US7294125B2 - Methods of delivering energy to body portions to produce a therapeutic response - Google Patents
Methods of delivering energy to body portions to produce a therapeutic response Download PDFInfo
- Publication number
- US7294125B2 US7294125B2 US10/887,324 US88732404A US7294125B2 US 7294125 B2 US7294125 B2 US 7294125B2 US 88732404 A US88732404 A US 88732404A US 7294125 B2 US7294125 B2 US 7294125B2
- Authority
- US
- United States
- Prior art keywords
- energy
- ultrasound
- tissue
- sphincter
- esophageal sphincter
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related, expires
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N7/00—Ultrasound therapy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N7/00—Ultrasound therapy
- A61N7/02—Localised ultrasound hyperthermia
Definitions
- the invention relates to methods of applying energy to various body portions to produce a therapeutic response.
- the applied energy is ultrasound and may be delivered by an ultrasound focusing device deployed in the body, for example, by using an endoscope.
- the energy may be used to effect a variety of therapeutic responses, including ablating lesions in the gall bladder or other organs, treating gastrointestinal reflux disease, or treating fecal incontinence.
- Minimizing the invasiveness of medical procedures is desirable. Generally, more invasive procedures are more expensive and result in more complications and more discomfort to the patient. For example, open surgery is an invasive medical procedure with significant attendant risks. Due to relatively large incisions necessary in the performance of open surgery, relatively large amounts of blood may be lost, the risk of infection increases, and the potential for post-operative hernias is high. Further, the relatively large incisions necessary in the performance of open surgery require extended recovery times for the incisions to heal.
- Laparoscopic procedures are less invasive than open surgery. Laparoscopic procedures often involve performing laparotomies for trocar ports (penetrations of the abdominal walls), percutaneous endoscopic gastronomies (incisions through the skin into the various bodily organs), and the installation of ports through which medical devices are inserted. This procedure also requires incisions through, for example, the abdominal walls, risking infection. The location of the incision in the bodily wall also presents a risk of other negative effects, such as sepsis, which can be caused by leakage of septic fluid contained in the stomach.
- Medical treatments that would benefit from less invasive procedures include, for example, vascular sealing, ablating lesions in the gall bladder or other body organs, treatment of gastrointestinal reflux disease, or treatment of fecal incontinence, among others.
- Vascular sealing is often necessary in medical procedures that access the vascular system of the patient.
- various means may be used to obtain access into a vein or artery, typically access is obtained by inserting a cannula or introducer sheath through the skin and into the selected blood vessel.
- a medical device or diagnostic instrument such as a guide wire, guiding catheter, balloon angioplasty device, atherectomy device, or the like, then is inserted into the vascular system through the cannula or introducer sheath.
- the introducer sheath must be of a relatively large diameter. Introducer sheaths typically have a diameter in the range between one millimeter and six millimeters, thus creating a significant puncture in the blood vessel, such as a puncture 17 into a vessel 10 as shown in FIGS. 1A and 1B .
- puncture 17 After the intravascular medical procedure is completed, puncture 17 must be closed and bleeding from the blood vessel 10 stopped. In the examples shown in FIGS. 1A and 1B , the puncture 17 goes through the subcutaneous tissue 13 , the vascular sheath 12 , and the vessel wall 11 .
- the vessel wall includes the adventitia 16 , media layer 15 , and intima layer 14 , as shown in FIGS. 1C and 1D .
- bleeding may be stopped by the application of direct digital pressure over the puncture by a trained physician or other suitably trained medical personnel.
- direct pressure must be applied for a sufficiently long time for hemostasis to occur so that the puncture is effectively closed and further bleeding is prevented.
- the pressure is generally applied for twenty to thirty minutes, but it may be necessary to apply pressure for longer and/or through other devices that apply pressure.
- Stopping bleeding using digital pressure is not an efficient use of medical professional services.
- the procedure also results in a substantial reduction, if not virtual arrest, of blood flow through the vessel. Since thrombosis is one of the major problems that can occur in the immediate post-operative period, reduction in blood flow caused by the application of digital pressure is undesirable.
- digital pressure is applied, an undesirable bruise or hematoma can form at the entry site, since internal bleeding of the punctured artery continues until clotting blocks the puncture. There is also a risk that upon movement by the patient, the puncture will reopen and begin bleeding again, resulting in a hematoma or other complications.
- the introducer sheath is generally left inside the patient for a relatively long period of time in order for the anticoagulants to clear from the blood. Because the patient may be required to remain immobile and because of the risk of complications, patients are usually required to remain overnight in the hospital for observation, increasing the cost of the overall procedure.
- Ablating portions of a gall bladder, or other bodily organs, is sometimes necessary to treat and/or destroy cancerous or noncancerous lesions that may exist on the surface or in the interior of the gall bladder, such as a lesion 51 in gall bladder 50 shown in FIG. 2 . Removal of these lesions is desirable to prevent them from spreading and/or infecting additional portions of the gall bladder, as even lesions in a noncancerous form may interfere with the proper functioning of the gall bladder.
- Gastroesophageal reflux occurs when stomach acid enters the esophagus from the stomach. (See a portion of the gastrointestinal tract, as shown in FIGS. 3A and 3B , that depicts esophagus 33 and stomach 30 ). This reflux of acid into the esophagus occurs naturally in healthy individuals, but also may become a pathological condition in others. Effects from gastroesophageal reflux range from mild to severe. Mild effects include heartburn, a burning sensation experienced behind the breastbone.
- More severe effects include a variety of complications, such as esophageal erosion, esophageal ulcers, esophageal stricture, abnormal epithelium (e.g., Barrett's esophagus), and/or pulmonary aspiration.
- GSD Gastroesophageal Reflux Disease
- LES lower esophageal sphincter
- the LES is a ring of smooth muscle and increased annular thickness existing in approximately the last four centimeters of the esophagus.
- the LES creates a region of high pressure (approximately 15-30 mm Hg above intragastric pressure) at the opening of the esophagus into the stomach. This pressure essentially closes the esophagus so that contents of the stomach cannot pass back into the esophagus.
- the LES opens in response to swallowing and peristaltic motion in the esophagus, allowing food to pass into the stomach. After opening, however, a properly functioning LES should return to the resting, or closed state. Transient relaxations of the LES do occur in healthy individuals, typically resulting in occasional bouts of heartburn.
- the physical interaction occurring between the gastric fundus and the esophagus also prevents gastroesophageal reflux.
- the gastric fundus shown as element 35 in FIGS. 3A and 3B , is a lobe of the stomach situated at the top of the stomach distal to the esophagus. In asymptomatic individuals, the fundus presses against the opening of the esophagus when the stomach is full of food and/or gas. This effectively closes off the esophageal opening to the stomach and helps to prevent acid reflux back into the esophagus.
- the food bolus As the food bolus is immersed in gastric acid, it releases gas which causes the fundus of the stomach to expand and thereby exert pressure on the distal esophagus causing it to collapse.
- the collapse of the esophagus lumen reduces the space for the stomach acid to splash past the closed esophagus lumen and thereby protect the proximal esophagus from its destructive contact.
- the LES functions abnormally, either due to an increase in transient LES relaxations, decreased muscle tone of the LES during resting, or an inability of the esophageal tissue to resist injury or repair itself after injury. These conditions often are exacerbated by overeating, intake of caffeine, chocolate or fatty foods, smoking, and/or hiatal hernia. Avoiding these exacerbating mechanisms helps curb the negative side effects associated with GERD, but does not change the underlying disease mechanism. Accordingly, some type of medical treatment is often necessary to correct, or at least improve, this condition.
- a surgical procedure known generally as fundoplication, has been developed to prevent acid reflux in patients whose normal LES functioning has been impaired, either as a result of GERD or other adverse effects.
- This procedure involves bringing the fundus wall into closer proximity of the esophageal wall to help close off the esophageal opening into the stomach.
- this procedure has been performed as an open surgery, but also has been performed laparoscopically.
- fundoplication involves certain attendant risks.
- an alternative, minimally invasive technique is preferable.
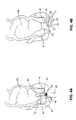
- Fecal incontinence which is most common in the elderly, is the loss of voluntary control to retain stool in the rectum, shown as element 40 in FIGS. 4A and 4B . In most cases, fecal incontinence is the result of an impaired involuntary internal anal sphincter 41 .
- the internal sphincter may be incompetent due to laxity or discontinuity. Discontinuity, or disruption of the internal anal sphincter, can be caused by a number of different muscle injuries.
- Biofeedback is successful in approximately two-thirds of patients who retain some degree of rectal sensation and functioning of the external anal sphincter 42 .
- multiple sessions are often necessary, and patients need to be highly motivated.
- Electronic home biofeedback systems are available and may be helpful as adjunctive therapy.
- Patients may have mechanical devices implanted in their body, for example, to treat cancer, or mechanical devices placed in the body to perform a treatment.
- mechanical devices that may be implanted in the body or placed in the body to perform a treatment include stents, grafts, needles, and knives. Some of these mechanical devices may, once implanted or placed in the body, require activation.
- One method of activating these mechanical devices is to run a wire from the mechanical device to the exterior of the patient's body, and then apply energy to the mechanical device through the wire.
- running wires through the body for prolonged periods of time are especially undesirable as it may lead to medical complications such as internal hemorrhaging. Accordingly, a less invasive way of activating the mechanical devices is desired.
- an embodiment of the invention includes a method of. treating a patient with gastrointestinal reflux disease by applying energy to an esophageal sphincter to produce a therapeutic response.
- an embodiment of the invention includes a method of treating a patient with fecal incontinence by applying energy to an anal sphincter to produce a therapeutic response.
- an embodiment of the invention includes a method of ablating tissue in a gall bladder by applying energy to the gall bladder to produce a therapeutic response.
- the applied energy is ultrasound, and the amount of applied energy may be adjusted as desired.
- an endoscope with a working channel is inserted into the body, and an ultrasound device is advanced through the working channel to the desired body portion.
- the energy is applied via an ultrasound device.
- the energy is applied below the surface of the desired body portion.
- the energy from the ultrasound device is focusing on the desired body portion.
- the ultrasound device is positioned outside of the patient.
- FIG. 1A is a schematic view of an exemplary method of sealing vascular punctures.
- FIG. 1B is a schematic view of another exemplary method of sealing vascular punctures.
- FIG. 1C is a schematic view of a sealed vascular puncture resulting from the method shown in FIG. 1A .
- FIG. 1D is a schematic view of a sealed vascular puncture resulting from the method shown in FIG. 1B .
- FIG. 2A is a schematic view of an exemplary method of ablating a gall bladder.
- FIG. 2B is a schematic view of another exemplary method of ablating a gall bladder.
- FIG. 3A is a schematic view of an exemplary method of treating GERD.
- FIG. 3B is a schematic view of another exemplary method of treating GERD.
- FIG. 4A is a schematic view of an exemplary method of treating fecal incontinence.
- FIG. 4B is a schematic view of another exemplary method of treating fecal incontinence.
- FIG. 5A is a schematic view of a method of activating a mechanical implant.
- FIG. 5B is. a schematic view of another method of activating a mechanical implant.
- Ultrasound may be used to minimize the invasiveness of medical procedures to treat the body.
- Ultrasound can provide therapeutic responses to internal and external body portions without the need for cuts or incisions, and can act on tissue below the surface of the tissue or organ.
- An ultrasound device that may be used in medical procedures is an ultrasound transducer or ultrasound focusing device disclosed in U.S. patent application Publication No. 2001/0031922 A1 to Weng et al. (“Weng”), the complete disclosure of which is incorporated herein by reference. Weng discloses using the ultrasound transducer for imaging and therapeutic purposes.
- imaging purposes for example, imaging internal human organs for diagnostic purposes
- ultrasound in the frequency range of 2-20 MHz and/or an intensity of less that 0.1 watts per square centimeter is used.
- a much more high intensity ultrasound above 200 watts per square centimeter is used.
- Such high intensity ultrasound can raise the temperature of a desired tissue region to above 60 degrees Celsius in a few seconds.
- FIGS. 1A-1D show the sealing of blood vessel punctures through the use of ultrasound 22 , for example, by using the ultrasound focusing device 20 disclosed in Weng.
- ultrasound focusing device 20 is positioned proximate the blood vessel puncture 17 . If the blood vessel puncture 17 is close enough to the surface of the skin, the ultrasound focusing device 20 may be placed outside of the body, as shown in FIG. 1A .
- a catheter 24 with a lumen 25 may be advanced through the cardiovascular system until it is proximate the blood vessel puncture 17 , and then the ultrasound focusing device 20 is advanced through the lumen 25 of catheter 24 until it also is proximate the blood vessel puncture 17 , as shown in FIG. 1B .
- the ultrasound focusing device 20 is preferably past the distal end of catheter 24 .
- Ultrasound focusing device 20 then is positioned, configured, and calibrated such that the focal point 23 of the ultrasound 22 will be centered on the portion of the puncture 17 to be sealed.
- Methods of positioning, configuring, and calibrating the ultrasound focusing device 20 for example by adjusting an ultrasound array 21 , are disclosed in Weng, the disclosure of which is incorporated by reference herein.
- the geometry and/or positioning of the ultrasound focusing device 20 can be adjusted so that the focal point 23 will be at a proper distance from the ultrasound focusing device 20 , particularly if the desired focal point 23 is below the surface of the subcutaneous tissue 13 and/or blood vessel wall 11 , as shown in FIGS. 1B and 1D .
- the desired focal point may also be on a surface of the tissue 13 or blood vessel wall 11 , as shown in FIGS. 1A and 1C .
- the geometry and positioning of the ultrasound focusing device 20 can also be adjusted such that it will only act on a specified volume of tissue at and/or surrounding the focal point 23 .
- Both the focal point 23 and the volume of tissue to be energized can be adjusted at any time during the procedure, for example, prior to, during, and/or subsequent to the application of energy to various tissue portions (i.e. any or all of the subcutaneous tissue 13 , vascular sheath 12 , adventitia 16 , media layer 15 , or intima layer 14 ) via the ultrasound focusing device 20 .
- the ultrasound focusing device 20 is activated such that energy is applied via ultrasound 22 to the tissue at the desired focal point 23 .
- the amount of energy supplied by the ultrasound focusing device 20 to the desired focal point 23 is rapidly and significantly increased such that the temperature of the tissue at the focal point 23 rises very rapidly.
- energy in the frequency range of 10 kHz to 300 GHz may be applied to the tissue for between 1 and 10 seconds such that the temperature of the tissue at the focal point 23 rises to over 60 degrees Celsius.
- the frequency may be above 25 kHz.
- the rapid rise in temperature causes the tissue at the focal point 23 , for example the adventitia 16 and the vascular sheath 12 , to denature so that the puncture 17 is closed, and then fuse so that the puncture 17 is sealed. In effect, the tissue is cauterized so as to effect sealing, as shown in FIGS. 1C and 1D .
- the heat generated at the focal point 23 may cause thrombosis and/or coagulate the blood around the focal point 23 and/or in the vascular puncture 17 , which may further facilitating the vascular sealing.
- FIGS. 2A and 2B disclose exemplary methods of ablating a portion of the gall bladder 50 during inside-out surgery through the use of ultrasound 22 , for example, by using the ultrasound focusing device 20 disclosed in Weng.
- Inside-out surgery is the performance of surgery from the inside of the body portion (e.g. without making any cuts or incisions in that body portion).
- an ultrasound focusing device 20 is positioned proximate the gall bladder 50 .
- an endoscope 26 with a working channel 27 is advanced through the gastrointestinal system until it is proximate the gall bladder 50 .
- the endoscope 26 is advanced through the gastrointestinal tract due to the tract's close proximity to the gall bladder 50 , however, advancing an endoscope or other access device through any body lumen that provides access to the gall bladder 50 is also contemplated. Then, the ultrasound focusing device 20 is advanced through the working channel. 27 of the endoscope 26 until it is distal the end of endoscope 26 and proximate the gall bladder 50 , as shown in FIG. 2A . In another example, however, the ultrasound focusing device 20 may be positioned external to the body such that it is proximate the gall bladder 50 , as shown in FIG. 2B .
- the ultrasound focusing device 20 is then positioned, configured, and calibrated such that the focal point 23 of the ultrasound 22 will be centered on the portion of the gall bladder 50 with the lesion 51 , for example, using methods disclosed in Weng for the ultrasound array 21 .
- the geometry and or positioning of the ultrasound focusing device 20 can be adjusted so that the focal point 23 will be at a proper distance from the ultrasound focusing device 20 , as in this case the lesion 51 is either inside the gall bladder 50 or on the gall bladder's surface.
- the geometry and positioning of the ultrasound focusing device 20 can also be adjusted such that it will only act on a specified volume of tissue at and/or surrounding the focal point 23 (i.e.
- Both the focal point 23 and the volume of tissue can be adjusted at any time during the procedure, for example, prior to, during, and/or subsequent to the application of energy.
- the ultrasound focusing device 20 is activated such that energy is applied via ultrasound 22 to the lesion 51 or at least a portion of the lesion 51 .
- the amount of energy supplied by the ultrasound focusing device 20 to the lesion 51 at the desired focal point 23 is rapidly and significantly increased such that the temperature of the lesion tissue 51 rises very rapidly.
- energy in the frequency range of 10 kHz to 300 GHz may be applied to the tissue for between 1 and 10 seconds such that the temperature of the tissue at the focal point 23 rise to over 60 degrees Celsius.
- the frequency may be above 25 kHz.
- the rapid rise in temperature causes almost immediate tissue ablation and/or tissue necrosis of at least a portion of the lesion 51 , and effectively destroys the tissue. If the entire lesion 51 has not been destroyed at this point, the position and/or volume of the focal point 23 of the ultrasound focusing device 20 can be shifted around, for example by adjusting the ultrasound array 21 , so as to destroy other portions of the lesion for which destruction is desired. Such shifting of the position and/or volume of the focal point 23 may occur while the energy is still being applied, or during periods where energy is not applied. In some cases, however, destruction of the entire lesion 51 may not be desired, as destroying all of the lesion 51 may also cause the destruction of healthy tissue.
- the surrounding healthy tissue should be relatively unaffected as only the energy from the ultrasound 22 converging at the focal point 23 should have enough magnitude to affect the tissue in any significant way.
- the ultrasound focusing device 20 and endoscope 26 can be removed.
- the healthy gall bladder tissue 50 should absorb the destroyed tissue and/or remnants of the lesion 51 .
- FIGS. 3A and 3B depict exemplary methods of treating gastrointestinal reflux disease (GERD) through the use of ultrasound 22 , for example, by using the ultrasound focusing device 20 disclosed in Weng.
- an endoscope 26 may be advanced through the esophagus 33 until its distal end is proximate the LES 34 .
- the ultrasound focusing device 20 is then advanced through the working channel 27 of the endoscope 26 until it also is proximate the LES 34 , as shown in FIG. 3A .
- the ultrasound focusing device 20 may be positioned external to the body such that it is proximate the LES 34 , as shown in FIG. 3B .
- ultrasound focusing device 20 is then adjusted, for example as described above in other ultrasound applications, such that the focal point 23 of the device 20 is targeted at the desired tissue portion, for example by adjusting the ultrasound array 21 using methods disclosed above.
- ultrasound focusing device 20 can be used to induce hyperplasia in the esophageal wall 36 , and particularly to the LES 34 , to help close off the esophageal opening into the stomach and/or to accommodate the proper placement of the diaphragm 31 relative to the LES 34 .
- the ultrasound focusing device can also be used to heat the collagen in the esophagus 33 , specifically the esophageal wall 36 , which will cause the lumen of the esophagus 33 to shrink. This will also help close off the esophageal opening into the stomach and/or accommodate the proper placement of the diaphragm 31 relative to the LES 34 .
- the ultrasound focusing device 20 For hyperplasia, once the ultrasound focusing device 20 , for example the ultrasound array 21 , has been positioned and/or adjusted, ultrasound energy is applied to the tissue portion in sufficient amounts to cause injury to the desired tissue portion.
- the surrounding tissue should be relatively unaffected as only the energy from the ultrasound 22 converging at the focal point 23 should have enough magnitude to affect the desired tissue in any significant way.
- the ultrasound focusing device 20 is either moved to another body portion, or completely removed from the body. The injured tissue and the surrounding tissue of esophageal wall 36 and LES 34 then invokes its natural healing response and repairs the damaged tissue.
- hyperplasia In injuring the tissue, hyperplasia often results where new tissue is formed in and/or around the injured tissue, thus adding bulk and mass to the tissue portion.
- the amount of ultrasound 22 applied to the targeted tissue area the amount of tissue injury, and hence the amount of tissue that will be added by the hyperplasia process, can be controlled.
- the amount and/or duration of energy applied may also take into account other factors, such as the characteristics of the tissue being affected, distance of the desired focal point 23 from the ultrasound focusing device 20 , distance of the desired focal point 23 from the surface of the tissue, and/or acceptable risk that overexposing the tissue will not result in the amount of hyperplasia desired.
- the ultrasound focusing device 20 For heating the collagen in the esophageal wall 36 , once the ultrasound focusing device 20 , for example the ultrasound array 21 , has been positioned and/or adjusted, ultrasound energy is applied to the desired collagen portions in sufficient amounts to heat the collagen to the temperature at which it breaks down and/or denatures. This denaturing and/or breaking down will cause the collagen to shrink, and hence cause the lumen of the esophagus 33 to shrink as well.
- the surrounding tissue should be relatively unaffected as only the energy from the ultrasound 22 converging at the focal point 23 should have enough magnitude to affect the desired tissue in any significant way.
- the ultrasound focusing device 20 is either moved to another body portion, or completely removed from the body.
- the heated collagen portions shrink, also causing shrinkage to the lumen of the esophagus 33 .
- the amount of ultrasound 22 applied to the targeted collagen portions the amount of heat applied to the collagen, and hence the amount of shrinkage that the lumen of the esophagus 33 will undergo, can be controlled.
- the amount and/or duration of energy applied may also take into account other factors, such as the. characteristics of the collagen being affected, distance of the desired focal point 23 from the ultrasound focusing device 20 , distance of the desired focal point 23 from the surface of the collagen, and/or acceptable risk that overexposing the collagen will destroy it.
- FIGS. 4A-4B disclose exemplary methods of treating fecal incontinence through the use of ultrasound 22 , for example, by using the ultrasound focusing device 20 disclosed in Weng.
- an ultrasound focusing device can be used to rebuild the anal sphincter, specifically by inducing hyperplasia.
- the ultrasound focusing device 20 is positioned such that its distal end is proximate either the internal anal sphincter 41 , as shown in FIG. 4A , or external anal sphincter 42 , as shown in FIG. 4B .
- the ultrasound focusing device can be positioned outside the body, as shown in FIG. 4B .
- the ultrasound focusing device 20 may also be positioned just inside the body in the lower colon 40 for treating either the internal anal sphincter 41 or external anal sphincter 42 , as shown in FIG. 4A .
- an endoscope 26 first may be positioned with its distal end 40 proximate the sphincter 41 , 42 .
- the ultrasound focusing device then may be advanced through the working channel 27 of the endoscope 26 until it is proximate the anal sphincter 41 , 42 .
- the ultrasound focusing device 20 is then adjusted, for example as in the procedures disclosed in this application for adjusting the ultrasound array 21 , such that the focal point 23 of the device 20 is targeted at the desired tissue portion.
- the desired tissue portion is a part of the anal sphincter 41 , 42 which, when hyperplasia is induced, will close the gap in the anal sphincter 41 , 42 .
- the focal area 23 of the ultrasound focusing device 20 will also be adjusted so that the ultrasound 22 will be directed to the proper volume of tissue at and/or surrounding the focal point 23 .
- the ultrasound focusing device 20 Once the ultrasound focusing device 20 has been adjusted, energy is applied via ultrasound 22 to the tissue portion in sufficient amounts to only cause injury to the desired tissue portion, for example using one of the methods disclosed above.
- the surrounding tissue should be relatively unaffected as only the energy from the ultrasound 22 converging at the focal point 23 should have enough magnitude to affect the desired tissue in any significant way.
- the ultrasound focusing device is moved.
- the injured tissue and the surrounding tissue here the internal anal sphincter 41 or external anal sphincter 42 , then invokes its natural healing response and repairs the damaged tissue.
- hyperplasia often results where new tissue is formed in and/or around the injured tissue, thus adding bulk and mass to the tissue portion.
- the amount of ultrasound 22 applied to the targeted tissue area the amount of tissue injury, and hence the amount of bulk and mass that will be added to the tissue by the hyperplasia process, can be controlled.
- the amount and/or duration of energy applied may also take into account other factors, such as the characteristics of the tissue being affected, distance of the desired focal point 23 from the ultrasound focusing device 20 , distance of the desired focal point 23 from the surface of the tissue, and/or acceptable risk that overexposing the tissue will not result in the amount of hyperplasia desired.
- FIGS. 5A and 5B depict exemplary methods of activating a mechanical implant 60 .
- a mechanical device 60 may require activation.
- an ultrasound focusing device 20 may be positioned proximate the mechanical implant 60 using any of the methods disclosed above.
- the ultrasound focusing device 20 may be advanced through the working channel 27 of an endoscope 26 disposed in the gastrointestinal tract, as shown in FIG. 5A , or it may be positioned external to the body proximate the mechanical implant 60 , as shown in FIG. 5B .
- the ultrasound focusing device 20 is then adjusted, for example as in the procedures disclosed in this application for adjusting the ultrasound array 21 , such that the focal point 23 of the device 20 is targeted at the mechanical implant 60 or the desired portion of the mechanical implant 60 .
- the desired implant portion is a part of the implant 60 which requires activation.
- the focal area 23 of the ultrasound focusing device 20 will also be adjusted so that the ultrasound 22 will be directed to the proper volume of the implant 60 and/or tissue at and/or surrounding the focal point 23 .
- the ultrasound focusing device 20 Once the ultrasound focusing device 20 has been adjusted, energy is applied via ultrasound 22 to the implant 60 in sufficient amounts to activate the implant 60 , for example using one of the methods disclosed above.
- the surrounding tissue should be relatively unaffected as only the energy from the ultrasound 22 converging at the focal point 23 should have enough magnitude to affect the implant 60 and/or the tissue in any significant way.
- the ultrasound focusing device After sufficiently activating the implant 60 , the ultrasound focusing device is moved.
- the implant 60 then performs its allotted function.
- the amount of ultrasound 22 applied to the implant 60 the amount of activation, and hence the amount of treatment by the implant, can be controlled.
- the amount and/or duration of energy applied may also take into account other factors, such as the characteristics of the implant 60 and/or tissue being affected, distance of the desired focal point 23 from the ultrasound focusing device 20 , distance of the desired focal point 23 from the surface of the tissue, and/or acceptable risk that overexposing the implant 60 will not result in the desired activation and/or treatment.
- the activation may only occur while energy is being applied to implant 60 via ultrasound, or may continue even after application of energy has stopped.
- a mechanical device such as a surgical instrument placed in the body to perform a treatment, as opposed to an implant in the body, may be activated through ultrasound.
- the delivery of ultrasonic energy to treat various internal organs or tissue is exemplary.
- Other types of energy and/or energy delivery devices may be used, including radio frequency, microwaves, lasers, electromagnetics, cryogenic fluids, heat, mechanical devices, or other energy delivery systems known in the art.
- the application of energy for example via ultrasound 22
- the application of energy via ultrasound 22 may be used to treat tumors, lesions, cancerous tissue, non-cancerous tissue, benign tissue growths, and other desirable or undesirable tissue.
- steps in above described methods can be interchanged and substituted into any of the other above described methods.
- tissue necrosis in a portion of the esophagus 33 , LES 34 , fundus wall 35 , or esophageal wall 36 may be desirable to cause tissue necrosis in any of the aforementioned portions.
- tissue necrosis in a portion of the anal sphincter 41 , 42 it may be desirable to cause tissue necrosis in a portion of the anal sphincter 41 , 42 .
- some of the steps in the method of ablating tissue in the gall bladder 50 may be used to cause tissue necrosis in a portion of the anal sphincter 41 , 42 .
- some of the steps in the method of increasing tissue mass in either the LES 34 or anal sphincter 41 , 42 may be used to increase mass in a section of the gall bladder 50 .
- any of the steps in any of the above-described methods may be repeated as necessary, for example, to treat multiple sections of the esophagus 33 , ablate multiple lesions 51 , or close multiple punctures 17 .
- certain steps in any of the above-described methods may be deleted as desired or necessary.
- devices other than an endoscope may be used to deliver the ultrasound focusing device 20 .
- the use and/or treatment of specific organs and/or body parts in describing each of the above methods is exemplary.
- Other organs and/or body parts may be treated or used for access to treated tissue or organs, as desired.
- the method for ablating tissue is not be limited to the gall bladder 50 , as other internal organs (e.g., the liver, lung, kidney, pancreas, prostate, bladder, uterus, or brain) from which tissue removal is required may also have tissue removed by ablation substantially as described above.
- the method for bulking up tissue is not be limited to portions of the gastrointestinal tract, as other organs, vessels, or tissue in the body that requires an increase in bulk or mass may also be treated using the methods described above.
- the method for sealing is not limited to blood vessels 10 , as other tissue, organs, or walls which has a puncture 17 , whether artificial or natural, that requires sealing may be sealed using the method as described above.
- the use of ultrasound to produce a therapeutic response in the body and the use of ultrasound for imaging purposes may be combined.
- an ultrasound device may be used to both view the desired body portion and also to direct the ultrasound focusing device in the desired body portion. Any other method of viewing the desired body portion and/or directing the ultrasound focusing device is also contemplated.
Abstract
Description
Claims (27)
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/887,324 US7294125B2 (en) | 2003-08-22 | 2004-07-09 | Methods of delivering energy to body portions to produce a therapeutic response |
| US11/984,059 US20080132809A1 (en) | 2003-08-22 | 2007-11-13 | Methods of delivering energy to body portions to produce a therapeutic response |
| US14/094,131 US20140323925A1 (en) | 2003-08-22 | 2013-12-02 | Methods of delivering energy to body portions to produce a therapeutic response |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US49692603P | 2003-08-22 | 2003-08-22 | |
| US10/887,324 US7294125B2 (en) | 2003-08-22 | 2004-07-09 | Methods of delivering energy to body portions to produce a therapeutic response |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/984,059 Continuation US20080132809A1 (en) | 2003-08-22 | 2007-11-13 | Methods of delivering energy to body portions to produce a therapeutic response |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20050055053A1 US20050055053A1 (en) | 2005-03-10 |
| US7294125B2 true US7294125B2 (en) | 2007-11-13 |
Family
ID=34228607
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/887,324 Expired - Fee Related US7294125B2 (en) | 2003-08-22 | 2004-07-09 | Methods of delivering energy to body portions to produce a therapeutic response |
| US11/984,059 Abandoned US20080132809A1 (en) | 2003-08-22 | 2007-11-13 | Methods of delivering energy to body portions to produce a therapeutic response |
| US14/094,131 Abandoned US20140323925A1 (en) | 2003-08-22 | 2013-12-02 | Methods of delivering energy to body portions to produce a therapeutic response |
Family Applications After (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/984,059 Abandoned US20080132809A1 (en) | 2003-08-22 | 2007-11-13 | Methods of delivering energy to body portions to produce a therapeutic response |
| US14/094,131 Abandoned US20140323925A1 (en) | 2003-08-22 | 2013-12-02 | Methods of delivering energy to body portions to produce a therapeutic response |
Country Status (1)
| Country | Link |
|---|---|
| US (3) | US7294125B2 (en) |
Cited By (135)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080221491A1 (en) * | 2004-09-16 | 2008-09-11 | Guided Therapy Systems, Inc. | Method and system for combined energy therapy profile |
| US7846108B2 (en) | 2002-04-16 | 2010-12-07 | Vivant Medical, Inc. | Localization element with energized tip |
| US7875024B2 (en) | 2003-07-18 | 2011-01-25 | Vivant Medical, Inc. | Devices and methods for cooling microwave antennas |
| US8068921B2 (en) | 2006-09-29 | 2011-11-29 | Vivant Medical, Inc. | Microwave antenna assembly and method of using the same |
| US8292880B2 (en) | 2007-11-27 | 2012-10-23 | Vivant Medical, Inc. | Targeted cooling of deployable microwave antenna |
| US8690868B2 (en) | 1999-06-17 | 2014-04-08 | Covidien Lp | Needle kit and method for microwave ablation, track coagulation, and biopsy |
| US8808282B2 (en) | 2002-04-16 | 2014-08-19 | Covidien Lp | Microwave antenna having a curved configuration |
| US8857438B2 (en) | 2010-11-08 | 2014-10-14 | Ulthera, Inc. | Devices and methods for acoustic shielding |
| US8880185B2 (en) | 2010-06-11 | 2014-11-04 | Boston Scientific Scimed, Inc. | Renal denervation and stimulation employing wireless vascular energy transfer arrangement |
| US8915853B2 (en) | 2004-10-06 | 2014-12-23 | Guided Therapy Systems, Llc | Methods for face and neck lifts |
| US8915870B2 (en) | 2004-10-06 | 2014-12-23 | Guided Therapy Systems, Llc | Method and system for treating stretch marks |
| US8932224B2 (en) | 2004-10-06 | 2015-01-13 | Guided Therapy Systems, Llc | Energy based hyperhidrosis treatment |
| US8939970B2 (en) | 2004-09-10 | 2015-01-27 | Vessix Vascular, Inc. | Tuned RF energy and electrical tissue characterization for selective treatment of target tissues |
| US8951251B2 (en) | 2011-11-08 | 2015-02-10 | Boston Scientific Scimed, Inc. | Ostial renal nerve ablation |
| US8974451B2 (en) | 2010-10-25 | 2015-03-10 | Boston Scientific Scimed, Inc. | Renal nerve ablation using conductive fluid jet and RF energy |
| US9011337B2 (en) | 2011-07-11 | 2015-04-21 | Guided Therapy Systems, Llc | Systems and methods for monitoring and controlling ultrasound power output and stability |
| US9023034B2 (en) | 2010-11-22 | 2015-05-05 | Boston Scientific Scimed, Inc. | Renal ablation electrode with force-activatable conduction apparatus |
| US9028472B2 (en) | 2011-12-23 | 2015-05-12 | Vessix Vascular, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9028485B2 (en) | 2010-11-15 | 2015-05-12 | Boston Scientific Scimed, Inc. | Self-expanding cooling electrode for renal nerve ablation |
| US9039619B2 (en) | 2004-10-06 | 2015-05-26 | Guided Therapy Systems, L.L.C. | Methods for treating skin laxity |
| US9039617B2 (en) | 2009-11-24 | 2015-05-26 | Guided Therapy Systems, Llc | Methods and systems for generating thermal bubbles for improved ultrasound imaging and therapy |
| US9044254B2 (en) | 2012-08-07 | 2015-06-02 | Covidien Lp | Microwave ablation catheter and method of utilizing the same |
| US9050106B2 (en) | 2011-12-29 | 2015-06-09 | Boston Scientific Scimed, Inc. | Off-wall electrode device and methods for nerve modulation |
| US9060761B2 (en) | 2010-11-18 | 2015-06-23 | Boston Scientific Scime, Inc. | Catheter-focused magnetic field induced renal nerve ablation |
| US9079000B2 (en) | 2011-10-18 | 2015-07-14 | Boston Scientific Scimed, Inc. | Integrated crossing balloon catheter |
| US9084609B2 (en) | 2010-07-30 | 2015-07-21 | Boston Scientific Scime, Inc. | Spiral balloon catheter for renal nerve ablation |
| US9089350B2 (en) | 2010-11-16 | 2015-07-28 | Boston Scientific Scimed, Inc. | Renal denervation catheter with RF electrode and integral contrast dye injection arrangement |
| US9114247B2 (en) | 2004-09-16 | 2015-08-25 | Guided Therapy Systems, Llc | Method and system for ultrasound treatment with a multi-directional transducer |
| US9119632B2 (en) | 2011-11-21 | 2015-09-01 | Boston Scientific Scimed, Inc. | Deflectable renal nerve ablation catheter |
| US9119600B2 (en) | 2011-11-15 | 2015-09-01 | Boston Scientific Scimed, Inc. | Device and methods for renal nerve modulation monitoring |
| US9121774B2 (en) | 2012-06-22 | 2015-09-01 | Covidien Lp | Microwave thermometry for microwave ablation systems |
| US9125666B2 (en) | 2003-09-12 | 2015-09-08 | Vessix Vascular, Inc. | Selectable eccentric remodeling and/or ablation of atherosclerotic material |
| US9125667B2 (en) | 2004-09-10 | 2015-09-08 | Vessix Vascular, Inc. | System for inducing desirable temperature effects on body tissue |
| US9149658B2 (en) | 2010-08-02 | 2015-10-06 | Guided Therapy Systems, Llc | Systems and methods for ultrasound treatment |
| US9155589B2 (en) | 2010-07-30 | 2015-10-13 | Boston Scientific Scimed, Inc. | Sequential activation RF electrode set for renal nerve ablation |
| US9162046B2 (en) | 2011-10-18 | 2015-10-20 | Boston Scientific Scimed, Inc. | Deflectable medical devices |
| US9173696B2 (en) | 2012-09-17 | 2015-11-03 | Boston Scientific Scimed, Inc. | Self-positioning electrode system and method for renal nerve modulation |
| US9186209B2 (en) | 2011-07-22 | 2015-11-17 | Boston Scientific Scimed, Inc. | Nerve modulation system having helical guide |
| US9186210B2 (en) | 2011-10-10 | 2015-11-17 | Boston Scientific Scimed, Inc. | Medical devices including ablation electrodes |
| US9192435B2 (en) | 2010-11-22 | 2015-11-24 | Boston Scientific Scimed, Inc. | Renal denervation catheter with cooled RF electrode |
| US9192790B2 (en) | 2010-04-14 | 2015-11-24 | Boston Scientific Scimed, Inc. | Focused ultrasonic renal denervation |
| US9216276B2 (en) | 2007-05-07 | 2015-12-22 | Guided Therapy Systems, Llc | Methods and systems for modulating medicants using acoustic energy |
| US9220561B2 (en) | 2011-01-19 | 2015-12-29 | Boston Scientific Scimed, Inc. | Guide-compatible large-electrode catheter for renal nerve ablation with reduced arterial injury |
| US9220558B2 (en) | 2010-10-27 | 2015-12-29 | Boston Scientific Scimed, Inc. | RF renal denervation catheter with multiple independent electrodes |
| US9263663B2 (en) | 2012-04-13 | 2016-02-16 | Ardent Sound, Inc. | Method of making thick film transducer arrays |
| US9265969B2 (en) | 2011-12-21 | 2016-02-23 | Cardiac Pacemakers, Inc. | Methods for modulating cell function |
| US9272162B2 (en) | 1997-10-14 | 2016-03-01 | Guided Therapy Systems, Llc | Imaging, therapy, and temperature monitoring ultrasonic method |
| US9277955B2 (en) | 2010-04-09 | 2016-03-08 | Vessix Vascular, Inc. | Power generating and control apparatus for the treatment of tissue |
| US9283409B2 (en) | 2004-10-06 | 2016-03-15 | Guided Therapy Systems, Llc | Energy based fat reduction |
| US9283410B2 (en) | 2004-10-06 | 2016-03-15 | Guided Therapy Systems, L.L.C. | System and method for fat and cellulite reduction |
| US9297845B2 (en) | 2013-03-15 | 2016-03-29 | Boston Scientific Scimed, Inc. | Medical devices and methods for treatment of hypertension that utilize impedance compensation |
| US9320537B2 (en) | 2004-10-06 | 2016-04-26 | Guided Therapy Systems, Llc | Methods for noninvasive skin tightening |
| US9327100B2 (en) | 2008-11-14 | 2016-05-03 | Vessix Vascular, Inc. | Selective drug delivery in a lumen |
| US9326751B2 (en) | 2010-11-17 | 2016-05-03 | Boston Scientific Scimed, Inc. | Catheter guidance of external energy for renal denervation |
| US9358365B2 (en) | 2010-07-30 | 2016-06-07 | Boston Scientific Scimed, Inc. | Precision electrode movement control for renal nerve ablation |
| US9364284B2 (en) | 2011-10-12 | 2016-06-14 | Boston Scientific Scimed, Inc. | Method of making an off-wall spacer cage |
| US9408661B2 (en) | 2010-07-30 | 2016-08-09 | Patrick A. Haverkost | RF electrodes on multiple flexible wires for renal nerve ablation |
| US9420955B2 (en) | 2011-10-11 | 2016-08-23 | Boston Scientific Scimed, Inc. | Intravascular temperature monitoring system and method |
| US9433760B2 (en) | 2011-12-28 | 2016-09-06 | Boston Scientific Scimed, Inc. | Device and methods for nerve modulation using a novel ablation catheter with polymeric ablative elements |
| US9452302B2 (en) | 2011-07-10 | 2016-09-27 | Guided Therapy Systems, Llc | Systems and methods for accelerating healing of implanted material and/or native tissue |
| US9463062B2 (en) | 2010-07-30 | 2016-10-11 | Boston Scientific Scimed, Inc. | Cooled conductive balloon RF catheter for renal nerve ablation |
| US9486270B2 (en) | 2002-04-08 | 2016-11-08 | Medtronic Ardian Luxembourg S.A.R.L. | Methods and apparatus for bilateral renal neuromodulation |
| US9486355B2 (en) | 2005-05-03 | 2016-11-08 | Vessix Vascular, Inc. | Selective accumulation of energy with or without knowledge of tissue topography |
| US9504446B2 (en) | 2010-08-02 | 2016-11-29 | Guided Therapy Systems, Llc | Systems and methods for coupling an ultrasound source to tissue |
| US9510802B2 (en) | 2012-09-21 | 2016-12-06 | Guided Therapy Systems, Llc | Reflective ultrasound technology for dermatological treatments |
| US9566454B2 (en) | 2006-09-18 | 2017-02-14 | Guided Therapy Systems, Llc | Method and sysem for non-ablative acne treatment and prevention |
| US9579030B2 (en) | 2011-07-20 | 2017-02-28 | Boston Scientific Scimed, Inc. | Percutaneous devices and methods to visualize, target and ablate nerves |
| US9610122B2 (en) | 2013-03-29 | 2017-04-04 | Covidien Lp | Step-down coaxial microwave ablation applicators and methods for manufacturing same |
| USRE46362E1 (en) | 2009-11-16 | 2017-04-11 | Covidien Lp | Twin sealing chamber hub |
| US9649156B2 (en) | 2010-12-15 | 2017-05-16 | Boston Scientific Scimed, Inc. | Bipolar off-wall electrode device for renal nerve ablation |
| US9668811B2 (en) | 2010-11-16 | 2017-06-06 | Boston Scientific Scimed, Inc. | Minimally invasive access for renal nerve ablation |
| US9687166B2 (en) | 2013-10-14 | 2017-06-27 | Boston Scientific Scimed, Inc. | High resolution cardiac mapping electrode array catheter |
| US9693821B2 (en) | 2013-03-11 | 2017-07-04 | Boston Scientific Scimed, Inc. | Medical devices for modulating nerves |
| US9694212B2 (en) | 2004-10-06 | 2017-07-04 | Guided Therapy Systems, Llc | Method and system for ultrasound treatment of skin |
| US9700340B2 (en) | 2004-10-06 | 2017-07-11 | Guided Therapy Systems, Llc | System and method for ultra-high frequency ultrasound treatment |
| US9707036B2 (en) | 2013-06-25 | 2017-07-18 | Boston Scientific Scimed, Inc. | Devices and methods for nerve modulation using localized indifferent electrodes |
| US9713730B2 (en) | 2004-09-10 | 2017-07-25 | Boston Scientific Scimed, Inc. | Apparatus and method for treatment of in-stent restenosis |
| US9770606B2 (en) | 2013-10-15 | 2017-09-26 | Boston Scientific Scimed, Inc. | Ultrasound ablation catheter with cooling infusion and centering basket |
| US9808300B2 (en) | 2006-05-02 | 2017-11-07 | Boston Scientific Scimed, Inc. | Control of arterial smooth muscle tone |
| US9808311B2 (en) | 2013-03-13 | 2017-11-07 | Boston Scientific Scimed, Inc. | Deflectable medical devices |
| US9827039B2 (en) | 2013-03-15 | 2017-11-28 | Boston Scientific Scimed, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9827449B2 (en) | 2004-10-06 | 2017-11-28 | Guided Therapy Systems, L.L.C. | Systems for treating skin laxity |
| US9833283B2 (en) | 2013-07-01 | 2017-12-05 | Boston Scientific Scimed, Inc. | Medical devices for renal nerve ablation |
| US9895194B2 (en) | 2013-09-04 | 2018-02-20 | Boston Scientific Scimed, Inc. | Radio frequency (RF) balloon catheter having flushing and cooling capability |
| US9907609B2 (en) | 2014-02-04 | 2018-03-06 | Boston Scientific Scimed, Inc. | Alternative placement of thermal sensors on bipolar electrode |
| US9907535B2 (en) | 2000-12-28 | 2018-03-06 | Ardent Sound, Inc. | Visual imaging system for ultrasonic probe |
| US9925001B2 (en) | 2013-07-19 | 2018-03-27 | Boston Scientific Scimed, Inc. | Spiral bipolar electrode renal denervation balloon |
| US9943365B2 (en) | 2013-06-21 | 2018-04-17 | Boston Scientific Scimed, Inc. | Renal denervation balloon catheter with ride along electrode support |
| US9956033B2 (en) | 2013-03-11 | 2018-05-01 | Boston Scientific Scimed, Inc. | Medical devices for modulating nerves |
| US9962223B2 (en) | 2013-10-15 | 2018-05-08 | Boston Scientific Scimed, Inc. | Medical device balloon |
| US9974607B2 (en) | 2006-10-18 | 2018-05-22 | Vessix Vascular, Inc. | Inducing desirable temperature effects on body tissue |
| US10022182B2 (en) | 2013-06-21 | 2018-07-17 | Boston Scientific Scimed, Inc. | Medical devices for renal nerve ablation having rotatable shafts |
| US10039938B2 (en) | 2004-09-16 | 2018-08-07 | Guided Therapy Systems, Llc | System and method for variable depth ultrasound treatment |
| US10085799B2 (en) | 2011-10-11 | 2018-10-02 | Boston Scientific Scimed, Inc. | Off-wall electrode device and methods for nerve modulation |
| US10265122B2 (en) | 2013-03-15 | 2019-04-23 | Boston Scientific Scimed, Inc. | Nerve ablation devices and related methods of use |
| US10271898B2 (en) | 2013-10-25 | 2019-04-30 | Boston Scientific Scimed, Inc. | Embedded thermocouple in denervation flex circuit |
| US10293190B2 (en) | 2002-04-08 | 2019-05-21 | Medtronic Ardian Luxembourg S.A.R.L. | Thermally-induced renal neuromodulation and associated systems and methods |
| US10321946B2 (en) | 2012-08-24 | 2019-06-18 | Boston Scientific Scimed, Inc. | Renal nerve modulation devices with weeping RF ablation balloons |
| US10335280B2 (en) | 2000-01-19 | 2019-07-02 | Medtronic, Inc. | Method for ablating target tissue of a patient |
| US10342609B2 (en) | 2013-07-22 | 2019-07-09 | Boston Scientific Scimed, Inc. | Medical devices for renal nerve ablation |
| US10363094B2 (en) | 2011-04-08 | 2019-07-30 | Covidien Lp | Flexible microwave catheters for natural or artificial lumens |
| US10376309B2 (en) | 2016-08-02 | 2019-08-13 | Covidien Lp | Ablation cable assemblies and a method of manufacturing the same |
| US10398464B2 (en) | 2012-09-21 | 2019-09-03 | Boston Scientific Scimed, Inc. | System for nerve modulation and innocuous thermal gradient nerve block |
| US10413357B2 (en) | 2013-07-11 | 2019-09-17 | Boston Scientific Scimed, Inc. | Medical device with stretchable electrode assemblies |
| US10420960B2 (en) | 2013-03-08 | 2019-09-24 | Ulthera, Inc. | Devices and methods for multi-focus ultrasound therapy |
| US10537304B2 (en) | 2008-06-06 | 2020-01-21 | Ulthera, Inc. | Hand wand for ultrasonic cosmetic treatment and imaging |
| US10549127B2 (en) | 2012-09-21 | 2020-02-04 | Boston Scientific Scimed, Inc. | Self-cooling ultrasound ablation catheter |
| US10561862B2 (en) | 2013-03-15 | 2020-02-18 | Guided Therapy Systems, Llc | Ultrasound treatment device and methods of use |
| US10589130B2 (en) | 2006-05-25 | 2020-03-17 | Medtronic, Inc. | Methods of using high intensity focused ultrasound to form an ablated tissue area containing a plurality of lesions |
| US10603521B2 (en) | 2014-04-18 | 2020-03-31 | Ulthera, Inc. | Band transducer ultrasound therapy |
| US10624697B2 (en) | 2014-08-26 | 2020-04-21 | Covidien Lp | Microwave ablation system |
| US10660703B2 (en) | 2012-05-08 | 2020-05-26 | Boston Scientific Scimed, Inc. | Renal nerve modulation devices |
| US10660698B2 (en) | 2013-07-11 | 2020-05-26 | Boston Scientific Scimed, Inc. | Devices and methods for nerve modulation |
| US10695124B2 (en) | 2013-07-22 | 2020-06-30 | Boston Scientific Scimed, Inc. | Renal nerve ablation catheter having twist balloon |
| US10722300B2 (en) | 2013-08-22 | 2020-07-28 | Boston Scientific Scimed, Inc. | Flexible circuit having improved adhesion to a renal nerve modulation balloon |
| US10813692B2 (en) | 2016-02-29 | 2020-10-27 | Covidien Lp | 90-degree interlocking geometry for introducer for facilitating deployment of microwave radiating catheter |
| US10813691B2 (en) | 2014-10-01 | 2020-10-27 | Covidien Lp | Miniaturized microwave ablation assembly |
| US10835305B2 (en) | 2012-10-10 | 2020-11-17 | Boston Scientific Scimed, Inc. | Renal nerve modulation devices and methods |
| US10864385B2 (en) | 2004-09-24 | 2020-12-15 | Guided Therapy Systems, Llc | Rejuvenating skin by heating tissue for cosmetic treatment of the face and body |
| US10945786B2 (en) | 2013-10-18 | 2021-03-16 | Boston Scientific Scimed, Inc. | Balloon catheters with flexible conducting wires and related methods of use and manufacture |
| US10952790B2 (en) | 2013-09-13 | 2021-03-23 | Boston Scientific Scimed, Inc. | Ablation balloon with vapor deposited cover layer |
| US11000679B2 (en) | 2014-02-04 | 2021-05-11 | Boston Scientific Scimed, Inc. | Balloon protection and rewrapping devices and related methods of use |
| US11065053B2 (en) | 2016-08-02 | 2021-07-20 | Covidien Lp | Ablation cable assemblies and a method of manufacturing the same |
| US11197715B2 (en) | 2016-08-02 | 2021-12-14 | Covidien Lp | Ablation cable assemblies and a method of manufacturing the same |
| US11202671B2 (en) | 2014-01-06 | 2021-12-21 | Boston Scientific Scimed, Inc. | Tear resistant flex circuit assembly |
| US11207548B2 (en) | 2004-10-07 | 2021-12-28 | Guided Therapy Systems, L.L.C. | Ultrasound probe for treating skin laxity |
| US11224895B2 (en) | 2016-01-18 | 2022-01-18 | Ulthera, Inc. | Compact ultrasound device having annular ultrasound array peripherally electrically connected to flexible printed circuit board and method of assembly thereof |
| US11235179B2 (en) | 2004-10-06 | 2022-02-01 | Guided Therapy Systems, Llc | Energy based skin gland treatment |
| US11241218B2 (en) | 2016-08-16 | 2022-02-08 | Ulthera, Inc. | Systems and methods for cosmetic ultrasound treatment of skin |
| US11246654B2 (en) | 2013-10-14 | 2022-02-15 | Boston Scientific Scimed, Inc. | Flexible renal nerve ablation devices and related methods of use and manufacture |
| US11338156B2 (en) | 2004-10-06 | 2022-05-24 | Guided Therapy Systems, Llc | Noninvasive tissue tightening system |
| US11717661B2 (en) | 2007-05-07 | 2023-08-08 | Guided Therapy Systems, Llc | Methods and systems for ultrasound assisted delivery of a medicant to tissue |
| US11724133B2 (en) | 2004-10-07 | 2023-08-15 | Guided Therapy Systems, Llc | Ultrasound probe for treatment of skin |
| US11883688B2 (en) | 2004-10-06 | 2024-01-30 | Guided Therapy Systems, Llc | Energy based fat reduction |
| US11944849B2 (en) | 2018-02-20 | 2024-04-02 | Ulthera, Inc. | Systems and methods for combined cosmetic treatment of cellulite with ultrasound |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050267453A1 (en) * | 2004-05-27 | 2005-12-01 | Wong Serena H | High intensity focused ultrasound for imaging and treatment of arrhythmias |
| US20070142884A1 (en) * | 2005-12-16 | 2007-06-21 | Acoustx Corporation | Methods and apparatuses for treating an esophageal disorder such as gastroesophageal reflux disease |
| US20070142699A1 (en) * | 2005-12-16 | 2007-06-21 | Acoustx Corporation | Methods and implantable apparatuses for treating an esophageal disorder such as gastroesophageal reflux disease |
| US7662177B2 (en) | 2006-04-12 | 2010-02-16 | Bacoustics, Llc | Apparatus and methods for pain relief using ultrasound waves in combination with cryogenic energy |
| CN101500649A (en) * | 2006-08-09 | 2009-08-05 | 皇家飞利浦电子股份有限公司 | A device for and a method of activating a physiologically effective substance by ultrasonic waves, and a capsule |
| US20080255550A1 (en) * | 2006-11-30 | 2008-10-16 | Minos Medical | Systems and methods for less invasive neutralization by ablation of tissue including the appendix and gall bladder |
| KR20090121292A (en) * | 2007-02-22 | 2009-11-25 | 라모트 앳 텔-아비브 유니버시티 리미티드 | Treating weakened vessel wall such as vulnerable plaque or aneurysms |
| US8460314B2 (en) * | 2007-02-26 | 2013-06-11 | Olympus Medical Systems Corp. | Application of procedure through natural orifice |
| JP5270417B2 (en) * | 2009-03-26 | 2013-08-21 | 東洋ゴム工業株式会社 | Pneumatic tire |
| CN108653939B (en) * | 2017-03-31 | 2019-12-24 | 宝健科技股份有限公司 | Invasive dip-angle shock wave device |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6063085A (en) | 1992-04-23 | 2000-05-16 | Scimed Life Systems, Inc. | Apparatus and method for sealing vascular punctures |
| US6156032A (en) | 1998-09-30 | 2000-12-05 | Scimed Life Systems, Inc. | Method for causing a stricture of a body passageway |
| US20010031922A1 (en) | 1999-12-23 | 2001-10-18 | Therus Corporation | Ultrasound transducers for imaging and therapy |
| US6409723B1 (en) * | 1999-04-02 | 2002-06-25 | Stuart D. Edwards | Treating body tissue by applying energy and substances |
| US20020133148A1 (en) | 2001-01-11 | 2002-09-19 | Daniel Steven A. | Bone-treatment instrument and method |
| US6635054B2 (en) | 2000-07-13 | 2003-10-21 | Transurgical, Inc. | Thermal treatment methods and apparatus with focused energy application |
| US20040082859A1 (en) * | 2002-07-01 | 2004-04-29 | Alan Schaer | Method and apparatus employing ultrasound energy to treat body sphincters |
| US6740082B2 (en) | 1998-12-29 | 2004-05-25 | John H. Shadduck | Surgical instruments for treating gastro-esophageal reflux |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4802487A (en) * | 1987-03-26 | 1989-02-07 | Washington Research Foundation | Endoscopically deliverable ultrasound imaging system |
| US5018508A (en) * | 1988-06-03 | 1991-05-28 | Fry Francis J | System and method using chemicals and ultrasound or ultrasound alone to replace more conventional surgery |
| US4929246A (en) * | 1988-10-27 | 1990-05-29 | C. R. Bard, Inc. | Method for closing and sealing an artery after removing a catheter |
| US5415657A (en) * | 1992-10-13 | 1995-05-16 | Taymor-Luria; Howard | Percutaneous vascular sealing method |
| US5603323A (en) * | 1996-02-27 | 1997-02-18 | Advanced Technology Laboratories, Inc. | Medical ultrasonic diagnostic system with upgradeable transducer probes and other features |
| US5785644A (en) * | 1996-07-12 | 1998-07-28 | Circon Corporation | Pivotal handle assembly for a video operating laparoscope |
| US5906580A (en) * | 1997-05-05 | 1999-05-25 | Creare Inc. | Ultrasound system and method of administering ultrasound including a plurality of multi-layer transducer elements |
| US6007499A (en) * | 1997-10-31 | 1999-12-28 | University Of Washington | Method and apparatus for medical procedures using high-intensity focused ultrasound |
| US6508815B1 (en) * | 1998-05-08 | 2003-01-21 | Novacept | Radio-frequency generator for powering an ablation device |
| US6425867B1 (en) * | 1998-09-18 | 2002-07-30 | University Of Washington | Noise-free real time ultrasonic imaging of a treatment site undergoing high intensity focused ultrasound therapy |
| US6451013B1 (en) * | 2000-01-19 | 2002-09-17 | Medtronic Xomed, Inc. | Methods of tonsil reduction using high intensity focused ultrasound to form an ablated tissue area containing a plurality of lesions |
| US6672312B2 (en) * | 2001-01-31 | 2004-01-06 | Transurgical, Inc. | Pulmonary vein ablation with myocardial tissue locating |
-
2004
- 2004-07-09 US US10/887,324 patent/US7294125B2/en not_active Expired - Fee Related
-
2007
- 2007-11-13 US US11/984,059 patent/US20080132809A1/en not_active Abandoned
-
2013
- 2013-12-02 US US14/094,131 patent/US20140323925A1/en not_active Abandoned
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6063085A (en) | 1992-04-23 | 2000-05-16 | Scimed Life Systems, Inc. | Apparatus and method for sealing vascular punctures |
| US6156032A (en) | 1998-09-30 | 2000-12-05 | Scimed Life Systems, Inc. | Method for causing a stricture of a body passageway |
| US6740082B2 (en) | 1998-12-29 | 2004-05-25 | John H. Shadduck | Surgical instruments for treating gastro-esophageal reflux |
| US6409723B1 (en) * | 1999-04-02 | 2002-06-25 | Stuart D. Edwards | Treating body tissue by applying energy and substances |
| US20010031922A1 (en) | 1999-12-23 | 2001-10-18 | Therus Corporation | Ultrasound transducers for imaging and therapy |
| US6635054B2 (en) | 2000-07-13 | 2003-10-21 | Transurgical, Inc. | Thermal treatment methods and apparatus with focused energy application |
| US20020133148A1 (en) | 2001-01-11 | 2002-09-19 | Daniel Steven A. | Bone-treatment instrument and method |
| US20040082859A1 (en) * | 2002-07-01 | 2004-04-29 | Alan Schaer | Method and apparatus employing ultrasound energy to treat body sphincters |
Non-Patent Citations (37)
| Title |
|---|
| Ablatherm: EDAP-Prostate Cancer treated by HIFU, High Intensity Focused Ultrasound: Homepage, Website of EDAP-The HIFU Company, Jul. 9, 2004 (1 page). http://www.edap-hifu.com. |
| Ablatherm: High Intensity Focused Ultrasound Treatment for Localized Prostate Cancer: Patient Information, Patient Brochure issued by EDAP-The HIFU Company, Jul. 2003 (16 pages). |
| About Therus, Therus Corporation, Aug. 10, 2003 (1 page). http://www.therus.com/about.html. |
| Boston Scientific forms ties to Therus, Boston Business Journal, May 2, 2002 (2 pages). http://boston.bizjournals.com/boston/stories/2002/04/29/daily45.html. |
| Clinical Applications, Therus Corporation, Aug. 10, 2003 (1 page). http://www.therus.com/clinical<SUB>-</SUB>apps.html. |
| EDAP: Prostate Cancer Treated by HIFU, High Intensity Focused Ultrasound: Headlines, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/physicians/news/index.htm. |
| HIFU Treatment With Ablatherm-History-EDAP-HIFU: How, When and Where did it Start!, Website of EDAP-The HIFU Company, Jul. 9, 2004 (1 page). http://www.edap-hifu.com/eng/patients/prostate<SUB>-</SUB>cancer/2b<SUB>-</SUB>hifu<SUB>-</SUB>history.htm. |
| Leslie, Mitch, Inside-Out Ultrasound: A New Way to View the Digestive System, Stanford Medicine, vol. 18, No. 1, Winter/Spring 2001. http://www.med.stanford.edu/center/communications/stanmed/2001winterspring/ultrasoud.html. |
| Prostate Cancer Curative Treatments-EDAP-HIFU: What are the Curative Treatments for Prostate Cancer?, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/patients/prostate<SUB>-</SUB>cancer/1f<SUB>-</SUB>info <SUB>-</SUB>curative <SUB>-</SUB>treat.htm. |
| Prostate Cancer Treated by HIFU, Ablatherm, Clinical Information: Histological Changes After Ablatherm, Website of EDAP-The HIFU Company, Jul. 9, 2004 (1 page). http://www.edap-hifu.com/eng/physicians/hifu/4b<SUB>-</SUB>info<SUB>-</SUB>histology.htm. |
| Prostate Cancer Treated by HIFU, Ablatherm, Clinical Information: MRI Post Treatment, Website of EDAP-The HIFU Company, Jul. 9, 2004 (1 page). http://www.edap-hifu.com/eng/physicians/hifu/4c<SUB>-</SUB>info<SUB>-</SUB>mri.htm. |
| Prostate Cancer Treated by HIFU, Ablatherm, Clinical Information: PSA Evolution Post Treatment, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/physicians/hifu/4d<SUB>-</SUB>info<SUB>-</SUB>psa.htm. |
| Prostate Cancer Treated by HIFU, Ablatherm, Clinical Result: Large Cohort Studies, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/physicians/hifu/5a<SUB>-</SUB>results<SUB>-</SUB>standard.htm. |
| Prostate Cancer Treated by HIFU, Ablatherm, Clinical Result: Long Term Results, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/physicians/hifu/5b<SUB>-</SUB>results<SUB>-</SUB>longterm.htm. |
| Prostate Cancer Treated by HIFU, Ablatherm, Clinical Result: PSA Stability, Website of EDAP-The HIFU Company, Jul. 9, 2004 (1 page). http://www.edap-hifu.com/eng/physicians/hifu/5c<SUB>-</SUB>results<SUB>-</SUB>psa.htm. |
| Prostate Cancer Treated by HIFU, Ablatherm, Clinical Result: Safety Results, Website of EDAP-The HIFU Company, Jul. 9, 2004 (1 page). http://www.edap-hifu.com/eng/physicians/hifu/5c<SUB>-</SUB>results<SUB>-</SUB>safety.htm. |
| Prostate Cancer Treated by HIFU, Ablatherm, Clinical Trials: Europe, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/physicians/hifu/6<SUB>-</SUB>trials<SUB>-</SUB>europe.htm. |
| Prostate Cancer Treated by HIFU, Ablatherm, Published Articles, Abstracts, Website of EDAP-The HIFU Company, Jul. 9, 2004 (11 pages). http://www.edap-hifu.com/eng/physicians/hifu/7<SUB>-</SUB>published.htm. |
| Prostate Cancer Treated by HIFU, Ablatherm: A Brief History of Ablatherm Development, Website of EDAP-The HIFU Company, Jul. 9, 2004 (1 page). http://www.edap-hifu.com/eng/physicians/hifu/3b<SUB>-</SUB>treatment<SUB>-</SUB>history.htm. |
| Prostate Cancer Treated by HIFU, Ablatherm: How is Ablatherm Treatment Performed?, Website of EDAP-The HIFU Company, Jul. 9, 2004 (3 pages). http://www.edap-hifu.com/eng/physicians/hifu/3c<SUB>-</SUB>treatment<SUB>-</SUB>treat-description.htm. |
| Prostate Cancer Treated by HIFU, Ablatherm: Overview of HIFU Treatment With Ablatherm, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/physicians/hifu/3a<SUB>-</SUB>treatment<SUB>-</SUB>overview.htm. |
| Prostate Cancer Treated by HIFU, Ablatherm: What Kind of Follow-up After Ablatherm?, Website of EDAP-The HIFU Company, Jul. 9, 2004 (1 page). http://www.edap-hifu.com/eng/physicians/hifu/3d<SUB>-</SUB>treatment<SUB>-</SUB>followup.htm. |
| Prostate Cancer Treated by HIFU, High Intensity Focused Ultrasound, Principles: History: How, When and Where Did It Start?, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/physicians/hifu/2b<SUB>-</SUB>hifu<SUB>-</SUB>history.htm. |
| Prostate Cancer Treated by HIFU, High Intensity Focused Ultrasound, Principles: How Does HIFU Create a Lesion?, Website of EDAP-The HIFU Company, Jul. 9, 2004 ( 1 page). http://www.edap-hifu.com/eng/physicians/hifu/2d<SUB>-</SUB>hifu<SUB>-</SUB>lesion.htm. |
| Prostate Cancer Treated by HIFU, High Intensity Focused Ultrasound, Principles: What are the Physical Principles?, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/physicians/hifu/2c<SUB>-</SUB>physical.htm. |
| Prostate Cancer Treated by HIFU, High Intensity Focused Ultrasound, Principles: What is HIFU?, Website of EDAP-The HIFU Company, Jul. 9, 2004 (1 page). http://www.edap-hifu.com/eng/physicians/hifu/2a<SUB>-</SUB>hifu<SUB>-</SUB>overview.htm. |
| Prostate Cancer Treated by HIFU, High Intensity Focused Ultrasound: Mainpage, Website of EDAP-The HIFU Company, Jul. 9, 2004 (3 pages). http://www.edap-hifu.com/eng/physicians/hifu/index.htm. |
| Prostate Cancer Treatment With Ablatherm-EDAP-HIFU: What is Ablatherm?, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/patients/prostate<SUB>-</SUB>cancer/2c<SUB>-</SUB>hifu<SUB>-</SUB>whatis.htm. |
| Prostate Cancer Treatment with Focalised Ultrasound-EDAP-HIFU: Frequently Asked Questions, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/patients/prostate<SUB>-</SUB>cancer/5<SUB>-</SUB>FAQ.htm. |
| Prostate Cancer Treatments With Ablatherm-EDAP-HIFU: How is Ablatherm Treatment Performed?, Website of EDAP-The HIFU Company, Jul. 9, 2004 (3 pages). http://www.edap-hifu.com/eng/patients/prostate<SUB>-</SUB>cancer/2d<SUB>-</SUB>hifu<SUB>-</SUB>treatment.htm. |
| Prostate Cancer Treatments With Ablatherm-EDAP-HIFU: What Are Possible Side Effects?, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/patients/prostate<SUB>-</SUB>cancer/2g<SUB>-</SUB>hifu<SUB>-</SUB>sideeffects.htm. |
| Prostate Cancer Treatments With Ablatherm-EDAP-HIFU: What are the Benefits and Goals?, Website of EDAP-The HIFU Company, Jul. 9, 2004 (1 page). http://www.edap-hifu.com/eng/patients/prostate<SUB>-</SUB>cancer/2h<SUB>-</SUB>hifu<SUB>-</SUB>benefits.htm. |
| Prostate Cancer Treatments With Ablatherm-EDAP-HIFU: What Happens After the Treatment?, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/patients/prostate<SUB>-</SUB>cancer/2e<SUB>-</SUB>hifu<SUB>-</SUB>followup.htm. |
| Prostate Cancer Treatments With Ablatherm-EDAP-HIFU: What Results Could You Expect?, Website of EDAP-The HIFU Company, Jul. 9, 2004 (1 page). http://www.edap-hifu.com/eng/patients/prostate<SUB>-</SUB>cancer/2f<SUB>-</SUB>hifu<SUB>-</SUB>results.htm. |
| Prostate Cancer Tretments With Ablatherm-EDAP-HIFU: Quick Overview of the HIFU-Ablatherm Therapy, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/patients/prostate<SUB>-</SUB>cancer/2a<SUB>-</SUB>hifu<SUB>-</SUB>overview.htm. |
| Prostate Contact Treatment Clinical Trial-EDAP-HIFU, Website of EDAP-The HIFU Company, Jul. 9, 2004 (2 pages). http://www.edap-hifu.com/eng/patients/prostate<SUB>-</SUB>cancer/3<SUB>-</SUB>clinical<SUB>-</SUB>trial<SUB>-</SUB>europe.htm. |
| Therus Theraseal, Graphical Renderings of Product Prototypes developed by the Stratos Product Development Group, Aug. 10, 2003 (3 pages). http://www.stratos.com/HTML/work/therus-theraseal.shtml. |
Cited By (222)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9272162B2 (en) | 1997-10-14 | 2016-03-01 | Guided Therapy Systems, Llc | Imaging, therapy, and temperature monitoring ultrasonic method |
| US8690868B2 (en) | 1999-06-17 | 2014-04-08 | Covidien Lp | Needle kit and method for microwave ablation, track coagulation, and biopsy |
| US10335280B2 (en) | 2000-01-19 | 2019-07-02 | Medtronic, Inc. | Method for ablating target tissue of a patient |
| US9907535B2 (en) | 2000-12-28 | 2018-03-06 | Ardent Sound, Inc. | Visual imaging system for ultrasonic probe |
| US9486270B2 (en) | 2002-04-08 | 2016-11-08 | Medtronic Ardian Luxembourg S.A.R.L. | Methods and apparatus for bilateral renal neuromodulation |
| US10293190B2 (en) | 2002-04-08 | 2019-05-21 | Medtronic Ardian Luxembourg S.A.R.L. | Thermally-induced renal neuromodulation and associated systems and methods |
| US7846108B2 (en) | 2002-04-16 | 2010-12-07 | Vivant Medical, Inc. | Localization element with energized tip |
| US10363097B2 (en) | 2002-04-16 | 2019-07-30 | Coviden Lp | Ablation system having multiple energy sources |
| US8808282B2 (en) | 2002-04-16 | 2014-08-19 | Covidien Lp | Microwave antenna having a curved configuration |
| US11045253B2 (en) | 2002-04-16 | 2021-06-29 | Covidien Lp | Electrosurgical energy channel splitters and systems for delivering electrosurgical energy |
| US10143520B2 (en) | 2002-04-16 | 2018-12-04 | Covidien Lp | Microwave antenna guide assembly |
| US10039602B2 (en) | 2002-04-16 | 2018-08-07 | Covidien Lp | Electrosurgical energy channel splitters and systems for delivering electrosurgical energy |
| US9480528B2 (en) | 2003-07-18 | 2016-11-01 | Covidien Lp | Devices and methods for cooling microwave antennas |
| US10405921B2 (en) | 2003-07-18 | 2019-09-10 | Covidien Lp | Devices and methods for cooling microwave antennas |
| US7875024B2 (en) | 2003-07-18 | 2011-01-25 | Vivant Medical, Inc. | Devices and methods for cooling microwave antennas |
| US9468499B2 (en) | 2003-07-18 | 2016-10-18 | Covidien Lp | Devices and methods for cooling microwave antennas |
| US9820814B2 (en) | 2003-07-18 | 2017-11-21 | Covidien Lp | Devices and methods for cooling microwave antennas |
| US9125666B2 (en) | 2003-09-12 | 2015-09-08 | Vessix Vascular, Inc. | Selectable eccentric remodeling and/or ablation of atherosclerotic material |
| US9510901B2 (en) | 2003-09-12 | 2016-12-06 | Vessix Vascular, Inc. | Selectable eccentric remodeling and/or ablation |
| US10188457B2 (en) | 2003-09-12 | 2019-01-29 | Vessix Vascular, Inc. | Selectable eccentric remodeling and/or ablation |
| US9713730B2 (en) | 2004-09-10 | 2017-07-25 | Boston Scientific Scimed, Inc. | Apparatus and method for treatment of in-stent restenosis |
| US8939970B2 (en) | 2004-09-10 | 2015-01-27 | Vessix Vascular, Inc. | Tuned RF energy and electrical tissue characterization for selective treatment of target tissues |
| US9125667B2 (en) | 2004-09-10 | 2015-09-08 | Vessix Vascular, Inc. | System for inducing desirable temperature effects on body tissue |
| US10039938B2 (en) | 2004-09-16 | 2018-08-07 | Guided Therapy Systems, Llc | System and method for variable depth ultrasound treatment |
| US9011336B2 (en) * | 2004-09-16 | 2015-04-21 | Guided Therapy Systems, Llc | Method and system for combined energy therapy profile |
| US20080221491A1 (en) * | 2004-09-16 | 2008-09-11 | Guided Therapy Systems, Inc. | Method and system for combined energy therapy profile |
| US9114247B2 (en) | 2004-09-16 | 2015-08-25 | Guided Therapy Systems, Llc | Method and system for ultrasound treatment with a multi-directional transducer |
| US10864385B2 (en) | 2004-09-24 | 2020-12-15 | Guided Therapy Systems, Llc | Rejuvenating skin by heating tissue for cosmetic treatment of the face and body |
| US10328289B2 (en) | 2004-09-24 | 2019-06-25 | Guided Therapy Systems, Llc | Rejuvenating skin by heating tissue for cosmetic treatment of the face and body |
| US9895560B2 (en) | 2004-09-24 | 2018-02-20 | Guided Therapy Systems, Llc | Methods for rejuvenating skin by heating tissue for cosmetic treatment of the face and body |
| US11590370B2 (en) | 2004-09-24 | 2023-02-28 | Guided Therapy Systems, Llc | Rejuvenating skin by heating tissue for cosmetic treatment of the face and body |
| US9095697B2 (en) | 2004-09-24 | 2015-08-04 | Guided Therapy Systems, Llc | Methods for preheating tissue for cosmetic treatment of the face and body |
| US10888718B2 (en) | 2004-10-06 | 2021-01-12 | Guided Therapy Systems, L.L.C. | Ultrasound probe for treating skin laxity |
| US11338156B2 (en) | 2004-10-06 | 2022-05-24 | Guided Therapy Systems, Llc | Noninvasive tissue tightening system |
| US9533175B2 (en) | 2004-10-06 | 2017-01-03 | Guided Therapy Systems, Llc | Energy based fat reduction |
| US9707412B2 (en) | 2004-10-06 | 2017-07-18 | Guided Therapy Systems, Llc | System and method for fat and cellulite reduction |
| US9700340B2 (en) | 2004-10-06 | 2017-07-11 | Guided Therapy Systems, Llc | System and method for ultra-high frequency ultrasound treatment |
| US9694212B2 (en) | 2004-10-06 | 2017-07-04 | Guided Therapy Systems, Llc | Method and system for ultrasound treatment of skin |
| US9694211B2 (en) | 2004-10-06 | 2017-07-04 | Guided Therapy Systems, L.L.C. | Systems for treating skin laxity |
| US10265550B2 (en) | 2004-10-06 | 2019-04-23 | Guided Therapy Systems, L.L.C. | Ultrasound probe for treating skin laxity |
| US10252086B2 (en) | 2004-10-06 | 2019-04-09 | Guided Therapy Systems, Llc | Ultrasound probe for treatment of skin |
| US10245450B2 (en) | 2004-10-06 | 2019-04-02 | Guided Therapy Systems, Llc | Ultrasound probe for fat and cellulite reduction |
| US10238894B2 (en) | 2004-10-06 | 2019-03-26 | Guided Therapy Systems, L.L.C. | Energy based fat reduction |
| US11717707B2 (en) | 2004-10-06 | 2023-08-08 | Guided Therapy Systems, Llc | System and method for noninvasive skin tightening |
| US11179580B2 (en) | 2004-10-06 | 2021-11-23 | Guided Therapy Systems, Llc | Energy based fat reduction |
| US8915853B2 (en) | 2004-10-06 | 2014-12-23 | Guided Therapy Systems, Llc | Methods for face and neck lifts |
| US8915870B2 (en) | 2004-10-06 | 2014-12-23 | Guided Therapy Systems, Llc | Method and system for treating stretch marks |
| US10046181B2 (en) | 2004-10-06 | 2018-08-14 | Guided Therapy Systems, Llc | Energy based hyperhidrosis treatment |
| US10046182B2 (en) | 2004-10-06 | 2018-08-14 | Guided Therapy Systems, Llc | Methods for face and neck lifts |
| US9522290B2 (en) | 2004-10-06 | 2016-12-20 | Guided Therapy Systems, Llc | System and method for fat and cellulite reduction |
| US8932224B2 (en) | 2004-10-06 | 2015-01-13 | Guided Therapy Systems, Llc | Energy based hyperhidrosis treatment |
| US9039619B2 (en) | 2004-10-06 | 2015-05-26 | Guided Therapy Systems, L.L.C. | Methods for treating skin laxity |
| US11697033B2 (en) | 2004-10-06 | 2023-07-11 | Guided Therapy Systems, Llc | Methods for lifting skin tissue |
| US10010726B2 (en) | 2004-10-06 | 2018-07-03 | Guided Therapy Systems, Llc | Ultrasound probe for treatment of skin |
| US10010721B2 (en) | 2004-10-06 | 2018-07-03 | Guided Therapy Systems, L.L.C. | Energy based fat reduction |
| US11400319B2 (en) | 2004-10-06 | 2022-08-02 | Guided Therapy Systems, Llc | Methods for lifting skin tissue |
| US10010725B2 (en) | 2004-10-06 | 2018-07-03 | Guided Therapy Systems, Llc | Ultrasound probe for fat and cellulite reduction |
| US10010724B2 (en) | 2004-10-06 | 2018-07-03 | Guided Therapy Systems, L.L.C. | Ultrasound probe for treating skin laxity |
| US10525288B2 (en) | 2004-10-06 | 2020-01-07 | Guided Therapy Systems, Llc | System and method for noninvasive skin tightening |
| US9283409B2 (en) | 2004-10-06 | 2016-03-15 | Guided Therapy Systems, Llc | Energy based fat reduction |
| US9283410B2 (en) | 2004-10-06 | 2016-03-15 | Guided Therapy Systems, L.L.C. | System and method for fat and cellulite reduction |
| US10888716B2 (en) | 2004-10-06 | 2021-01-12 | Guided Therapy Systems, Llc | Energy based fat reduction |
| US9320537B2 (en) | 2004-10-06 | 2016-04-26 | Guided Therapy Systems, Llc | Methods for noninvasive skin tightening |
| US10532230B2 (en) | 2004-10-06 | 2020-01-14 | Guided Therapy Systems, Llc | Methods for face and neck lifts |
| US9713731B2 (en) | 2004-10-06 | 2017-07-25 | Guided Therapy Systems, Llc | Energy based fat reduction |
| US11207547B2 (en) | 2004-10-06 | 2021-12-28 | Guided Therapy Systems, Llc | Probe for ultrasound tissue treatment |
| US9974982B2 (en) | 2004-10-06 | 2018-05-22 | Guided Therapy Systems, Llc | System and method for noninvasive skin tightening |
| US10960236B2 (en) | 2004-10-06 | 2021-03-30 | Guided Therapy Systems, Llc | System and method for noninvasive skin tightening |
| US11167155B2 (en) | 2004-10-06 | 2021-11-09 | Guided Therapy Systems, Llc | Ultrasound probe for treatment of skin |
| US11235179B2 (en) | 2004-10-06 | 2022-02-01 | Guided Therapy Systems, Llc | Energy based skin gland treatment |
| US10603523B2 (en) | 2004-10-06 | 2020-03-31 | Guided Therapy Systems, Llc | Ultrasound probe for tissue treatment |
| US10603519B2 (en) | 2004-10-06 | 2020-03-31 | Guided Therapy Systems, Llc | Energy based fat reduction |
| US9421029B2 (en) | 2004-10-06 | 2016-08-23 | Guided Therapy Systems, Llc | Energy based hyperhidrosis treatment |
| US10610706B2 (en) | 2004-10-06 | 2020-04-07 | Guided Therapy Systems, Llc | Ultrasound probe for treatment of skin |
| US9427600B2 (en) | 2004-10-06 | 2016-08-30 | Guided Therapy Systems, L.L.C. | Systems for treating skin laxity |
| US9427601B2 (en) | 2004-10-06 | 2016-08-30 | Guided Therapy Systems, Llc | Methods for face and neck lifts |
| US10610705B2 (en) | 2004-10-06 | 2020-04-07 | Guided Therapy Systems, L.L.C. | Ultrasound probe for treating skin laxity |
| US9440096B2 (en) | 2004-10-06 | 2016-09-13 | Guided Therapy Systems, Llc | Method and system for treating stretch marks |
| US9833639B2 (en) | 2004-10-06 | 2017-12-05 | Guided Therapy Systems, L.L.C. | Energy based fat reduction |
| US9833640B2 (en) | 2004-10-06 | 2017-12-05 | Guided Therapy Systems, L.L.C. | Method and system for ultrasound treatment of skin |
| US9827449B2 (en) | 2004-10-06 | 2017-11-28 | Guided Therapy Systems, L.L.C. | Systems for treating skin laxity |
| US10888717B2 (en) | 2004-10-06 | 2021-01-12 | Guided Therapy Systems, Llc | Probe for ultrasound tissue treatment |
| US11883688B2 (en) | 2004-10-06 | 2024-01-30 | Guided Therapy Systems, Llc | Energy based fat reduction |
| US11235180B2 (en) | 2004-10-06 | 2022-02-01 | Guided Therapy Systems, Llc | System and method for noninvasive skin tightening |
| US9827450B2 (en) | 2004-10-06 | 2017-11-28 | Guided Therapy Systems, L.L.C. | System and method for fat and cellulite reduction |
| US11207548B2 (en) | 2004-10-07 | 2021-12-28 | Guided Therapy Systems, L.L.C. | Ultrasound probe for treating skin laxity |
| US11724133B2 (en) | 2004-10-07 | 2023-08-15 | Guided Therapy Systems, Llc | Ultrasound probe for treatment of skin |
| US9486355B2 (en) | 2005-05-03 | 2016-11-08 | Vessix Vascular, Inc. | Selective accumulation of energy with or without knowledge of tissue topography |
| US9808300B2 (en) | 2006-05-02 | 2017-11-07 | Boston Scientific Scimed, Inc. | Control of arterial smooth muscle tone |
| US10589130B2 (en) | 2006-05-25 | 2020-03-17 | Medtronic, Inc. | Methods of using high intensity focused ultrasound to form an ablated tissue area containing a plurality of lesions |
| US9566454B2 (en) | 2006-09-18 | 2017-02-14 | Guided Therapy Systems, Llc | Method and sysem for non-ablative acne treatment and prevention |
| US9333032B2 (en) | 2006-09-29 | 2016-05-10 | Covidien Lp | Microwave antenna assembly and method of using the same |
| US8068921B2 (en) | 2006-09-29 | 2011-11-29 | Vivant Medical, Inc. | Microwave antenna assembly and method of using the same |
| US9974607B2 (en) | 2006-10-18 | 2018-05-22 | Vessix Vascular, Inc. | Inducing desirable temperature effects on body tissue |
| US10413356B2 (en) | 2006-10-18 | 2019-09-17 | Boston Scientific Scimed, Inc. | System for inducing desirable temperature effects on body tissue |
| US10213252B2 (en) | 2006-10-18 | 2019-02-26 | Vessix, Inc. | Inducing desirable temperature effects on body tissue |
| US9216276B2 (en) | 2007-05-07 | 2015-12-22 | Guided Therapy Systems, Llc | Methods and systems for modulating medicants using acoustic energy |
| US11717661B2 (en) | 2007-05-07 | 2023-08-08 | Guided Therapy Systems, Llc | Methods and systems for ultrasound assisted delivery of a medicant to tissue |
| US8292880B2 (en) | 2007-11-27 | 2012-10-23 | Vivant Medical, Inc. | Targeted cooling of deployable microwave antenna |
| US11123039B2 (en) | 2008-06-06 | 2021-09-21 | Ulthera, Inc. | System and method for ultrasound treatment |
| US10537304B2 (en) | 2008-06-06 | 2020-01-21 | Ulthera, Inc. | Hand wand for ultrasonic cosmetic treatment and imaging |
| US11723622B2 (en) | 2008-06-06 | 2023-08-15 | Ulthera, Inc. | Systems for ultrasound treatment |
| US9327100B2 (en) | 2008-11-14 | 2016-05-03 | Vessix Vascular, Inc. | Selective drug delivery in a lumen |
| USRE46362E1 (en) | 2009-11-16 | 2017-04-11 | Covidien Lp | Twin sealing chamber hub |
| US9345910B2 (en) | 2009-11-24 | 2016-05-24 | Guided Therapy Systems Llc | Methods and systems for generating thermal bubbles for improved ultrasound imaging and therapy |
| US9039617B2 (en) | 2009-11-24 | 2015-05-26 | Guided Therapy Systems, Llc | Methods and systems for generating thermal bubbles for improved ultrasound imaging and therapy |
| US9277955B2 (en) | 2010-04-09 | 2016-03-08 | Vessix Vascular, Inc. | Power generating and control apparatus for the treatment of tissue |
| US9192790B2 (en) | 2010-04-14 | 2015-11-24 | Boston Scientific Scimed, Inc. | Focused ultrasonic renal denervation |
| US8880185B2 (en) | 2010-06-11 | 2014-11-04 | Boston Scientific Scimed, Inc. | Renal denervation and stimulation employing wireless vascular energy transfer arrangement |
| US9463062B2 (en) | 2010-07-30 | 2016-10-11 | Boston Scientific Scimed, Inc. | Cooled conductive balloon RF catheter for renal nerve ablation |
| US9408661B2 (en) | 2010-07-30 | 2016-08-09 | Patrick A. Haverkost | RF electrodes on multiple flexible wires for renal nerve ablation |
| US9358365B2 (en) | 2010-07-30 | 2016-06-07 | Boston Scientific Scimed, Inc. | Precision electrode movement control for renal nerve ablation |
| US9084609B2 (en) | 2010-07-30 | 2015-07-21 | Boston Scientific Scime, Inc. | Spiral balloon catheter for renal nerve ablation |
| US9155589B2 (en) | 2010-07-30 | 2015-10-13 | Boston Scientific Scimed, Inc. | Sequential activation RF electrode set for renal nerve ablation |
| US9149658B2 (en) | 2010-08-02 | 2015-10-06 | Guided Therapy Systems, Llc | Systems and methods for ultrasound treatment |
| US10183182B2 (en) | 2010-08-02 | 2019-01-22 | Guided Therapy Systems, Llc | Methods and systems for treating plantar fascia |
| US9504446B2 (en) | 2010-08-02 | 2016-11-29 | Guided Therapy Systems, Llc | Systems and methods for coupling an ultrasound source to tissue |
| US8974451B2 (en) | 2010-10-25 | 2015-03-10 | Boston Scientific Scimed, Inc. | Renal nerve ablation using conductive fluid jet and RF energy |
| US9220558B2 (en) | 2010-10-27 | 2015-12-29 | Boston Scientific Scimed, Inc. | RF renal denervation catheter with multiple independent electrodes |
| US8857438B2 (en) | 2010-11-08 | 2014-10-14 | Ulthera, Inc. | Devices and methods for acoustic shielding |
| US9848946B2 (en) | 2010-11-15 | 2017-12-26 | Boston Scientific Scimed, Inc. | Self-expanding cooling electrode for renal nerve ablation |
| US9028485B2 (en) | 2010-11-15 | 2015-05-12 | Boston Scientific Scimed, Inc. | Self-expanding cooling electrode for renal nerve ablation |
| US9668811B2 (en) | 2010-11-16 | 2017-06-06 | Boston Scientific Scimed, Inc. | Minimally invasive access for renal nerve ablation |
| US9089350B2 (en) | 2010-11-16 | 2015-07-28 | Boston Scientific Scimed, Inc. | Renal denervation catheter with RF electrode and integral contrast dye injection arrangement |
| US9326751B2 (en) | 2010-11-17 | 2016-05-03 | Boston Scientific Scimed, Inc. | Catheter guidance of external energy for renal denervation |
| US9060761B2 (en) | 2010-11-18 | 2015-06-23 | Boston Scientific Scime, Inc. | Catheter-focused magnetic field induced renal nerve ablation |
| US9192435B2 (en) | 2010-11-22 | 2015-11-24 | Boston Scientific Scimed, Inc. | Renal denervation catheter with cooled RF electrode |
| US9023034B2 (en) | 2010-11-22 | 2015-05-05 | Boston Scientific Scimed, Inc. | Renal ablation electrode with force-activatable conduction apparatus |
| US9649156B2 (en) | 2010-12-15 | 2017-05-16 | Boston Scientific Scimed, Inc. | Bipolar off-wall electrode device for renal nerve ablation |
| US9220561B2 (en) | 2011-01-19 | 2015-12-29 | Boston Scientific Scimed, Inc. | Guide-compatible large-electrode catheter for renal nerve ablation with reduced arterial injury |
| US10363094B2 (en) | 2011-04-08 | 2019-07-30 | Covidien Lp | Flexible microwave catheters for natural or artificial lumens |
| US9452302B2 (en) | 2011-07-10 | 2016-09-27 | Guided Therapy Systems, Llc | Systems and methods for accelerating healing of implanted material and/or native tissue |
| US9011337B2 (en) | 2011-07-11 | 2015-04-21 | Guided Therapy Systems, Llc | Systems and methods for monitoring and controlling ultrasound power output and stability |
| US9579030B2 (en) | 2011-07-20 | 2017-02-28 | Boston Scientific Scimed, Inc. | Percutaneous devices and methods to visualize, target and ablate nerves |
| US9186209B2 (en) | 2011-07-22 | 2015-11-17 | Boston Scientific Scimed, Inc. | Nerve modulation system having helical guide |
| US9186210B2 (en) | 2011-10-10 | 2015-11-17 | Boston Scientific Scimed, Inc. | Medical devices including ablation electrodes |
| US9420955B2 (en) | 2011-10-11 | 2016-08-23 | Boston Scientific Scimed, Inc. | Intravascular temperature monitoring system and method |
| US10085799B2 (en) | 2011-10-11 | 2018-10-02 | Boston Scientific Scimed, Inc. | Off-wall electrode device and methods for nerve modulation |
| US9364284B2 (en) | 2011-10-12 | 2016-06-14 | Boston Scientific Scimed, Inc. | Method of making an off-wall spacer cage |
| US9079000B2 (en) | 2011-10-18 | 2015-07-14 | Boston Scientific Scimed, Inc. | Integrated crossing balloon catheter |
| US9162046B2 (en) | 2011-10-18 | 2015-10-20 | Boston Scientific Scimed, Inc. | Deflectable medical devices |
| US8951251B2 (en) | 2011-11-08 | 2015-02-10 | Boston Scientific Scimed, Inc. | Ostial renal nerve ablation |
| US9119600B2 (en) | 2011-11-15 | 2015-09-01 | Boston Scientific Scimed, Inc. | Device and methods for renal nerve modulation monitoring |
| US9119632B2 (en) | 2011-11-21 | 2015-09-01 | Boston Scientific Scimed, Inc. | Deflectable renal nerve ablation catheter |
| US9265969B2 (en) | 2011-12-21 | 2016-02-23 | Cardiac Pacemakers, Inc. | Methods for modulating cell function |
| US9186211B2 (en) | 2011-12-23 | 2015-11-17 | Boston Scientific Scimed, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9174050B2 (en) | 2011-12-23 | 2015-11-03 | Vessix Vascular, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9072902B2 (en) | 2011-12-23 | 2015-07-07 | Vessix Vascular, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9592386B2 (en) | 2011-12-23 | 2017-03-14 | Vessix Vascular, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9028472B2 (en) | 2011-12-23 | 2015-05-12 | Vessix Vascular, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9037259B2 (en) | 2011-12-23 | 2015-05-19 | Vessix Vascular, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9402684B2 (en) | 2011-12-23 | 2016-08-02 | Boston Scientific Scimed, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9433760B2 (en) | 2011-12-28 | 2016-09-06 | Boston Scientific Scimed, Inc. | Device and methods for nerve modulation using a novel ablation catheter with polymeric ablative elements |
| US9050106B2 (en) | 2011-12-29 | 2015-06-09 | Boston Scientific Scimed, Inc. | Off-wall electrode device and methods for nerve modulation |
| US9263663B2 (en) | 2012-04-13 | 2016-02-16 | Ardent Sound, Inc. | Method of making thick film transducer arrays |
| US10660703B2 (en) | 2012-05-08 | 2020-05-26 | Boston Scientific Scimed, Inc. | Renal nerve modulation devices |
| US10363095B2 (en) | 2012-06-22 | 2019-07-30 | Covidien Lp | Microwave thermometry for microwave ablation systems |
| US9743986B2 (en) | 2012-06-22 | 2017-08-29 | Covidien Lp | Microwave thermometry for microwave ablation systems |
| US9151680B2 (en) | 2012-06-22 | 2015-10-06 | Covidien Lp | Microwave thermometry for microwave ablation systems |
| US9121774B2 (en) | 2012-06-22 | 2015-09-01 | Covidien Lp | Microwave thermometry for microwave ablation systems |
| US9127989B2 (en) | 2012-06-22 | 2015-09-08 | Covidien Lp | Microwave thermometry for microwave ablation systems |
| US9993295B2 (en) | 2012-08-07 | 2018-06-12 | Covidien Lp | Microwave ablation catheter and method of utilizing the same |
| US9044254B2 (en) | 2012-08-07 | 2015-06-02 | Covidien Lp | Microwave ablation catheter and method of utilizing the same |
| US9247992B2 (en) | 2012-08-07 | 2016-02-02 | Covidien, LP | Microwave ablation catheter and method of utilizing the same |
| US9993296B2 (en) | 2012-08-07 | 2018-06-12 | Covidien Lp | Microwave ablation catheter and method of utilizing the same |
| US9259269B2 (en) | 2012-08-07 | 2016-02-16 | Covidien Lp | Microwave ablation catheter and method of utilizing the same |
| US11678934B2 (en) | 2012-08-07 | 2023-06-20 | Covidien Lp | Microwave ablation system |
| US9370398B2 (en) | 2012-08-07 | 2016-06-21 | Covidien Lp | Microwave ablation catheter and method of utilizing the same |
| US9247993B2 (en) | 2012-08-07 | 2016-02-02 | Covidien, LP | Microwave ablation catheter and method of utilizing the same |
| US10321946B2 (en) | 2012-08-24 | 2019-06-18 | Boston Scientific Scimed, Inc. | Renal nerve modulation devices with weeping RF ablation balloons |
| US9173696B2 (en) | 2012-09-17 | 2015-11-03 | Boston Scientific Scimed, Inc. | Self-positioning electrode system and method for renal nerve modulation |
| US10398464B2 (en) | 2012-09-21 | 2019-09-03 | Boston Scientific Scimed, Inc. | System for nerve modulation and innocuous thermal gradient nerve block |
| US10549127B2 (en) | 2012-09-21 | 2020-02-04 | Boston Scientific Scimed, Inc. | Self-cooling ultrasound ablation catheter |
| US9510802B2 (en) | 2012-09-21 | 2016-12-06 | Guided Therapy Systems, Llc | Reflective ultrasound technology for dermatological treatments |
| US9802063B2 (en) | 2012-09-21 | 2017-10-31 | Guided Therapy Systems, Llc | Reflective ultrasound technology for dermatological treatments |
| US10835305B2 (en) | 2012-10-10 | 2020-11-17 | Boston Scientific Scimed, Inc. | Renal nerve modulation devices and methods |
| US11517772B2 (en) | 2013-03-08 | 2022-12-06 | Ulthera, Inc. | Devices and methods for multi-focus ultrasound therapy |
| US10420960B2 (en) | 2013-03-08 | 2019-09-24 | Ulthera, Inc. | Devices and methods for multi-focus ultrasound therapy |
| US9956033B2 (en) | 2013-03-11 | 2018-05-01 | Boston Scientific Scimed, Inc. | Medical devices for modulating nerves |
| US9693821B2 (en) | 2013-03-11 | 2017-07-04 | Boston Scientific Scimed, Inc. | Medical devices for modulating nerves |
| US9808311B2 (en) | 2013-03-13 | 2017-11-07 | Boston Scientific Scimed, Inc. | Deflectable medical devices |
| US9827039B2 (en) | 2013-03-15 | 2017-11-28 | Boston Scientific Scimed, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US10265122B2 (en) | 2013-03-15 | 2019-04-23 | Boston Scientific Scimed, Inc. | Nerve ablation devices and related methods of use |
| US9297845B2 (en) | 2013-03-15 | 2016-03-29 | Boston Scientific Scimed, Inc. | Medical devices and methods for treatment of hypertension that utilize impedance compensation |
| US10561862B2 (en) | 2013-03-15 | 2020-02-18 | Guided Therapy Systems, Llc | Ultrasound treatment device and methods of use |
| US11382692B2 (en) | 2013-03-29 | 2022-07-12 | Covidien Lp | Step-down coaxial microwave ablation applicators and methods for manufacturing same |
| US9610122B2 (en) | 2013-03-29 | 2017-04-04 | Covidien Lp | Step-down coaxial microwave ablation applicators and methods for manufacturing same |
| US9987087B2 (en) | 2013-03-29 | 2018-06-05 | Covidien Lp | Step-down coaxial microwave ablation applicators and methods for manufacturing same |
| US10383688B2 (en) | 2013-03-29 | 2019-08-20 | Covidien Lp | Step-down coaxial microwave ablation applicators and methods for manufacturing same |
| US9943365B2 (en) | 2013-06-21 | 2018-04-17 | Boston Scientific Scimed, Inc. | Renal denervation balloon catheter with ride along electrode support |
| US10022182B2 (en) | 2013-06-21 | 2018-07-17 | Boston Scientific Scimed, Inc. | Medical devices for renal nerve ablation having rotatable shafts |
| US9707036B2 (en) | 2013-06-25 | 2017-07-18 | Boston Scientific Scimed, Inc. | Devices and methods for nerve modulation using localized indifferent electrodes |
| US9833283B2 (en) | 2013-07-01 | 2017-12-05 | Boston Scientific Scimed, Inc. | Medical devices for renal nerve ablation |
| US10660698B2 (en) | 2013-07-11 | 2020-05-26 | Boston Scientific Scimed, Inc. | Devices and methods for nerve modulation |
| US10413357B2 (en) | 2013-07-11 | 2019-09-17 | Boston Scientific Scimed, Inc. | Medical device with stretchable electrode assemblies |
| US9925001B2 (en) | 2013-07-19 | 2018-03-27 | Boston Scientific Scimed, Inc. | Spiral bipolar electrode renal denervation balloon |
| US10695124B2 (en) | 2013-07-22 | 2020-06-30 | Boston Scientific Scimed, Inc. | Renal nerve ablation catheter having twist balloon |
| US10342609B2 (en) | 2013-07-22 | 2019-07-09 | Boston Scientific Scimed, Inc. | Medical devices for renal nerve ablation |
| US10722300B2 (en) | 2013-08-22 | 2020-07-28 | Boston Scientific Scimed, Inc. | Flexible circuit having improved adhesion to a renal nerve modulation balloon |
| US9895194B2 (en) | 2013-09-04 | 2018-02-20 | Boston Scientific Scimed, Inc. | Radio frequency (RF) balloon catheter having flushing and cooling capability |
| US10952790B2 (en) | 2013-09-13 | 2021-03-23 | Boston Scientific Scimed, Inc. | Ablation balloon with vapor deposited cover layer |
| US9687166B2 (en) | 2013-10-14 | 2017-06-27 | Boston Scientific Scimed, Inc. | High resolution cardiac mapping electrode array catheter |
| US11246654B2 (en) | 2013-10-14 | 2022-02-15 | Boston Scientific Scimed, Inc. | Flexible renal nerve ablation devices and related methods of use and manufacture |
| US9770606B2 (en) | 2013-10-15 | 2017-09-26 | Boston Scientific Scimed, Inc. | Ultrasound ablation catheter with cooling infusion and centering basket |
| US9962223B2 (en) | 2013-10-15 | 2018-05-08 | Boston Scientific Scimed, Inc. | Medical device balloon |
| US10945786B2 (en) | 2013-10-18 | 2021-03-16 | Boston Scientific Scimed, Inc. | Balloon catheters with flexible conducting wires and related methods of use and manufacture |
| US10271898B2 (en) | 2013-10-25 | 2019-04-30 | Boston Scientific Scimed, Inc. | Embedded thermocouple in denervation flex circuit |
| US11202671B2 (en) | 2014-01-06 | 2021-12-21 | Boston Scientific Scimed, Inc. | Tear resistant flex circuit assembly |
| US11000679B2 (en) | 2014-02-04 | 2021-05-11 | Boston Scientific Scimed, Inc. | Balloon protection and rewrapping devices and related methods of use |
| US9907609B2 (en) | 2014-02-04 | 2018-03-06 | Boston Scientific Scimed, Inc. | Alternative placement of thermal sensors on bipolar electrode |
| US10603521B2 (en) | 2014-04-18 | 2020-03-31 | Ulthera, Inc. | Band transducer ultrasound therapy |
| US11351401B2 (en) | 2014-04-18 | 2022-06-07 | Ulthera, Inc. | Band transducer ultrasound therapy |
| US10624697B2 (en) | 2014-08-26 | 2020-04-21 | Covidien Lp | Microwave ablation system |
| US10813691B2 (en) | 2014-10-01 | 2020-10-27 | Covidien Lp | Miniaturized microwave ablation assembly |
| US11839426B2 (en) | 2014-10-01 | 2023-12-12 | Covidien Lp | Miniaturized microwave ablation assembly |
| US11224895B2 (en) | 2016-01-18 | 2022-01-18 | Ulthera, Inc. | Compact ultrasound device having annular ultrasound array peripherally electrically connected to flexible printed circuit board and method of assembly thereof |
| US10813692B2 (en) | 2016-02-29 | 2020-10-27 | Covidien Lp | 90-degree interlocking geometry for introducer for facilitating deployment of microwave radiating catheter |
| US11065053B2 (en) | 2016-08-02 | 2021-07-20 | Covidien Lp | Ablation cable assemblies and a method of manufacturing the same |
| US10376309B2 (en) | 2016-08-02 | 2019-08-13 | Covidien Lp | Ablation cable assemblies and a method of manufacturing the same |
| US11197715B2 (en) | 2016-08-02 | 2021-12-14 | Covidien Lp | Ablation cable assemblies and a method of manufacturing the same |
| US11241218B2 (en) | 2016-08-16 | 2022-02-08 | Ulthera, Inc. | Systems and methods for cosmetic ultrasound treatment of skin |
| US11944849B2 (en) | 2018-02-20 | 2024-04-02 | Ulthera, Inc. | Systems and methods for combined cosmetic treatment of cellulite with ultrasound |
Also Published As
| Publication number | Publication date |
|---|---|
| US20050055053A1 (en) | 2005-03-10 |
| US20080132809A1 (en) | 2008-06-05 |
| US20140323925A1 (en) | 2014-10-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7294125B2 (en) | Methods of delivering energy to body portions to produce a therapeutic response | |
| EP1117339B1 (en) | Device for causing a stricture of a body passageway | |
| US6416545B1 (en) | Urological stent therapy system and method | |
| JP4387303B2 (en) | Apparatus and method for treating urinary incontinence in women | |
| US9827008B2 (en) | Snare with loop made of heat shrinkable shape memory material and method of use thereof | |
| US20060015151A1 (en) | Method of using endoscopic truncal vagoscopy with gastric bypass, gastric banding and other procedures | |
| US20050240249A1 (en) | Methods for treating mitral valve annulus with biodegradable compression element | |
| WO1993004727A1 (en) | Balloon-catheter | |
| US20080281204A1 (en) | System and Method for Treating Hemorrhoids | |
| WO2009072131A2 (en) | Prostatic capsulotomy for treatment of conditions | |
| US20120004546A1 (en) | System and Method for Treating Hemorrhoids | |
| US20200261137A1 (en) | Cryotherapy device with cryoprotection and methods for performing cryotherapy with cryoprotection | |
| RU2231991C2 (en) | Method for treating unresectable primary and metastatic hepatic tumors | |
| PERKASH | Use of contact laser crystal tip firing Nd: YAG to relieve urinary outflow obstruction in male neurogenic bladder patients | |
| Kanehira et al. | A technique of percutaneous endoscopic intragastric surgery | |
| Elmunzer et al. | 984 PER ORAL ENDOSCOPIC MYOTOMY FOR CRICOPHARYNGEAL BAR | |
| Nostrant et al. | Endoscopic treatment for gastroesophageal reflux disease | |
| Behm et al. | Endoluminal therapies for gastroesophageal reflux disease | |
| US20100234832A1 (en) | Method for vascular treatment | |
| Sportes et al. | Dissecção endoscópica da submucosa com túnel duplo para adenocarcinoma esofágico superficial | |
| Sportes et al. | Endoscopic submucosal double tunnel dissection for superficial esophageal adenocarcinoma | |
| Surel et al. | LESS |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: SCIMED LIFE SYSTEMS, INC., MINNESOTA Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:PHALEN, MICHAEL P.;MORECI, STEPHEN F.;REEL/FRAME:015381/0262;SIGNING DATES FROM 20041018 TO 20041025 |
|
| FEPP | Fee payment procedure |
Free format text: PAYOR NUMBER ASSIGNED (ORIGINAL EVENT CODE: ASPN); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| AS | Assignment |
Owner name: BOSTON SCIENTIFIC SCIMED, INC., MINNESOTA Free format text: CHANGE OF NAME;ASSIGNOR:SCIMED LIFE SYSTEMS, INC.;REEL/FRAME:018505/0868 Effective date: 20050101 Owner name: BOSTON SCIENTIFIC SCIMED, INC.,MINNESOTA Free format text: CHANGE OF NAME;ASSIGNOR:SCIMED LIFE SYSTEMS, INC.;REEL/FRAME:018505/0868 Effective date: 20050101 |
|
| FPAY | Fee payment |
Year of fee payment: 4 |
|
| REMI | Maintenance fee reminder mailed | ||
| LAPS | Lapse for failure to pay maintenance fees | ||
| STCH | Information on status: patent discontinuation |
Free format text: PATENT EXPIRED DUE TO NONPAYMENT OF MAINTENANCE FEES UNDER 37 CFR 1.362 |
|
| FP | Lapsed due to failure to pay maintenance fee |
Effective date: 20151113 |