05-09 | Laser desorption device, mass spectrometer assembly, and method for ambient liquid mass spectrometry |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1918974A2 true EP1918974A2 (en) | 2008-05-07 |
| EP1918974A3 EP1918974A3 (en) | 2010-03-24 |
Family
ID=39060250
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP07119671A Withdrawn EP1918974A3 (en) | 2006-10-30 | 2007-10-30 | Mass spectrometer assembly and method for ambient liquid mass spectrometry |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP1918974A3 (en) |
| JP (2) | JP2008147165A (en) |
| RU (1) | RU2007140232A (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2287600A1 (en) * | 2008-05-20 | 2011-02-23 | Shimadzu Corporation | Atmospheric pressure ionization mass spectrometer |
| EP2296791A1 (en) * | 2008-05-12 | 2011-03-23 | Shimadzu Corporation | "droplet pickup ion source" coupled to mobility analyzer apparatus and method |
| US8835179B2 (en) | 2010-10-29 | 2014-09-16 | Scinopharm Taiwan, Ltd. | Real-time monitor solid phase peptide synthesis by mass spectrometry |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| HU226837B1 (en) | 2006-05-31 | 2009-12-28 | Semmelweis Egyetem | Desorption ionization method and device operated by liquid stream |
| CA2763145C (en) | 2009-05-27 | 2021-05-25 | Medimass Kft | System and method for identification of biological tissues |
| US9281174B2 (en) | 2011-12-28 | 2016-03-08 | Micromass Uk Limited | System and method for rapid evaporative ionization of liquid phase samples |
| CN104254901B (en) | 2011-12-28 | 2018-05-04 | 英国质谱有限公司 | Collide ion generator and separator |
| JP6278223B2 (en) * | 2014-10-20 | 2018-02-14 | 株式会社島津製作所 | Atmospheric pressure ionizer |
| JP6593689B2 (en) * | 2015-08-27 | 2019-10-23 | 高知県公立大学法人 | Matrix for MALDI mass spectrometry, method for producing the same, and mass spectrometry using the same |
| WO2017221846A1 (en) * | 2016-06-24 | 2017-12-28 | 株式会社住化分析センター | Method for evaluating physical structure |
| KR102062447B1 (en) * | 2018-03-13 | 2020-01-03 | 가천대학교 산학협력단 | Target surfaces for MALDI mass spectrometry using Graphene films and Mass analysis method using the same |
| US11532472B2 (en) * | 2020-07-30 | 2022-12-20 | Ricoh Company, Ltd. | Mass spectrometry |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6849847B1 (en) * | 1998-06-12 | 2005-02-01 | Agilent Technologies, Inc. | Ambient pressure matrix-assisted laser desorption ionization (MALDI) apparatus and method of analysis |
| US20050056776A1 (en) * | 2000-06-09 | 2005-03-17 | Willoughby Ross C. | Laser desorption ion source |
| US20050199823A1 (en) * | 2004-01-20 | 2005-09-15 | Bruker Daltonik | Desorption and ionization of analyte molecules at atmospheric pressure |
| US20070176113A1 (en) * | 2006-01-27 | 2007-08-02 | Jentaie Shiea | Electrospray-assisted laser desorption ionization device, mass spectrometer, and method for mass spectrometry |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1876442A3 (en) * | 1998-09-17 | 2008-03-05 | Advion BioSciences, Inc. | Integrated monolithic microfabricated liquid chromatography system and method |
| DE10144250A1 (en) * | 2001-08-31 | 2003-04-03 | Fraunhofer Ges Forschung | Improved mass spectrometric analysis using nanoparticles |
| GB0128586D0 (en) * | 2001-11-29 | 2002-01-23 | Amersham Biosciences Ab | Graphite anchor targets |
| JP2005243466A (en) * | 2004-02-26 | 2005-09-08 | Japan Science & Technology Agency | Laser ionization device and its utilization |
| JP4366508B2 (en) * | 2004-03-30 | 2009-11-18 | 国立大学法人山梨大学 | Ionization method and apparatus for mass spectrometry |
-
2007
- 2007-09-28 JP JP2007253798A patent/JP2008147165A/en active Pending
- 2007-10-30 EP EP07119671A patent/EP1918974A3/en not_active Withdrawn
- 2007-10-30 RU RU2007140232/28A patent/RU2007140232A/en not_active Application Discontinuation
-
2013
- 2013-07-11 JP JP2013145875A patent/JP5707630B2/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6849847B1 (en) * | 1998-06-12 | 2005-02-01 | Agilent Technologies, Inc. | Ambient pressure matrix-assisted laser desorption ionization (MALDI) apparatus and method of analysis |
| US20050056776A1 (en) * | 2000-06-09 | 2005-03-17 | Willoughby Ross C. | Laser desorption ion source |
| US20050199823A1 (en) * | 2004-01-20 | 2005-09-15 | Bruker Daltonik | Desorption and ionization of analyte molecules at atmospheric pressure |
| US20070176113A1 (en) * | 2006-01-27 | 2007-08-02 | Jentaie Shiea | Electrospray-assisted laser desorption ionization device, mass spectrometer, and method for mass spectrometry |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2296791A1 (en) * | 2008-05-12 | 2011-03-23 | Shimadzu Corporation | "droplet pickup ion source" coupled to mobility analyzer apparatus and method |
| EP2296791A4 (en) * | 2008-05-12 | 2014-01-08 | Shimadzu Corp | "droplet pickup ion source" coupled to mobility analyzer apparatus and method |
| EP2287600A1 (en) * | 2008-05-20 | 2011-02-23 | Shimadzu Corporation | Atmospheric pressure ionization mass spectrometer |
| EP2287600A4 (en) * | 2008-05-20 | 2014-01-08 | Shimadzu Corp | Atmospheric pressure ionization mass spectrometer |
| US8835179B2 (en) | 2010-10-29 | 2014-09-16 | Scinopharm Taiwan, Ltd. | Real-time monitor solid phase peptide synthesis by mass spectrometry |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5707630B2 (en) | 2015-04-30 |
| JP2013257332A (en) | 2013-12-26 |
| RU2007140232A (en) | 2009-05-10 |
| EP1918974A3 (en) | 2010-03-24 |
| JP2008147165A (en) | 2008-06-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1918974A2 (en) | Mass spectrometer assembly and method for ambient liquid mass spectrometry | |
| US7696475B2 (en) | Electrospray-assisted laser desorption ionization device, mass spectrometer, and method for mass spectrometry | |
| US20080116366A1 (en) | Laser desorption device, mass spectrometer assembly, and method for ambient liquid mass spectrometry | |
| US11867684B2 (en) | Sample dispenser including an internal standard and methods of use thereof | |
| US11430648B2 (en) | System and methods for ionizing compounds using matrix-assistance for mass spectrometry and ion mobility spectrometry | |
| EP3633710B1 (en) | Ion generation using wetted porous material | |
| Takats et al. | Ambient mass spectrometry using desorption electrospray ionization (DESI): instrumentation, mechanisms and applications in forensics, chemistry, and biology | |
| Liu et al. | Development, characterization, and application of paper spray ionization | |
| US7718958B2 (en) | Mass spectroscopic reaction-monitoring method | |
| EP1741120B1 (en) | Method and system for desorption electrospray ionization | |
| US7847244B2 (en) | Enclosed desorption electrospray ionization | |
| US7968842B2 (en) | Apparatus and systems for processing samples for analysis via ion mobility spectrometry | |
| EP3486937B1 (en) | Mass spectrometry analysis of microorganisms in samples | |
| US8445842B2 (en) | Mechanical holder for surface analysis | |
| US7687772B2 (en) | Mass spectrometric imaging method under ambient conditions using electrospray-assisted laser desorption ionization mass spectrometry | |
| CN101173914A (en) | Atmospheric pressure liquid phase mass spectrometric analysis method and atmospheric pressure liquid phase mass spectrograph | |
| EP3465138B1 (en) | Systems and methods for analyzing an analyte extracted from a sample using an adsorbent material | |
| Walles et al. | Approaches for coupling solid-phase microextraction to nanospray | |
| Zhou et al. | Direct solid-phase microextraction-mass spectrometry facilitates rapid analysis and green analytical chemistry | |
| Zhu et al. | High-throughput screening using a synchronized pulsed self-aspiration vacuum electrospray ionization miniature mass spectrometer | |
| CN110931342A (en) | Photoelectron and ion image energy spectrum device based on liquid beam sampling | |
| Abu‐Rabie | Direct analysis of dried blood spot samples | |
| EP1193730A1 (en) | Atmospheric-pressure ionization device and method for analysis of a sample | |
| WO2023073587A1 (en) | Ultra high throughput screening combined with definitive testing in a single sample preparation step | |
| CN117413339A (en) | Analyzer system |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC MT NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA HR MK RS |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC MT NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA HR MK RS |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: H01J 49/14 20060101ALI20100215BHEP Ipc: H01J 49/16 20060101ALI20100215BHEP Ipc: H01J 49/04 20060101AFI20100215BHEP |
|
| AKY | No designation fees paid | ||
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: 8566 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 20100925 |