US8784465B2 - Implantable medical devices - Google Patents
Implantable medical devices Download PDFInfo
- Publication number
- US8784465B2 US8784465B2 US12/940,388 US94038810A US8784465B2 US 8784465 B2 US8784465 B2 US 8784465B2 US 94038810 A US94038810 A US 94038810A US 8784465 B2 US8784465 B2 US 8784465B2
- Authority
- US
- United States
- Prior art keywords
- stent
- expanded
- endoprosthesis
- polymer
- product
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related, expires
Links
- 0 *CC1CCCC1 Chemical compound *CC1CCCC1 0.000 description 5
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G61/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G61/02—Macromolecular compounds containing only carbon atoms in the main chain of the macromolecule, e.g. polyxylylenes
- C08G61/04—Macromolecular compounds containing only carbon atoms in the main chain of the macromolecule, e.g. polyxylylenes only aliphatic carbon atoms
- C08G61/06—Macromolecular compounds containing only carbon atoms in the main chain of the macromolecule, e.g. polyxylylenes only aliphatic carbon atoms prepared by ring-opening of carbocyclic compounds
- C08G61/08—Macromolecular compounds containing only carbon atoms in the main chain of the macromolecule, e.g. polyxylylenes only aliphatic carbon atoms prepared by ring-opening of carbocyclic compounds of carbocyclic compounds containing one or more carbon-to-carbon double bonds in the ring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/82—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/86—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure
- A61F2/88—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure the wire-like elements formed as helical or spiral coils
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/82—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/86—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure
- A61F2/90—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure
- A61F2/91—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure made from perforated sheet material or tubes, e.g. perforated by laser cuts or etched holes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/82—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/86—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure
- A61F2/90—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure
- A61F2/91—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure made from perforated sheet material or tubes, e.g. perforated by laser cuts or etched holes
- A61F2/915—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure made from perforated sheet material or tubes, e.g. perforated by laser cuts or etched holes with bands having a meander structure, adjacent bands being connected to each other
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L29/00—Materials for catheters, medical tubing, cannulae, or endoscopes or for coating catheters
- A61L29/14—Materials characterised by their function or physical properties, e.g. lubricating compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/30—Low-molecular-weight compounds
- C08G18/38—Low-molecular-weight compounds having heteroatoms other than oxygen
- C08G18/3893—Low-molecular-weight compounds having heteroatoms other than oxygen containing silicon
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/40—High-molecular-weight compounds
- C08G18/42—Polycondensates having carboxylic or carbonic ester groups in the main chain
- C08G18/4266—Polycondensates having carboxylic or carbonic ester groups in the main chain prepared from hydroxycarboxylic acids and/or lactones
- C08G18/4269—Lactones
- C08G18/4277—Caprolactone and/or substituted caprolactone
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/40—High-molecular-weight compounds
- C08G18/48—Polyethers
- C08G18/4833—Polyethers containing oxyethylene units
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L65/00—Compositions of macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain; Compositions of derivatives of such polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/82—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/848—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents having means for fixation to the vessel wall, e.g. barbs
- A61F2002/8483—Barbs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/82—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/86—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure
- A61F2/90—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure
- A61F2/91—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure made from perforated sheet material or tubes, e.g. perforated by laser cuts or etched holes
- A61F2/915—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure made from perforated sheet material or tubes, e.g. perforated by laser cuts or etched holes with bands having a meander structure, adjacent bands being connected to each other
- A61F2002/91533—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure made from perforated sheet material or tubes, e.g. perforated by laser cuts or etched holes with bands having a meander structure, adjacent bands being connected to each other characterised by the phase between adjacent bands
- A61F2002/91541—Adjacent bands are arranged out of phase
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/95—Instruments specially adapted for placement or removal of stents or stent-grafts
- A61F2002/9528—Instruments specially adapted for placement or removal of stents or stent-grafts for retrieval of stents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2220/00—Fixations or connections for prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2220/0008—Fixation appliances for connecting prostheses to the body
- A61F2220/0016—Fixation appliances for connecting prostheses to the body with sharp anchoring protrusions, e.g. barbs, pins, spikes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2250/00—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2250/0014—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof having different values of a given property or geometrical feature, e.g. mechanical property or material property, at different locations within the same prosthesis
- A61F2250/0039—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof having different values of a given property or geometrical feature, e.g. mechanical property or material property, at different locations within the same prosthesis differing in diameter
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2250/00—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2250/0058—Additional features; Implant or prostheses properties not otherwise provided for
- A61F2250/0067—Means for introducing or releasing pharmaceutical products into the body
- A61F2250/0068—Means for introducing or releasing pharmaceutical products into the body the pharmaceutical product being in a reservoir
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
- A61M25/1027—Making of balloon catheters
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2230/00—Compositions for preparing biodegradable polymers
Definitions
- This invention relates to implantable medical devices and methods of delivering the same.
- the body includes various passageways such as arteries, other blood vessels, and other body lumens. These passageways sometimes become occluded or weakened. For example, the passageways can be occluded by a tumor, restricted by plaque, or weakened by an aneurysm. When this occurs, the passageway can be reopened or reinforced, or even replaced, with a medical endoprosthesis.
- An endoprosthesis is typically a tubular member that is placed in a lumen in the body. Examples of endoprosthesis include stents and covered stents, sometimes called “stent-grafts”.
- An endoprosthesis can be delivered inside the body by a catheter that supports the endoprosthesis in a compacted or reduced-size form as the endoprosthesis is transported to a desired site. Upon reaching the site, the endoprosthesis is expanded, for example, so that it can contact the walls of the lumen.
- Prostate enlargement also known as benign prostate hyperplasia or benign prostate hypertrophy
- benign prostate hyperplasia is a common affliction among older men.
- the condition involves swelling of the prostate.
- the prostate surrounds the urethra, or urinary tract, and enlargement of the prostate may restrict passage of urine from the bladder towards the urethra.
- Benign prostate hyperplasia is uncomfortable because it makes urination difficult or impossible.
- the condition is also dangerous because it can lead to infection of the bladder and kidneys.
- Prostate enlargement can be treated with surgery known as resection. Resection can be accomplished by cutting away a large portion of the prostate gland. Prostate enlargement can also be treated with heat treatment, cold treatment, or ablation.
- a restricted urethra can be treated with a prostatic stent to support the urethra and keep it open despite pressure from the enlarged prostate.
- a prostatic stent may be implanted permanently or as an interim solution.
- the invention relates to implantable medical devices, for example, a stent including a polymer.
- the invention features a medical device.
- the medical device includes a balloon catheter having an expandable member, e.g., an inflatable balloon, at its distal end and a stent or other endoprosthesis.
- the stent is an apertured tubular member formed of a polymer and is assembled about the balloon.
- the stent has an initial diameter for delivery into the body and can be expanded to a larger diameter by inflating the balloon.
- the polymer does not flow substantially during expansion and substantial stress relaxation or creep does not occur so that the geometry of the stent is maintained.
- a tubular endoprosthesis including a polymer body is provided and delivered into a body lumen.
- the endoprosthesis is expanded in the body lumen under conditions of expanding pressure and temperature so that the wall thickness of the polymer body is substantially maintained.
- a polymer tube is formed to a first, large diameter.
- An aperture pattern is cut into the tube wall.
- the polymer is crosslinked or crystallized.
- the polymer tube is deformed to a second, small diameter.
- the polymer tube is expanded in a body lumen to a diameter larger than the second diameter by application of pressure and heat.
- a polymer tube is formed to a first, small diameter.
- An aperture pattern is provided in the tube wall.
- the polymer is crystallized or crosslinked.
- the tube is expanded in a body lumen by application of pressure and heat.
- an implantable medical apparatus in another aspect, includes an element operable for movement within the body by mechanical force applied to the element.
- the element includes a polymer having a melt or glass transition temperature in the range above body temperature to about 50° C. or 60° C. and exhibiting a plateau in a plot of storage modulus as a function of temperature at melt or glass transition.
- the element is a stent.
- the stent may be generally a tubular body that includes an apertured wall.
- the stent may be operable for expansion from a first, smaller diameter to a second larger diameter for implantation in a lumen.
- the thickness of the stent wall varies by about 1% or less between the first and second diameter.
- the invention features a medical device including a polymer having a melt or glass transition temperature above body temperature and exhibiting an approximate plateau in a plot of storage modulus as a function of temperature at melt or glass transition.
- the melt or glass transition temperature may be, for example, above about 37° C.
- the medical device may undergo a triggerable event at about the plateau.
- the triggerable event may be, for example, a change in the flexibility, a change in the porosity, a change in the coefficient of friction or a change in the surface roughness.
- the medical device may be, for example, a stent that has a portion that has a collapsed position that can be reverted to an expanded position by a trigger subsequent to insertion into the body.
- the polymer body optionally, includes apertures.
- the polymer body has a ratio of aperture open area to wall area of about 0.5 or more or 0.7 or more.
- the endoprosthesis is expanded by simultaneously applying an expanding pressure and heat to the endoprosthesis.
- the polymer body is heated above the melt or glass transition temperature of polymer in the polymer body.
- the polymer body is elastomeric at the melt or glass transition temperature.
- the polymer is elastomeric at body temperature.
- the polymer is crystalline.
- the polymer is crosslinked.
- the polymer is radiation crosslinked.
- the melt or glass transition temperature is about 40 to 50° C.
- the melt or glass transition temperature has a transition range of about 5° C. or less.
- the polymer exhibits a plateau in the melt or glass transition range in a plot of storage modulus as a function of temperature.
- the polymer body includes a drug, radiopaque agent or magnetic heating agent.
- the polymer is a shape memory polymer, e.g. capable of remembering a smaller diameter configuration after expansion.
- the polymer is, for example, polynorbornene, polycaprolactone, polyenes, nylons, polycyclooctene (PCO), blends of PCO and styrene-butadiene rubber, polyvinyl acetate/polyvinylidinefluoride (PVAc/PVDF), blends of PVAc/PVDF/poly-methylmethacrylate (PMMA), polyurethanes, styrene-butadiene copolymers, polyethylene, trans-isoprene, blends of polycaprolactone and n-butylacrylate, PVC, e.g., plasticized PVC, and blends thereof.
- An expansion pressure of about 1 atm or more is applied.
- the endoprosthesis is delivered on a catheter.
- the endoprosthesis is delivered to a site of occlusion and the site is simultaneously dilated while expanding the endoprosthesis.
- the endoprosthesis is delivered to a site of lumen curvature and the endoprosthesis is expanded at the site.
- the endoprosthesis is delivered to a vascular lumen.
- the endoprosthesis is delivered adjacent (into) the prostate.
- a heat applicator applies heat to the stent during inflation of the balloon to expand the balloon to the expanded diameter.
- the polymer has a melt or glass transition temperature in the range of about 40 to 50° C. and a modulus at the melt or glass transition temperature sufficient to maintain the stent geometry or under application of pressure and/or heat. The polymer exhibits a plateau in the storage modulus in the range of melt or glass transition temperatures.
- the stent has a wall thickness of about 0.005 to 5 mm.
- the stent has an initial unexpanded inner diameter in the range of about 1 mm to 5 mm.
- the stent has an expanded inner diameter of about 1 mm to 20 mm.
- the stent may be expandable to about 100% or 400% or more of the initial inner diameter.
- An example of a coronary stent has an initial inner diameter of about 2 mm, and expanded inner diameter of about 4 mm and the wall thickness is about 0.005 mm to 0.1 mm.
- the stent can be in the form of a tube including aperture areas provided in the tube.
- the aperture are in the shape of elongate slots, e.g., when the stent is in the small diameter condition.
- the apertures have a dimension of about 1 mm or less in the small diameter condition.
- the apertures are in the shape of diamond-like openings, e.g. when the stent is in an expanded condition.
- the stent can be a wire-form formed of one or more filaments configured to generally define a tube.
- the elastomeric nature of the polymer in the melted or glass state enhances the ability to maintain geometry as the stent is expanded. For example, the polymer exhibits minimal flow during expansion and the thickness of the stent remains substantially constant. Elastomeric properties in the crystalline or solid state enhance the ability to conform to torturous curvature in narrow body lumens. High compression resistance allows the stent to maintain the body lumen open and resist occluding forces such as elastic recoil or the growth of thrombus from the vessel wall.
- the invention features a polymeric stent having a portion that has a collapsed position that can be reverted to an expanded position by heating above a first temperature subsequent to insertion of the stent into a cavity or lumen.
- the stent may be in the form, for example, of a coiled elongated element (for example, a strand, a tape or a flattened tube).
- the stent may be further heated to a second temperature that is higher than the first temperature and removed as a substantially uncoiled element.
- the flattened tube may include a central opening that includes a medicament that can be released by the inserted stent.
- the medicament is compounded into the plastic or is a coating on the plastic.
- the portion is at an end of the stent and the portion is flared or stepped. In other implementations, the portion includes less than 50% of the length of the stent.
- the invention features a polymeric stent in the form of a coiled elongated element, and having a portion that has a collapsed position that can be reverted to an expanded position by heating above a first temperature subsequent to insertion of the stent into a cavity or lumen.
- the modulus of the element lowers sufficiently that the stent can be removed from the cavity or lumen as a substantially uncoiled element.
- the invention features a method of treating a non-vascular cavity or lumen.
- the method includes inserting a polymeric stent having a portion in a collapsed position that can be reverted, by heating, to an expanded position. Following insertion, the stent is heated sufficiently to revert the portion in the collapsed position to the expanded position.
- the method may further include heating the stent having the portion in the expanded position sufficiently to soften the stent, and removing the softened stent from the cavity or lumen.
- the stent may be, for example, a coiled elongated element (for example, a rod, a tape or flattened tube) and the heating of the stent prior to removal allows the stent to be removed in a substantially uncoiled state.
- This method provides ease of removal, for example, for removing prostatic stents that have been inserted on an interim basis.
- the heating may be performed, for example, on a delivery tube.
- the portion of the stent is at the end of the stent and may be flared when in the expanded position. In other embodiments, for example, the portion of the stent is not at an end of the stent.
- the invention features a polymeric stent including metal particles.
- a portion of the stent has a collapsed position that can be reverted to an expanded position by heating.
- the heating may be performed using inductive heating to revert the portion in the collapsed position to the expanded position.
- the invention features a stent having an exterior surface that includes a plurality of protruding elements that extend outwardly from the surface.
- the protruding elements may be useful in helping the stent retain its position, for example, after insertion into the prostatic urethra.
- the protruding elements are formed of monofilament.
- the monofilament may include a plurality of constrictions along its length.
- the stent is a polymeric stent and the stent has a portion that has a collapsed position that can be reverted to an expanded position by heating above a first temperature subsequent to insertion of the stent into a cavity or lumen.
- the invention features an implantable endoprothesis including a tubular member that includes a polymeric material.
- the tubular member has a wall having a first transverse dimension and a first longitudinal length, measured when at the first transverse dimension, sized for delivery into a lumen.
- the tubular member can be expanded to a second transverse dimension that is at least about fifty percent larger than the first transverse dimension within the lumen, the first and second transverse dimensions being measured from an outer surface of the wall of the tubular member.
- the tubular member also has a second longitudinal length, measured when at the second transverse dimension. After expansion from the first transverse dimension to the second transverse dimension, the second longitudinal length decreases by less than about fifty percent, measured relative to the first longitudinal length.
- the tubular member has a wall thickness, measured from an inner surface of the wall to the outer surface of the wall, and the wall thickness decreases by greater than about twenty percent, e.g., greater than about thirty percent, greater than about fifty percent, greater than about seventy-five percent, or greater than eighty-five percent, after expansion from the first transverse dimension to the second transverse dimension.
- the second longitudinal length decreases by less than about twenty percent, measured relative to the first longitudinal length.
- the tubular member can be, for example, approximately circular in transverse cross-section, or the tubular member can have other transverse shapes, e.g., non-circular, e.g., elliptical.
- the polymeric material has a softening temperature from about 40° C. to about 60° C., e.g., 45, 50, 55, or 58° C.
- the polymeric material can be cross-linked, non-cross-linked, a shape memory polymer, or a non-shape memory polymer.
- the polymeric material is, for example, polycyclooctene (PCO), a styrenic elastomer, a styrenic block copolymer, a styrene-butadiene rubber, a polyolefin, trans-isoprene, or blends of these materials.
- the polymeric material can include a filler, e.g., a radio-opaque agent, e.g., bismuth carbonate, barium sulfate, or mixtures of these materials.
- a filler e.g., a radio-opaque agent, e.g., bismuth carbonate, barium sulfate, or mixtures of these materials.
- Other fillers includes, for example, a thermal conductor, e.g., a boron nitride, other ceramics, or a metal.
- the tubular member is, for example, substantially straight before it is expanded.
- the tubular member is curved after it is expanded and/or the outer surface of the wall of the tubular member includes a protruding element that extends outwardly from the outer surface after the tubular member is expanded.
- the wall of the tubular member includes at least one aperture defined therein.
- the plastic has a elastic modulus of greater than about 50,000 psi, e.g., greater than about 75,000, greater than about 150,000, greater than about 250,000, or greater than about 500,000 psi.
- the invention features a method of treating a patient.
- the method includes placing the endoprosthesis just discussed on a delivery system.
- the delivery system then is used to deliver the endoprosthesis a lumen, e.g., a pulmonary lumen, an esophageal lumen, a biliary lumen, an enteral lumen, a ureteral lumen, and a urethral lumen.
- the endoprosthesis then is heated and expanded within the lumen.
- the delivery system includes a balloon catheter.
- FIGS. 1A and 1B are side views of a portion of a stent in a small diameter and expanded condition, respectively.
- FIGS. 1C and 1D are cross-sectional views of a portion of a stent in a small diameter and expanded condition, respectively.
- FIGS. 2A-2C illustrate delivery of a stent into a body lumen.
- FIG. 3 is a plot of storage modulus as a function of temperature.
- FIG. 3A is a plot of storage modulus as a function of temperature for samples of PCO with varying degrees of crosslinking
- FIG. 3B is a WAXS 2 ⁇ plot for samples of PCO with varying degrees of crosslinking.
- FIG. 4 illustrates manufacture and use of a stent.
- FIG. 5 illustrates manufacture and use of a stent.
- FIG. 6 is a perspective view of a stent with an end in an expanded position.
- FIG. 7 is a side view of the stent shown in FIG. 6 .
- FIG. 8 is a side view of the stent shown in FIG. 6 with the end in a collapsed position.
- FIG. 8A is a graph of heat flow as a function of temperature for several POSS polyurethanes.
- FIG. 8B is a graph of storage modulus as a function of temperature for several POSS polyurethanes.
- FIG. 9 is a perspective view of a wrapping fixture.
- FIG. 10 is a cross-sectional view of a restricted prostatic urethra.
- FIG. 11 is a cross-sectional view illustrating delivery of a stent to the prostatic urethra.
- FIG. 12 is a cross-sectional view of a prostatic stent deployed in the urethra.
- FIG. 13 is a side view of an alternative delivery system.
- FIG. 13A-13B are side views of an alternative delivery system.
- FIG. 14 is a cross-sectional view illustrating removal of a prostatic stent.
- FIG. 15 is a graph of storage modulus (E′) VS temperature for PCO.
- FIG. 16 is a graph of storage modulus (E′) VS temperature for PVAc/PVDF/PMMA blends.
- FIG. 17 is a side view of an alternative stent with two ends portions in expanded positions.
- FIG. 18 is the stent shown in FIG. 17 with ends in collapsed positions.
- FIG. 19 is a side view of an alternative stent with three portions in expanded positions.
- FIG. 20 is a side view of an alternative stent made with a flattened tube.
- FIG. 21 is a cross-sectional view of the stent shown in FIG. 20 , taken along 21 - 21 .
- FIG. 22 is a side view of an alternative stent made with a tape.
- FIG. 23 is a perspective view of a stent with a plurality of protruding elements, the end of the stent is in an expanded position.
- FIG. 24 is a side view of the stent shown in FIG. 23 .
- FIG. 25 is a side view of the stent shown in FIG. 23 with the end in a collapsed position.
- FIG. 26 is a perspective view of an alternative stent with a plurality of protruding elements, the end of the stent in an expanded position.
- FIG. 27 is a side view of the stent shown in FIG. 26 .
- FIG. 28 is a side view of the stent shown in FIG. 26 with an end in a collapsed position.
- FIGS. 29 and 30 are perspective views of a tubular stent in an unexpanded state and in an expanded state, respectively.
- FIGS. 31 and 32 are perspective views of an elongated, tubular stent in an unexpanded state and in an expanded state, respectively.
- FIG. 33 is a perspective view of a curved tubular stent in an expanded state.
- FIG. 34 is a perspective view of a tubular stent in an expanded state that has flared ends.
- FIG. 35 is a perspective view of an elongated tubular stent having an outer surface that includes a plurality of projections.
- a stent 10 includes a polymer body 12 generally defining a tube.
- the stent includes open areas 14 .
- the open areas are relatively small and defined as slots cut through the wall of the stent.
- the slots are widened to diamond-like shapes.
- the expansion mechanism of the stent utilizes a deformation (arrows) of the wall material about the open areas. As illustrated, the expansion results in a generally regular, symmetric, geometric pattern that resists and distributes inward compression of the stent by forces imposed by the lumen wall.
- the polymer does not substantially flow or thin out on expansion, so that a reliable expansion geometry and wall thickness can be achieved.
- Other stent constructions are suitable.
- filament-form stents in which filaments of polymer material are arranged to define a generally tubular structure can be used. Open areas are defined between the filaments.
- An example of a stent design including helical filaments is provided in Wallsten, U.S. Pat. No. 4,655,771. Suitable aperture wall designs are also described in Palmaz U.S. Pat. No. 4,733,665.
- Another suitable arrangement is exemplified by the Express stent, commercially available from Boston Scientific, Natick, Mass.
- the stent is delivered utilizing a catheter 24 , which includes a catheter body 25 that carries a balloon 26 at its distal end.
- the catheter includes an inflation apparatus 28 such as a syringe or pump apparatus which can be used to inject and circulate inflation fluid into the catheter, where it is directed by a lumen to the interior of the balloon so that the balloon can be inflated.
- the inflation apparatus can include a heating apparatus 30 , to heat the inflation fluid directed to the balloon.
- the catheter is delivered into a vessel 20 including body lumen 21 in a patient to the site of an obstruction 31 typically utilizing a guidewire 32 .
- the guidewire 32 extends through a lumen within the body 25 of the catheter.
- the stent 10 is positioned over the inflatable balloon 26 .
- the balloon is initially in a small diameter deflated condition.
- the stent is in a small diameter condition over the balloon.
- the balloon is inflated by actuating the inflation apparatus 28 .
- the inflation fluid is heated to heat the polymer body of the stent 10 .
- the stent is expanded into contact with body lumen 21 .
- the stent can be expanded simultaneously with the widening of the obstructed region.
- the temperature of the inflation fluid is typically decreased to reverse the softening of the stent body 10 .
- the temperature of the stent has been reduced in this manner, it remains implanted in the vessel to resist vessel recoil and reduce restenosis after the balloon is deflated and the catheter is removed from the body.
- Suitable polymers include those that maintain stent geometry under expansion conditions, allowing for intricate stent geometries such as apertured tubes having high open area to wall ratios.
- the stent can be expanded without fracture or substantial irreversible stress relaxation or creep.
- the stent is heated to or above the melt or glass transition temperature during expansion.
- the polymer is in a softened state. In this state, the polymer can be predictably deformed, typically about aperture regions during expansion.
- the soft condition permits proper apposition of the stent to the lumen wall without kinking and without damage due to excessive stiffness, which could straighten the lumen from its native curvature and lead to dissections or other trauma.
- the polymer After the stent is fully expanded and cooled, the polymer substantially sets in the proper apposition, e.g. about a native curvature. Excessive recoil of the stent to a linear configuration is avoided, reducing trauma about the vessel. At the same time, the polymer can have some elastomeric properties in the cooled, hardened state so that the stent can flex with natural vessel motion. After cooling, the stent exhibits sufficient resistance to inward radial force to reduce restenosis due to, e.g., lumen wall recoil. The polymer has sufficient strength so that the stent wall can be kept relatively thin while resisting restenosis from lumen wall forces.
- Suitable polymers include elastomers that are crosslinked, crystalline, or amorphous, e.g. plasticized PVC, e.g., PVC plasticized with a monomeric plasticizer, e.g., a phthalate, or a polymeric plasticizer.
- the crosslinked and/or crystalline nature is sufficient to resist excessive creep or stress relaxation when the polymer is heated and expanded.
- the polymer can be crosslinked so that it exhibits the desired elastomeric properties but not crosslinked to the degree that it becomes excessively brittle. Too little crosslinking does not establish sufficient resistance to flow during heating and expansion to maintain stent geometry.
- crosslinking can be adjusted to adjust the melt or glass transition temperature and transition temperature range.
- a narrow melt transition range is desirable, e.g. 5° C. or 10° C. or less.
- Crosslinking can be achieved by application of radiation such as e-beam, UV, gamma, x-ray radiation or by heat-activated chemical crosslinking techniques.
- Chemical crosslinking agents include peroxides, such as benzoyl peroxide or dicumyl peroxide (DCP), and azo compounds, such as 2,2′-azobis(2,4-dimethyl valeronitrile) or 2,2′-azobis[N-(2-propenyl)-2-methylpropionamide]. Radiation techniques provide the advantage that the polymer typically does not have to be substantially heated to achieve crosslinking.
- An intricate aperture pattern provided in a stent precursor tube can be maintained and heat-induced flow of pre-crosslinked polymer can be avoided.
- an exposure of about 50-300, e.g. 250 kilograys typically provides sufficient crosslinking Melting and crystallization temperatures are measured using a differential scanning calorimetry.
- the polymer can have elastomeric properties in the melted or softened state. Elastomeric properties at melt or glass transition can be investigated by measuring the modulus of elasticity or storage modulus as a function of temperature and determining the elastomeric nature of the material in the desired expansion temperature range. Referring to FIG. 3 , a plot of storage modulus as a function of temperature is provided. Storage modulus decreases as the material is heated. At the melt or glass transition, a plateau “P” is typically consistent with an elastomeric nature. At much higher temperatures, the modulus drops off more quickly, indicating a material which could flow under pressure. To determine storage modulus, a dynamic mechanical analyzer (Perkin Elmer) can be used.
- Perkin Elmer Perkin Elmer
- Dynamic mechanical analysis was carried out in tensile mode at an operating frequency of 1 Hz, a static force of 10 mN, and oscillation amplitude of 5 ⁇ m (approximately 0.1% strain) and an automatic tension setting of 125%. Temperature ramps were conducted at 4° C./minute over the range ⁇ 100° C. to 100° C.
- the storage modulus of neat PCO begins to decrease sharply to about 2 MPa at the completion of melting at 71° C. As found with DSC, this transition temperature is observed mechanically to decrease with increasing degree of crosslinking. For temperatures greater than T m , the modulus of neat PCO, trace (i), continues to decrease to a point where the material flows like a viscous liquid, not showing a persistent rubbery plateau ( FIG. 3 ). This feature hampers the applicability of neat PCO for use as a shape memory polymer due to an inability to be deformed as a rubber above T m without rapid stress relaxation.
- cured PCO which contains just 1% peroxide, represented by trace (ii), will allow significant shape memory effects owing to its persistent rubbery plateau above 72° C. As the amount of peroxide increases, the rubbery plateau modulus increases, allowing for enhanced mechanical energy storage, but the transition temperature and the steepness of the transition decrease.
- trace (v) in FIG. 3A the thermomechanical response that is observed is inconducive to shape memory effects as the fixing (crystallization) temperature is lower than room temperature so that shape fixing would require subambient cooling and the temporary shape would be expected to drift via partial melting. In addition, the melting transition is too broad for dramatic strain recovery to be expected.
- Suitable polymers include elastomers that are typically crosslinked and/or crystalline and exhibit melt or glass transitions at temperatures that are above body temperature and safe for use in the body, e.g. at about 40 to 50° C. Suitable polymers can have an elastic modulus of about 60,000 or 70,000 psi or more at 25° C. (ASTM D638M). Such polymers may have a variety of room temperature moduli, from rigid glassy materials having storage moduli of several GPa to compliant rubbers with moduli as low as tens of MPa. Moreover, the moduli may tuned over the range 0.5 ⁇ E ⁇ 10 MPa, as dictated by the end application.
- Suitable polymers include polynorbornene, polycaprolactone, polyenes, nylons, polycyclooctene (PCO), blends of PCO and styrene-butadiene rubber, polyvinyl acetate/polyvinylidinefluoride (PVAc/PVDF), blends of PVAc/PVDF/polymethylmethacrylate (PMMA), polyurethanes, styrene-butadiene copolymers, polyethylene (particularly, crosslinked polyethylene), trans-isoprene, block copolymers of polyethylene terephthalate (PET), blends of polycaprolactone and n-butylacrylate, and PVC, e.g., plasticized PVC, e.g., PVC plasticized with a monomeric plasticizer, e.g., a phthalate, or a polymeric plasticizer.
- PVC polyvinyl acetate/polyvinylidinefluoride
- a suitable PVAc/PVDF tube is formed by compounding 60-80 parts (by weight) PVAc (B-100, mw 500,000, ChemPoint, Cleveland, Ohio) with 40 to 20 parts PVDF (Grade 1010, Solvay Fluoropolymers, Houston, Tex.).
- the PVAc/PVDF is a crystalline material that can be utilized with or without crosslinking.
- the polymer body can be made of mixtures of polymers or multiple polymer layers.
- the polymer forming the stent body can be compounded to include a drug, which elutes from the polymer, or radiopaque material.
- the structural polymer body can be coated with other polymers to carry drug or control drug delivery from the structural polymer.
- the polymer body can also exhibit shape memory properties. This feature of the polymer can be used in combination with the expansion properties discussed above.
- the polymer can be configured to remember an enlarged or reduced diameter configuration.
- the stent can be delivered into the body, and expanded by a combination of heat and radial pressure as described above. After a time, the stent can be retrieved by reheating the stent.
- the heating causes the stent to revert its small diameter condition.
- the remembered stent diameter is less than the vessel diameter and the stent can be more easily removed from the vessel.
- Such an application might be useful, for example, for a stent delivered into the prostate where removal and replacement is desirable.
- a polymer tube 40 is constructed by extrusion or molding a suitable polymer to an initial diameter di which in the same or greater than the target lumen diameter.
- the strands can be formed by extrusion, followed by arranging the strands into a tube, e.g. by weaving or knitting.
- the tube wall is then cut to provide a pattern of open areas in a desirable geometric pattern, e.g. by laser cutting.
- the polymer can be recrystallized or crosslinked, if necessary.
- the catheter can be, e.g. an angioplasty balloon catheter with a relatively non-distendable inflating balloon suitable for expansion of occluded regions of a vascular lumen.
- the balloon may include a polymer such as PET, which has a burst pressure of about 1.5 to 5 atm or more.
- the stent can be heated by heating the balloon inflation fluid.
- the balloon inflation fluid can be heated by e.g., heating the fluid delivery device outside the body using e.g., resistive heating tape.
- the catheter can carry a heating device.
- a resistive heater or RF heater can be provided in the interior of the balloon.
- a heated balloon catheter is described in Abele et al. U.S. Pat. No. 5,496,311 and U.S.
- Vessel heating as described in the '311 can be used in combination with stent delivery as discussed herein.
- the stent can be heated directly.
- the polymer can be compounded to include a material, such as magnetic particles, which are susceptible to heating by magnetic effects, such as hysteresis effects.
- a magnetic field can be imposed on the stent body by a source on a catheter or outside the body. Particles are available as the Smartbond System from Triton Systems, Inc., Chelmsford, Mass. Heating by magnetic effects is discussed in U.S. Pat. No. 6,056,844.
- the stent can also be heated during delivery without applying expansion force to soften the stent, improving its flexibility and thus improving delivery to a treatment site through a tortuous vessel path.
- a polycyclooctene polymer (Vistenemer 8012 pellets, mw 90,000, Degussa, N.J.) is melt processed in an extruder to produce a tube having dimensions of about 0.118 inch O.D. and 0.070 inch I.D. (wall thickness about 0.024 inch). The tube is cut to a length of about 4 cm. The tube is subject to UV excimer laser ablation cutting to provide an aperture pattern of rectangular slots having a width of about 0.2 mm and a length of about 8 mm. Beam energy and pulse rate are selected to avoid substantial heating or melting of the polymer.
- the polymer can be compounded with about 10% TiO 2 (T8141, Dupont) to enhance absorption of laser radiation.
- a suitable pattern is consistent with the Express stent (commercially available from Boston Scientific, Natick, Mass.). (Alternatively, a pattern as described in Palmaz U.S. Pat. No. 4,733,665 can be used.)
- the tube is heated to a temperature below its melt point, e.g., to about 39 to 40° C. in a water bath and expanded by balloon catheter to a diameter of about 5 mm and positioned on a mandrel (PTFE tube) to maintain the expanded shape and diameter.
- the tube is then cooled to room temperature.
- the polymer is then crosslinked by e-beam radiation at 250 K Grays (Steris Isomedics Services, Northborough, Mass.). Crosslinking fixes the stent in the condition.
- the crosslinked PCO has an elastic (Youngs) modulus of about 74945 psi at about 25° C. (ASTM D638M)).
- the stent is heated to the polymer melt temperature, about 45° C. and collapsed over a deflated balloon (diameter of about 2 mm) with a 4 mm inflated maximum diameter and 2 cm length.
- a suitable balloon catheter is a 75 cm Meditech UltraThin Catheter, available from Boston Scientific, Natick, Mass.).
- the balloon and stent are immersed in a water bath of about 42 to 45° C. and water the same temperature is used to inflate the balloon.
- the stent is expanded to about 4 mm diameter (ID) at an inflation pressure of about 1 to 1.5 atm (measured at the delivery syringe). After expansion, the heating is discontinued and the balloon and inflation fluid allowed to cool to body temperature (while the balloon remains inflated). Alternatively, a cooled contrast fluid can be circulated to the balloon.
- ID mm diameter
- a cooled contrast fluid can be circulated to the balloon.
- the stent exhibits no visible reduction in wall thickness or irregular flow of polymer into the stent open areas.
- the stent in the heated, expanded state, the stent can be bent around a mandrel of about 0.75 cm radius without kinking. After the stent is cooled, it maintains the curved form.
- stent 100 includes a coiled rod 120 composed of a polymer.
- Stent 100 includes an elongated portion 140 having a general diameter d 1 and an end portion 160 having a maximum diameter d 2 .
- General diameter d 1 may be between about 3 mm and about 25 mm, more preferably between about 6 mm and about 14 mm
- maximum diameter d 2 may be between about 7 mm and about 30 mm, more preferably between about 10 mm and about 17 mm.
- the stent When stent 100 is designed, for example, for insertion into a urethra, the stent may have an overall length, for example, of between about 3 mm and about 15 mm, preferably between about 6 mm and 10 mm, and end portion 160 may have a length, for example, of from about 3 mm to about 15 mm, preferably from about 5 mm to about 10 mm. End portion 160 is in a flared position. Referring to FIG. 8 , stent 100 is shown with end portion 160 in a collapsed position that can be reverted with heating to the expanded position shown in FIGS. 6 and 7 .
- the portion of the stent in the collapsed position that can be reverted to the expanded position is, for example, greater than 5%, 10%, or even 25% of the overall length L of the stent, and less than 80% or 65% of the overall length L of the stent.
- the of the overall length L of the stent may be between 10% and 65% of the overall length L of the stent.
- the polymers preferably are cross-linked and/or crystalline elastomers that have melt or glass transition temperatures that are above body temperature, for example, greater than 45° C. or 55° C.
- the degree of cross-linking can be used to adjust, for example, the melt or glass transition temperature, and range, of the polymer.
- the polymer preferably has a relatively narrow, for example, less that 5° C. or 10° C., melt or glass transition temperature range.
- the polymer preferably has elastomer properties in its melted or softened state.
- Preferred polymers have an elastic modulus, for example, of about 60,000 psi or 70,000 psi or more at 25° C. (ASTM D638).
- polymers include polynorbornene and copolymers of polynorbornene, blends of polynorbornene with KRATON® (thermoplastic elastomer) and polyethylene, styrenic block copolymer elastomers (e.g., styrene-butadiene), polymethylmethacrylate (PMMA), polyethylene, polyurethane, polyisoprene, polycaprolactone and copolymers of polycaprolactone, polylactic acid (PLA) and copolymers of polylactic acid, polyglycolic acid (PGA) and copolymers of polyglycolic acid, copolymers of PLA and PGA, polyenes, nylons, polycyclooctene (PCO), polyvinyl acetate (PVAc), polyvinylidene fluoride (PVDF), blends of polyvinyl acetate/polyvinylidine fluoride (PVAc/PVDF), blends of polyvin
- the polymers above are also useful for the stents of FIGS. 1 , 4 and 5 .
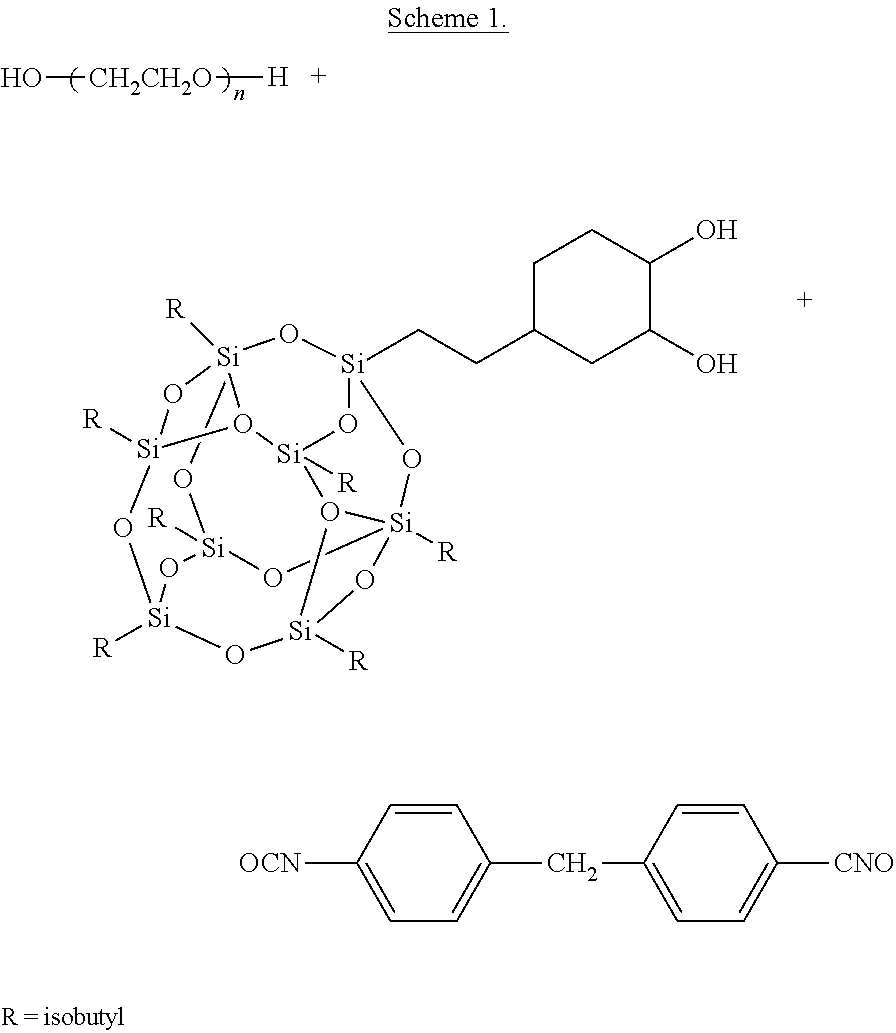
- Particular polyurethanes are made by reacting (A) a polyol, (B) a chain extender dihydroxyl-terminated POSS and (C) a diisocyanate, where POSS stands for a polyhedral oligomeric silsesquioxane diol.
- the polyol (A) can be polyethylene glycol (PEG), polycaprolactone (PCL), polycyclooctene (PCO), trans-1,4 butadiene, transisoprene, polynorbornene diol and polymethacrylate copolymer
- the chain extender (B) can be TMP cyclopentyldiol-POSS, TMP cyclohexyldiol-POSS, TMP isobutyldiol-POSS, trans-cyclohexanediolcyclohexane-POSS, or transcyclohexanediolisobutyl-POSS
- the diisocyanate (C) can be selected from a large number of diisocyanates and is preferably 4,4′ diphenyl methylene diisocyanate (MDI).
- diisocyanates (C) that are suitable for use in the synthesis of hybrid polyurethane SMPs include: toluene-2,4-diisocyanate (TDI), toluene-2,6diisocyanate, hexamethylene-1,6-diisocyanate (HDI), 4,4′ diphenylmethane diisocyanate (MDI), isophorone diisocyanate (IPDI), and hydrogenate 4,4′-diphenylmethane diisocyanate (H12MDI).
- TDI toluene-2,4-diisocyanate
- HDI hexamethylene-1,6-diisocyanate
- MDI 4,4′ diphenylmethane diisocyanate
- IPDI isophorone diisocyanate
- H12MDI hydrogenate 4,4′-diphenylmethane diisocyanate
- FIG. 8A A graph of heat flow as a function of temperature for several POSS polyurethanes is shown in FIG. 8A and a graph of storage modulus as a function of temperature for several POSS polyurethanes is shown in FIG. 8B .
- This scheme shows an example of synthesis of TPU using polyethylene glycol as polyol, TMP Isobutyldiol-POSS as chain extender to react with 4,4′ diphenyl methylene diisocyanate in toluene.
- This scheme shows an example of synthesis of TPU using polycaprolactone diol as polyol, TMP Isobutyldiol-POSS as chain extender to react with 4,4′ diphenyl methylene diisocyanate.
- This scheme shows an example of synthesis of TPU using polyocyclooctene as polyol, TMP Isobutyldiol-POSS as chain extender to react with 4,4′ diphenyl methylene diisocyanate.
- any of the polymers mentioned above may be filled with, for example, nanoparticles of clay and silica to, for example, increase the modulus of the plastic.
- Dispersing agents and/or compatibilizing agents may be used, for example, to improve the blending of polymers and the blending of polymers with fillers.
- Dispersing agents and/or compatibilizing agents include, for example, ACRAWAX® (ethylene bis-stearamide), polyurethanes and ELVALOY® (acrylic-functionalized polyethylene).
- the polymers can be cross-linked by application of radiation such as e-beam, UV, gamma, x-ray radiation or by heat-activated chemical crosslinking techniques. Radiation techniques provide the advantage that the polymer typically does not have to be substantially heated to achieve crosslinking. For e-beam radiation, an exposure of about 200-300, e.g. 250 kilograys, typically provides sufficient crosslinking.
- wrapping fixture 200 can be used for making coiled stent 100 .
- Wrapping fixture 200 includes a base 220 for support, a mandrel 240 with a flared end 260 , slits 280 for fixing the plastic rod 120 , an aperture 290 for fixing the plastic rod 120 on the non-flared end and a fixing screw 299 for releasably fixing mandrel 240 to wrapping fixture 200 .
- the rod 120 is inserted into aperture 290 that is machined through mandrel 240 to fix the rod at the starting end.
- the rod 120 is tightly wrapped around mandrel 240 , including flared end 260 .
- plastic rod 120 is pushed into slits 280 .
- the overall length of the stent 100 may be, for example, about 2 mm to about 150 mm or more. The overall length required depends upon the application.
- the plastic rod 120 now fixed in place on the mandrel 240 , is heated to above the softening point of the material and maintained at that temperature long enough to anneal the rod and fix the shape. Typically, the time required to fix the shape is from about 0.25 hr to 10 hr or more.
- stent 100 is removed from mandrel 240 .
- the flared end of the coil is tapered down and collapsed so that the diameter along the entire length of the stent is approximately d 1 . Collapsing the flared end 160 of stent 100 allows for ease of insertion, for example, into a restricted prostatic urethra.
- a 56:24:20 mixture of PVAc/PVDF/PMMA is dry bended and loaded into the hopper of an extruder.
- the PVAc is grade B-100
- the PVDF is Solvay SOLEF® 1010
- the PMMA is Atofina PLEXIGLAS® V045.
- the mixture is melt processed to produce 1.27 mm (0.05 inch) monofilament.
- the rod is made into a coil by winding it around wrapping fixture 200 .
- the fixture and the rod are immersed into a 50° C. water bath. At this temperature, the rod becomes malleable enough to wind easily around the mandrel and secured in place to prohibit the uncoiling of the helical shape.
- the mandrel is removed from the fixture with the stent locked in place and placed into an oven at 110° C. for one hour to anneal the stent.
- This annealing process locks the permanent shape of the coil.
- the mandrel and coil are cooled to room temperature, and the stent is removed from the mandrel.
- the stent had on overall length of approximately 73 mm and a flared end portion length of approximately 7 mm.
- the diameter d 1 of the body is approximately 6 mm and the maximum diameter d 2 of the flare on the open end is approximately 11 mm.
- the flared end of the coil is tapered down with brief heating to 50° C. and manipulation, followed by cooling, so that the diameter is approximately 6 mm along the entire length of the stent.
- a 70:30 mixture of PVAc/PVDF is dry bended and loaded into the hopper of an extruder.
- the mixture is melt processed to produce 1.27 mm (0.05 inch) monofilament.
- the rod is made into a coil by winding it around wrapping fixture 200 .
- the fixture and the rod are immersed into a 50° C. water bath. At this temperature, the rod becomes malleable enough to wind easily around the mandrel and secured in place to prohibit the uncoiling of the helical shape.
- the mandrel is removed from the fixture with the stent locked in place and placed into an oven at 110° C. for one hour to anneal the stent. This annealing process locks the permanent shape of the coil.
- the mandrel and coil are cooled to room temperature, and the stent is removed from the mandrel.
- the stent had on overall length of approximately 73 mm and a flared end portion length of approximately 7 mm.
- the diameter d 1 of the body is approximately 6 mm and the maximum diameter d 2 of the flare on the open end is approximately 11 mm.
- the flared end of the coil is tapered down with brief heating to 50° C. and manipulation, followed by cooling, so that the diameter is approximately 6 mm along the entire length of the stent.
- stent 100 may be, for example, inserted into restricted urethra 300 on delivery tube 320 .
- end portion 160 is in a collapsed position.
- warm water e.g., 45° C.-55° C.
- Heating reverts the collapsed end 160 to a flared, expanded position ( FIG. 12 ).
- the flared, expanded position allows stent 100 to remain fixed in position, for example, in the prostatic urethra.
- delivery tube 320 is a long cylindrical tube into which a ureteral scope 380 is inserted.
- Delivery tube 320 has a distal end 330 over which stent 100 is placed.
- Delivery 320 is fitted with a side port 350 including a stopcock 370 through which saline can be flushed for irrigation.
- Delivery tube 320 with stent 100 in place is delivered into, for example, the prostatic urethra with the aid of a ureteral scope.
- hot saline is flushed through port 350 to revert the collapsed end 160 to a flared, expanded position ( FIG. 12 ).
- the flared, expanded end allows stent 100 to remain fixed in position, for example, in or adjacent the prostatic urethra or external sphincter between the prostate and the bladder to prevent migration.
- the direction of the flare can, of course, be oriented in other directions.
- the scope and delivery tube 320 are withdrawn, leaving stent 100 in place.
- an alternative delivery system is illustrated that includes a tube 320 with a screw on tip 331 onto which a stent 100 is placed after collapsing the end portion 160 .
- the assembly is inserted into, for example, the prostatic urethra.
- Stent 100 may be, for example, inserted into restricted urethra 300 on balloon catheter (not shown). During insertion, end portion 160 is in a collapsed position. After insertion, warm water is flushed through the guide wire lumen of the balloon catheter to flood the area and to heat the stent. Heating of the stent by the water reverts the collapsed end 160 to a flared, expanded position The flared, expanded position allows stent 100 to remain fixed in position, for example, in the prostatic urethra. If there is an obstruction in the lumen into which the stent is deployed the stricture can be dilated using the balloon to help the stent open fully and maintain a uniform diameter inside the vessel.
- coiled stent 100 with an end 160 in the expanded position can be removed with the aid of a catheter equipped with a grasping device 360 and a ureteral scope 380 for visualizing stent 100 in, for example, the prostatic urethra.
- a catheter equipped with a grasping device 360 and a ureteral scope 380 for visualizing stent 100 in, for example, the prostatic urethra.
- end 400 of stent 100 has been visualized with ureteral scope 380
- the stent is grasped with grasping device 360 .
- ureteral scope 380 is removed from the catheter and is replaced with a heating device (not shown), e.g. a catheter. Heating stent 100 above the softening point of the polymer, e.g., from about 45° C. to about 55° C. for polycyclooctene (PCO), and pulling the end 400 of stent 100 through the orifice 420 allows the stent to be removed in
- FIG. 11 shows heating of stent 100 with a warm liquid on a delivery tube
- heating may be accomplished with the use of IR, RF or inductive heating.
- stent 10 into other body lumens or cavities.
- other body lumens or cavities include the biliary duct, cystic duct, a ureter, a bulbar urethra or a hepatic duct.
- the modulus of polycyclooctene (PCO) that can, for example, be the polymer of stent 100 is shown as a function of temperature. Below approximately ⁇ 65° C. (T g , region A), PCO exists a rigid, glassy polymer. Above T g , but below T m , PCO exists as a flexible elastomer (region B). Above T m , PCO exists as a relatively low modulus elastomer. Above T m , for example, stent 100 composed of PCO can be removed from a lumen or cavity of the body, the prostatic urethra, for example, in a substantially uncoiled state.
- PCO polycyclooctene
- the modulus of a ternary blend of PVAc/PVDF/PMMA that can, for example, be the polymer of stent 100 is shown as a function of temperature. Adding PMMA offers the advantage, for example, of increasing the modulus of the blend.
- Stent 100 reverts from its collapsed position to its expanded position upon re-heating above T m (region C) because the modulus of the material lowers sufficiently to release the residual stress that was “frozen” into stent 100 during the rapid cooling.
- FIGS. 17-28 show other examples of stents.
- coiled stent 600 has two end portions 620 and 660 and a central portion 640 in expanded positions. All three portions may be collapsed (not shown), and then reverted to expanded positions.
- coiled stent 700 made from a flattened tube 710 has an end portion 720 in an expanded position.
- the flattened tube can, for example, add strength to the stent.
- the tube can have a major diameter, for example, of between about 1.0 mm to about 3.0 mm, more preferably between about 1.5 mm to about 2.25 mm, and major inner diameter, for example, of between about 0.5 mm to about 2.5 mm, more preferably between about 1.25 mm and about 1.75 mm.
- End portion 720 may be collapsed (not shown) and then reverted to an expanded position.
- flattened tube 710 has an interior 740 that may be filled with, for example, a medicament.
- the medicament may be triclosan or salicylic acid. Release of medicament from flattened tube 710 , for example, may reduce the risk of infection. Interior 740 may also be filled with, for example, paclitaxel or mitoxantrone. Release of the these medicaments from interior 740 may be, for example, useful for treating prostate cancer and reducing prostatic hyperplasia.
- any filler e.g., a therapeutic agent
- a therapeutic agent can be a genetic therapeutic agent, a non-genetic therapeutic agent, or cells.
- Therapeutic agents can be used singlularly, or in combination.
- Therapeutic agents can be, for example, nonionic, or they may be anionic and/or cationic in nature.
- non-genetic therapeutic agents include: (a) anti-thrombotic agents such as heparin, heparin derivatives, urokinase, and PPack (dextrophenylalanine proline arginine chloromethylketone); (b) anti-inflammatory agents such as dexamethasone, prednisolone, corticosterone, budesonide, estrogen, sulfasalazine and mesalamine; (c) anti-neoplastic/antiproliferative/anti-miotic agents such as paclitaxel, 5-fluorouracil, cisplatin, vinblastine, vincristine, epothilones, endostatin, angiostatin, angiopeptin, monoclonal antibodies capable of blocking smooth muscle cell proliferation, and thymidine kinase inhibitors; (d) anesthetic agents such as lidocaine, bupivacaine and ropivacaine; (e) anti-coagulant
- Exemplary genetic therapeutic agents include anti-sense DNA and RNA as well as DNA coding for: (a) anti-sense RNA, (b) tRNA or rRNA to replace defective or deficient endogenous molecules, (c) angiogenic factors including growth factors such as acidic and basic fibroblast growth factors, vascular endothelial growth factor, epidermal growth factor, transforming growth factor ⁇ and ⁇ , platelet-derived endothelial growth factor, platelet-derived growth factor, tumor necrosis factor ⁇ , hepatocyte growth factor and insulin-like growth factor, (d) cell cycle inhibitors including CD inhibitors, and (e) thymidine kinase (“TK”) and other agents useful for interfering with cell proliferation.
- angiogenic factors including growth factors such as acidic and basic fibroblast growth factors, vascular endothelial growth factor, epidermal growth factor, transforming growth factor ⁇ and ⁇ , platelet-derived endothelial growth factor, platelet-derived growth factor, tumor necrosis
- BMP's bone morphogenic proteins
- BMP-3, BMP-4, BMP-5, BMP-6 and BMP-7 are any of BMP-2, BMP-3, BMP-4, BMP-5, BMP-6 and BMP-7.
- These dimeric proteins can be provided as homodimers, heterodimers, or combinations thereof, alone or together with other molecules.
- molecules capable of inducing an upstream or downstream effect of a BMP can be provided.
- Such molecules include any of the “hedgehog” proteins, or the DNA's encoding them.
- Vectors for delivery of genetic therapeutic agents include viral vectors such as adenoviruses, gutted adenoviruses, adeno-associated virus, retroviruses, alpha virus (Semliki Forest, Sindbis, etc.), lentiviruses, herpes simplex virus, replication competent viruses (e.g., ONYX-015) and hybrid vectors; and non-viral vectors such as artificial chromosomes and mini-chromosomes, plasmid DNA vectors (e.g., pCOR), cationic polymers (e.g., polyethyleneimine, polyethyleneimine (PEI)), graft copolymers (e.g., polyether-PEI and polyethylene oxide-PEI), neutral polymers PVP, SP1017 (SUPRATEK), lipids such as cationic lipids, liposomes, lipoplexes, nanoparticles, or microparticles, with and without targeting sequences such as the protein transduction domain (PTD).
- Cells for use include cells of human origin (autologous or allogeneic), including whole bone marrow, bone marrow derived mono-nuclear cells, progenitor cells (e.g., endothelial progenitor cells), stem cells (e.g., mesenchymal, hematopoietic, neuronal), pluripotent stem cells, fibroblasts, myoblasts, satellite cells, pericytes, cardiomyocytes, skeletal myocytes or macrophage, or from an animal, bacterial or fungal source (xenogeneic), which can be genetically engineered, if desired, to deliver proteins of interest.
- progenitor cells e.g., endothelial progenitor cells
- stem cells e.g., mesenchymal, hematopoietic, neuronal
- pluripotent stem cells fibroblasts, myoblasts, satellite cells, pericytes, cardiomyocytes, skeletal myocytes or macrophage, or
- coiled stent 800 made from tape 810 has an end portion 820 that may be collapsed and then reverted to an expanded position.
- Tape 810 may have a thickness from about 0.5 mm to about 2.0 mm, more preferably from about 0.75 mm to about 1.25 mm, and a width, for example, of from bout 1.0 mm to about 3.0 mm, more preferably from about 1.75 mm to about 3.00 mm.
- an aperture 840 is provided so that a trailing string (not shown) can be included for ease of removal with the grasping device shown in FIG. 14 .
- Protruding elements 940 are made, for example, by cutting into the plastic rod with, for example, a sharp edged instrument, for example, a knife, so as penetrate a depth into the plastic rod.
- the depth of penetration P is adjusted to provide acceptable frictional properties, while minimizing the impact on the mechanical properties of the plastic rod.
- the maximum depth of penetration into the plastic rod, measured inwardly from the outer surface of the plastic rod is, for example, from about 1 to about 50% of the average thickness of the plastic rod.
- the mechanical properties of the plastic rod may be reduced and if the depth of penetration is too low, the resulting protruding elements may be too small to provide the appropriate frictional properties when expanded in a body cavity or lumen, for example, the prostatic urethra.
- Other cutting means are possible, for example, water knife and laser cutting means, to reduce the impact of the cutting on the mechanical properties of the plastic rod.
- the shape of the plastic rod from which the stent is made may be of other forms than that shown above. For example, it may be in the form of, for example, a coiled elongated flattened tube and the flattened tube may include a central opening that includes a medicament that can be released by the inserted stent.
- stent 900 is manufactured from plastic rod made by a variety of methods known in the art (e.g., extrusion and coextrusion).
- the plastic rod may have a diameter, for example, of about 0.25 mm to about 2.5 mm or more.
- the protruding elements are put onto the plastic rod before wrapping the mandrel shown and discussed above. After wrapping the mandrel, the plastic rod and wrapping fixture 200 are heated to the softening temperature of the polymer, making the plastic rod malleable.
- the protrusions are annealed in the “up” position, that is, with the protruding elements extending outwardly by “prying up” the protruding elements that results from cutting.
- Prying up the protruding elements may be achieved by, for example, running a surface across the protruding elements in a direction opposite the cut direction. Annealing is continued to fix the shape. After cooling, the stent is removed from mandrel. Before packaging, the flared end of the coil is tapered down along with protruding elements and collapsed so that the diameter along the entire length of the stent is approximately d 1 . Collapsing the flared end and protruding elements allows for ease of insertion, for example, into a restricted prostatic urethra. In some implementations, the flared end and the main body are collapsed to have a diameter less than d 1 .
- stent 1000 with protruding elements 1060 is made by, for example, wrapping a thicker plastic rod with a thinner plastic rod, for example, a monofilament, that includes a plurality of constrictions, for example, knots along its length.
- the elevation E above an outer surface of the thicker plastic rod is adjusted to provide acceptable frictional properties.
- the maximum elevation above an outer surface of the thicker plastic rod is, for example, from about 1 to about 50% of the average thickness of thicker plastic rod.
- the shape of the rods from which the stent is made may be of other forms than that shown above.
- it may be in the form of, for example, a coiled elongated flattened tube and the flattened tube may include a central opening that includes a medicament that can be released by the inserted stent.
- stent 1000 is manufactured from plastic rod made by a variety of methods known in the art (e.g., extrusion and coextrusion).
- the thicker plastic rod may have a diameter, for example, of about 0.25 mm to about 2.5 mm or more.
- the thinner plastic rod from which the protruding elements are fashioned may have a diameter of, for example, from about 0.2 mm to about 20 mm.
- the constrictions, for example, knots are placed on the thinner plastic rod and the thinner plastic rod is wrapped around the outer surface of the thicker plastic rod. The ends of the thinner plastic rod are heat staked to hold the thinner plastic rod onto the outer surface of the thicker plastic rod.
- the assembly of the thinner and thicker plastic rod is wrapped around the mandrel shown and discussed above.
- the plastic rods and wrapping fixture 200 are heated to the softening temperature of the polymer of the thicker plastic rod, making the plastic rod malleable Annealing is continued to fix the shape.
- the stent is removed from mandrel.
- the flared end of the coil is tapered down so that the diameter along the entire length of the stent is approximately d 1 . Collapsing the flared end and protruding elements allows for ease of insertion, for example, into a restricted prostatic urethra.
- the thinner plastic rod may contain a medicament that is released upon expansion, for example, in the prostatic urethra.
- the thinner plastic rod is made of a degradable material and the degradable material is filled with a medicament.
- the entire stent for example, the stents of FIGS. 4 , 5 , 20 , 23 and 26 , may have an expanded position.
- an implantable stent includes a tubular member 1102 that is formed from a polymeric material, e.g., PCO.
- Tubular member 1102 includes a wall with thickness W 1 and has a first transverse dimension OD 1 , and a first longitudinal length L 1 that is measured at first transverse dimension.
- Tubular member 1102 is sized for delivery into a lumen.
- an elevated temperature e.g., 40, 50, or 60° C.
- tubular member 1102 can be expanded ( FIG.
- Second transverse dimension OD 2 that is, e.g., about fifty percent larger than the first transverse dimension OD 1 within the lumen.
- First and second transverse dimensions are measured from an outer surface 1106 , 1108 of the tubular structure in its unexpanded and expanded state, respectively.
- the tubular member In its expanded state, the tubular member has a wall with thickness W 2 and a second longitudinal length L 2 , measured when at the second transverse dimension.
- the second longitudinal length L 2 decreases by less than about fifty percent, measured relative to the first longitudinal length L 1 . This reduced forshortening can improve, for example, placement accuracy within the lumen.
- the wall thickness of the tubular member decreases by greater than about twenty percent, e.g., greater than about fifty percent, greater than about fifty-five percent, greater than about sixty percent, greater than about sixty-five percent, greater than about seventy-five percent, or more, e.g., greater than about ninety percent, after expansion from the first transverse dimension to the second transverse dimension.
- a relatively large decrease in the wall thickness in going from the unexpanded state to the expanded state at least partially explains the observed reduced foreshortening.
- the second longitudinal length decreases by less than about thirty percent, e.g., less than twenty-five percent, less than twenty percent, less than fifteen percent, or less than ten percent, measured relative to the first longitudinal length.
- the polymeric material has relatively low softening temperature so that high temperatures do not need to used within the body.
- the polymer can have a softening temperature from about 40° C. to about 60° C., e.g., 45, 50, 55, or 58° C.
- the polymeric material can be non-cross-linked, cross-linked, shape memory, or non-shape memory.
- suitable polymeric materials include those discussed above, e.g., nylons, polyurethanes, or PVAc/PVDF blends, and those discussed below.
- Specific polymeric materials include polycyclooctene (PCO), styrenic elastomers, styrenic block copolymers, styrene-butadiene rubber, polyolefins, trans-isoprene, plasticized PVC, e.g., PVC plasticized with a monomeric plasticizer, e.g., a phthalate, or a polymeric plasticizer, or blends of these polymers.
- PCO polycyclooctene
- styrenic elastomers elastomers
- styrenic block copolymers styrene-butadiene rubber
- polyolefins s
- the polymeric material has an elastic modulus of greater than about 50,000 psi, e.g., greater than about 75,000, greater than about 100,000, greater than about 200,000, greater than about 250,000, or more, e.g., greater than about 500,000 psi. Without wishing to be bound by any particular theory, it is believed that proper selection of the polymeric material at least partially explains the observed reduced foreshortening.
- a stent can have a shape in memory, e.g., a curved shape.
- the stent can have an unexpanded shape that is substantially straight, and an expanded shape that is curved ( FIG. 33 ).
- a curved tubular member can enable better retention in a deployed region of the lumen such that the stent has a reduced likelihood for movement within a lumen.
- Other memorized shapes are possible.
- the stent may have a flared end, or two flared ends after expansion, as shown in FIG. 34 . Flared ends can also enable better retention in a deployed region of a lumen.
- a tubular member can include apertures 1114 defined in a wall, when this is desired.
- apertures are advantageous because they can allow tissue to grow into the apertures, thereby enabling better retention in the lumen.
- an unexpanded tubular stent is cylindrical in shape, has a smooth outer surface, and is made of PCO filled with about forty percent by weight of a boron nitride for radio-opacity, and for enhanced thermal conductivity.
- the stent has an unexpanded wall thickness of about 3 mm, an outer diameter of approximately 10 french, and an unexpanded length of approximately 25 mm. After expansion on a heated balloon at 50° C., followed by cooling to set the shape of the stent, an expanded wall thickness is approximately 1 mm, an outer diameter is approximately 20 french, and an expanded length is approximately 20 mm.
- PVDF poly(vinylidene fluoride)
- PVA polylactide
- PEG poly(ethylene glycol)
- PEG polyethylene
- PVC polyethylene-co-vinyl acetate
- PVDC poly(vinylidene chloride)
- PVDC copolymers of poly vinylidene chloride
- PVDC polyvinylidene chlor
- PVAc poly(vinyl acetate)
- PLA semicrystalline polylactide
- PVDF poly(vinylidene fluoride)
- the polymers show complete miscibility at all blending ratios with a single glass transition temperature, while crystallization (exclusive of PVAc) is partially maintained.
- the T g 's of the blends are employed as the critical temperature for triggering the shape recovery while the crystalline phases serve as physical crosslinking sites for elastic deformation above T g , but below T m .
- PVAc poly(vinyl chloride) with poly(butyl acrylate) or poly(butyl methacrylate)
- PVC/PBA poly(butyl methacrylate)
- PVAc simultaneously lowers the crystallinity of PVDF while increasing T g .
- PVC may serve the same role as PVDF, but it already has a low degree of crystallinity, but a relatively high T g ( ⁇ 80° C.).
- the second component (PBA) serves only the role of decreasing T g .
- small molecule plasticizers most notably dioctylphthalate (DOP), but is preferred to use a biocompatible polymeric plasticizer for intended implantable applications.
- DOP dioctylphthalate
- the range of PBA compositions is 10-40%, with 20% being the most advantageous, yielding a T g ⁇ 40° C.
- the crystallization behavior of semicrystalline PLA was investigated via DSC.
- the PLA samples were first heat pressed at 180° C. for 10 minutes and then quenched to room temperature with water cooling.
- the storage modulus above T g shifts to about 200 MPa until melting, the increase being due to an increase of the degree of crystallinity on annealing to tune the rubbery modulus at equilibrium state.
- PLA was blended in different proportions to PVAc and annealed as above. Storage moduli for such blends were measured and the results are plotted in FIG. 39 . It can be seen that, below T g , all of the samples show similar large moduli while above T g the moduli decrease to a plateau whose magnitude depends on crystallinity and thus PLA content. This trend is in accordance with that of DSC and XRD, and can be explained by the fact that the increase of storage moduli came from the physical crosslinking formed by crystals and the filler effect of the high modulus crystalline phase.
- Bioabsorbable polymers include, for example, polyurethanes and polyurethane copolymers such as those described above with the general formula (directly below), where X/Y is, for example, 1 to 20, n is, for example, 2 to 1000, and the total degree of polymerization m is, for example, 2 to 100
- the bioabsorbability of the polymers is enhanced by copolymerization of polyurethane and POSS with suitable monomers.
- suitable monomers include caprolactone, ethyleneglycol, ethylene oxide, lactic acid, and glycolic acid.
- the copolymers from these monomers can hydrolyze and cleave the polymer linkage.
- the stent is sized (e.g., an expanded inner diameter of about 2 mm to about 20 mm) and configured for use in the vascular system, particularly the coronary arteries, and implanted after or simultaneously with an angioplasty procedure to maintain an open lumen and reduce restenosis.
- Vascular stents are described in U.S. Provisional Application No. 60/418,023, which is hereby incorporated in full by reference.
- a stent for coronary use can have an initial diameter of about 2 mm, an expanded diameter of about 4 mm, and a wall thickness of about 0.005 mm to 0.1 mm.
- Other exemplary applications include neuro, carotid, peripheral, and vasculature lumens.
- the vascular stent can be bioabsorbable or non-bioabsorbable.
- a stent e.g., a bioabsorbable or a non-bioabsorbable stent, is constructed for use in nonvascular lumens, such as the esophagus, ureteral, biliary, or prostate.
- the stent is conductive to allow an electrical current to pass through the stent, for example, to deliver electricity to an area of the body or to trigger, for example, a physical change in the stent, for example, a change in the diameter of the stent.
- the stent for example, of FIGS. 4 , 23 and 26 , is made porous by, for example, adding a chemical foaming agent to the polymer from which the stent is made during the production the plastic strand.
- the stent is porous and includes a medicament.
- the initial porosity of the stent can be reduced, for example, by the application of heat and pressure before deployment in the body. Upon deployment of the stent in the body, the porosity is increased by a triggering event, for example, the application of heat to the stent at the desired site of treatment.
Abstract
Description
The bioabsorbability of the polymers is enhanced by copolymerization of polyurethane and POSS with suitable monomers. Examples of suitable monomers include caprolactone, ethyleneglycol, ethylene oxide, lactic acid, and glycolic acid. The copolymers from these monomers can hydrolyze and cleave the polymer linkage.
Claims (20)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/940,388 US8784465B2 (en) | 2002-10-11 | 2010-11-05 | Implantable medical devices |
| US13/493,532 US9115245B2 (en) | 2002-10-11 | 2012-06-11 | Implantable medical devices |
Applications Claiming Priority (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US41802302P | 2002-10-11 | 2002-10-11 | |
| US41950602P | 2002-10-18 | 2002-10-18 | |
| US46640103P | 2003-04-29 | 2003-04-29 | |
| US48832303P | 2003-07-18 | 2003-07-18 | |
| US48859003P | 2003-07-18 | 2003-07-18 | |
| US10/683,314 US20050010275A1 (en) | 2002-10-11 | 2003-10-10 | Implantable medical devices |
| US10/958,435 US7794494B2 (en) | 2002-10-11 | 2004-10-05 | Implantable medical devices |
| US12/880,483 US20140135889A9 (en) | 2002-10-11 | 2010-09-13 | Implantable medical devices |
| US12/940,388 US8784465B2 (en) | 2002-10-11 | 2010-11-05 | Implantable medical devices |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/880,483 Division US20140135889A9 (en) | 2002-10-11 | 2010-09-13 | Implantable medical devices |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/493,532 Division US9115245B2 (en) | 2002-10-11 | 2012-06-11 | Implantable medical devices |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20110054591A1 US20110054591A1 (en) | 2011-03-03 |
| US8784465B2 true US8784465B2 (en) | 2014-07-22 |
Family
ID=35897979
Family Applications (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/958,435 Expired - Fee Related US7794494B2 (en) | 2002-10-11 | 2004-10-05 | Implantable medical devices |
| US12/880,483 Abandoned US20140135889A9 (en) | 2002-10-11 | 2010-09-13 | Implantable medical devices |
| US12/940,388 Expired - Fee Related US8784465B2 (en) | 2002-10-11 | 2010-11-05 | Implantable medical devices |
| US13/493,532 Expired - Fee Related US9115245B2 (en) | 2002-10-11 | 2012-06-11 | Implantable medical devices |
Family Applications Before (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/958,435 Expired - Fee Related US7794494B2 (en) | 2002-10-11 | 2004-10-05 | Implantable medical devices |
| US12/880,483 Abandoned US20140135889A9 (en) | 2002-10-11 | 2010-09-13 | Implantable medical devices |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/493,532 Expired - Fee Related US9115245B2 (en) | 2002-10-11 | 2012-06-11 | Implantable medical devices |
Country Status (5)
| Country | Link |
|---|---|
| US (4) | US7794494B2 (en) |
| EP (1) | EP1824412A2 (en) |
| AU (1) | AU2005294569A1 (en) |
| CA (1) | CA2583191A1 (en) |
| WO (1) | WO2006041767A2 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20150114143A1 (en) * | 2013-10-24 | 2015-04-30 | Spirit Aerosystems, Inc. | Remoldable contour sensor holder |
| US11096774B2 (en) | 2016-12-09 | 2021-08-24 | Zenflow, Inc. | Systems, devices, and methods for the accurate deployment of an implant in the prostatic urethra |
| US11890213B2 (en) | 2019-11-19 | 2024-02-06 | Zenflow, Inc. | Systems, devices, and methods for the accurate deployment and imaging of an implant in the prostatic urethra |
Families Citing this family (209)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2178541C (en) | 1995-06-07 | 2009-11-24 | Neal E. Fearnot | Implantable medical device |
| WO1999042156A1 (en) * | 1998-02-24 | 1999-08-26 | Boston Scientific Limited | High flow rate dialysis catheters and related methods |
| US7713297B2 (en) | 1998-04-11 | 2010-05-11 | Boston Scientific Scimed, Inc. | Drug-releasing stent with ceramic-containing layer |
| US20060177416A1 (en) | 2003-10-14 | 2006-08-10 | Medivas, Llc | Polymer particle delivery compositions and methods of use |
| AU2002345328A1 (en) | 2001-06-27 | 2003-03-03 | Remon Medical Technologies Ltd. | Method and device for electrochemical formation of therapeutic species in vivo |
| US20040030062A1 (en) * | 2002-05-02 | 2004-02-12 | Mather Patrick T. | Castable shape memory polymers |
| US7524914B2 (en) * | 2002-10-11 | 2009-04-28 | The University Of Connecticut | Shape memory polymers based on semicrystalline thermoplastic polyurethanes bearing nanostructured hard segments |
| US7208550B2 (en) * | 2002-10-11 | 2007-04-24 | The University Of Connecticut | Blends of amorphous and semicrystalline polymers having shape memory properties |
| CA2501551C (en) * | 2002-10-11 | 2012-12-11 | University Of Connecticut | Crosslinked polycyclooctene |
| US7976936B2 (en) * | 2002-10-11 | 2011-07-12 | University Of Connecticut | Endoprostheses |
| ATE499396T1 (en) * | 2002-10-11 | 2011-03-15 | Univ Connecticut | SHAPE MEMORY POLYMERS BASED ON SEMICRYSTALLINE THERMOPLASTIC POLYURETHANES THAT HAVE NANOSTRUCTURED HARD SEGMENTS |
| US7960935B2 (en) | 2003-07-08 | 2011-06-14 | The Board Of Regents Of The University Of Nebraska | Robotic devices with agent delivery components and related methods |
| US7004965B2 (en) * | 2003-12-17 | 2006-02-28 | Yosef Gross | Implant and delivery tool therefor |
| US9801527B2 (en) | 2004-04-19 | 2017-10-31 | Gearbox, Llc | Lumen-traveling biological interface device |
| US9011329B2 (en) | 2004-04-19 | 2015-04-21 | Searete Llc | Lumenally-active device |
| US7998060B2 (en) | 2004-04-19 | 2011-08-16 | The Invention Science Fund I, Llc | Lumen-traveling delivery device |
| US8092549B2 (en) | 2004-09-24 | 2012-01-10 | The Invention Science Fund I, Llc | Ciliated stent-like-system |
| US7850676B2 (en) | 2004-04-19 | 2010-12-14 | The Invention Science Fund I, Llc | System with a reservoir for perfusion management |
| US8019413B2 (en) | 2007-03-19 | 2011-09-13 | The Invention Science Fund I, Llc | Lumen-traveling biological interface device and method of use |
| US7857767B2 (en) | 2004-04-19 | 2010-12-28 | Invention Science Fund I, Llc | Lumen-traveling device |
| US8361013B2 (en) | 2004-04-19 | 2013-01-29 | The Invention Science Fund I, Llc | Telescoping perfusion management system |
| US8337482B2 (en) | 2004-04-19 | 2012-12-25 | The Invention Science Fund I, Llc | System for perfusion management |
| US8353896B2 (en) | 2004-04-19 | 2013-01-15 | The Invention Science Fund I, Llc | Controllable release nasal system |
| US8043361B2 (en) * | 2004-12-10 | 2011-10-25 | Boston Scientific Scimed, Inc. | Implantable medical devices, and methods of delivering the same |
| US7785291B2 (en) * | 2005-03-01 | 2010-08-31 | Tulip Medical Ltd. | Bioerodible self-deployable intragastric implants |
| US7699863B2 (en) * | 2005-03-01 | 2010-04-20 | Tulip Medical Ltd. | Bioerodible self-deployable intragastric implants |
| US7763037B2 (en) | 2005-03-18 | 2010-07-27 | Castlewood Surgical, Inc. | System and method for attaching a vein, an artery, or a tube in a vascular environment |
| US7758594B2 (en) * | 2005-05-20 | 2010-07-20 | Neotract, Inc. | Devices, systems and methods for treating benign prostatic hyperplasia and other conditions |
| US8628542B2 (en) | 2005-05-20 | 2014-01-14 | Neotract, Inc. | Median lobe destruction apparatus and method |
| US10195014B2 (en) | 2005-05-20 | 2019-02-05 | Neotract, Inc. | Devices, systems and methods for treating benign prostatic hyperplasia and other conditions |
| US20060271170A1 (en) * | 2005-05-31 | 2006-11-30 | Gale David C | Stent with flexible sections in high strain regions |
| JP5192384B2 (en) | 2005-09-22 | 2013-05-08 | メディバス エルエルシー | Bis- (α-amino) -diol-diester-containing poly (ester amide) and poly (ester urethane) compositions and methods of use |
| JP5178520B2 (en) | 2005-09-22 | 2013-04-10 | メディバス エルエルシー | Solid polymer delivery compositions and methods of use thereof |
| DE102005056532A1 (en) * | 2005-11-28 | 2007-05-31 | Mnemoscience Gmbh | Method for removal of tubular tissue supports e.g. stents from human or animal hollow organs, involves heating of tissue support to temperature below transition temperature, till softening |
| US20070135751A1 (en) * | 2005-12-09 | 2007-06-14 | Dicarlo Paul D | Medical devices |
| US8840660B2 (en) | 2006-01-05 | 2014-09-23 | Boston Scientific Scimed, Inc. | Bioerodible endoprostheses and methods of making the same |
| US20070158880A1 (en) * | 2006-01-06 | 2007-07-12 | Vipul Bhupendra Dave | Methods of making bioabsorbable drug delivery devices comprised of solvent cast tubes |
| US20070160672A1 (en) * | 2006-01-06 | 2007-07-12 | Vipul Bhupendra Dave | Methods of making bioabsorbable drug delivery devices comprised of solvent cast films |
| US20070162110A1 (en) * | 2006-01-06 | 2007-07-12 | Vipul Bhupendra Dave | Bioabsorbable drug delivery devices |
| US7951185B1 (en) * | 2006-01-06 | 2011-05-31 | Advanced Cardiovascular Systems, Inc. | Delivery of a stent at an elevated temperature |
| US8089029B2 (en) | 2006-02-01 | 2012-01-03 | Boston Scientific Scimed, Inc. | Bioabsorbable metal medical device and method of manufacture |
| US20070191931A1 (en) * | 2006-02-16 | 2007-08-16 | Jan Weber | Bioerodible endoprostheses and methods of making the same |
| US9526814B2 (en) * | 2006-02-16 | 2016-12-27 | Boston Scientific Scimed, Inc. | Medical balloons and methods of making the same |
| US20070224235A1 (en) | 2006-03-24 | 2007-09-27 | Barron Tenney | Medical devices having nanoporous coatings for controlled therapeutic agent delivery |
| US8187620B2 (en) | 2006-03-27 | 2012-05-29 | Boston Scientific Scimed, Inc. | Medical devices comprising a porous metal oxide or metal material and a polymer coating for delivering therapeutic agents |
| US8048150B2 (en) | 2006-04-12 | 2011-11-01 | Boston Scientific Scimed, Inc. | Endoprosthesis having a fiber meshwork disposed thereon |
| US20120035540A1 (en) | 2006-04-12 | 2012-02-09 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Event-based control of a lumen traveling device |
| US20080058785A1 (en) | 2006-04-12 | 2008-03-06 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Autofluorescent imaging and target ablation |
| EP2023869B1 (en) * | 2006-05-12 | 2019-09-18 | Cardinal Health Switzerland 515 GmbH | Balloon expandable bioabsorbable drug eluting flexible stent |
| EP2034927B1 (en) * | 2006-06-20 | 2012-01-25 | Boston Scientific Limited | Medical devices including composites |
| US8679096B2 (en) | 2007-06-21 | 2014-03-25 | Board Of Regents Of The University Of Nebraska | Multifunctional operational component for robotic devices |
| US9579088B2 (en) | 2007-02-20 | 2017-02-28 | Board Of Regents Of The University Of Nebraska | Methods, systems, and devices for surgical visualization and device manipulation |
| US8974440B2 (en) | 2007-08-15 | 2015-03-10 | Board Of Regents Of The University Of Nebraska | Modular and cooperative medical devices and related systems and methods |
| CA2991346C (en) | 2006-06-22 | 2020-03-10 | Board Of Regents Of The University Of Nebraska | Magnetically coupleable robotic devices and related methods |
| US8815275B2 (en) | 2006-06-28 | 2014-08-26 | Boston Scientific Scimed, Inc. | Coatings for medical devices comprising a therapeutic agent and a metallic material |
| WO2008002778A2 (en) | 2006-06-29 | 2008-01-03 | Boston Scientific Limited | Medical devices with selective coating |
| US7862830B2 (en) * | 2006-07-13 | 2011-01-04 | Abbott Cardiovascular Systems Inc. | Stereocomplex-forming composition and implantable medical device comprising same |
| EP2054537A2 (en) | 2006-08-02 | 2009-05-06 | Boston Scientific Scimed, Inc. | Endoprosthesis with three-dimensional disintegration control |
| DE102006038232A1 (en) * | 2006-08-07 | 2008-02-14 | Biotronik Vi Patent Ag | Endoprosthesis and method for producing such |
| US20080051759A1 (en) * | 2006-08-24 | 2008-02-28 | Boston Scientific Scimed, Inc. | Polycarbonate polyurethane venous access devices |
| ATE508708T1 (en) | 2006-09-14 | 2011-05-15 | Boston Scient Ltd | MEDICAL DEVICES WITH A DRUG-RELEASING COATING |
| WO2008034030A2 (en) * | 2006-09-15 | 2008-03-20 | Boston Scientific Limited | Magnetized bioerodible endoprosthesis |
| CA2663250A1 (en) | 2006-09-15 | 2008-03-20 | Boston Scientific Limited | Bioerodible endoprostheses and methods of making the same |
| CA2663220A1 (en) | 2006-09-15 | 2008-03-20 | Boston Scientific Limited | Medical devices and methods of making the same |
| US8057534B2 (en) | 2006-09-15 | 2011-11-15 | Boston Scientific Scimed, Inc. | Bioerodible endoprostheses and methods of making the same |
| EP2210625B8 (en) | 2006-09-15 | 2012-02-29 | Boston Scientific Scimed, Inc. | Bioerodible endoprosthesis with biostable inorganic layers |
| EP2076296A2 (en) | 2006-09-15 | 2009-07-08 | Boston Scientific Scimed, Inc. | Endoprosthesis with adjustable surface features |
| EP2068962B1 (en) | 2006-09-18 | 2013-01-30 | Boston Scientific Limited | Endoprostheses |
| US20080069858A1 (en) * | 2006-09-20 | 2008-03-20 | Boston Scientific Scimed, Inc. | Medical devices having biodegradable polymeric regions with overlying hard, thin layers |
| US20080081064A1 (en) * | 2006-09-28 | 2008-04-03 | Surmodics, Inc. | Implantable Medical Device with Apertures for Delivery of Bioactive Agents |
| US7981150B2 (en) | 2006-11-09 | 2011-07-19 | Boston Scientific Scimed, Inc. | Endoprosthesis with coatings |
| US20080132988A1 (en) * | 2006-12-01 | 2008-06-05 | Scimed Life Systems, Inc. | Balloon geometry for delivery and deployment of shape memory polymer stent with flares |
| JP5355418B2 (en) | 2006-12-28 | 2013-11-27 | ボストン サイエンティフィック リミテッド | Bioerodible endoprosthesis and method for manufacturing the bioerodible endoprosthesis |
| US8814930B2 (en) | 2007-01-19 | 2014-08-26 | Elixir Medical Corporation | Biodegradable endoprosthesis and methods for their fabrication |
| US20130150943A1 (en) * | 2007-01-19 | 2013-06-13 | Elixir Medical Corporation | Biodegradable endoprostheses and methods for their fabrication |
| US8431149B2 (en) | 2007-03-01 | 2013-04-30 | Boston Scientific Scimed, Inc. | Coated medical devices for abluminal drug delivery |
| US8070797B2 (en) | 2007-03-01 | 2011-12-06 | Boston Scientific Scimed, Inc. | Medical device with a porous surface for delivery of a therapeutic agent |
| US7649022B2 (en) | 2007-03-30 | 2010-01-19 | Medivas, Llc | Bioabsorbable elastomeric polymer networks, cross-linkers and methods of use |
| US8067054B2 (en) | 2007-04-05 | 2011-11-29 | Boston Scientific Scimed, Inc. | Stents with ceramic drug reservoir layer and methods of making and using the same |
| EP2157976A4 (en) * | 2007-04-19 | 2012-03-07 | Univ Massachusetts Medical | Thermal responsive polymer siloxanes, compositions, and methods and applications related thereto |
| WO2008142677A2 (en) * | 2007-05-18 | 2008-11-27 | Prostaplant, Ltd. | Prostate implant and methods for insertion and extraction thereof |
| US20100130815A1 (en) * | 2007-05-18 | 2010-05-27 | Prostaplant Ltd. | Intraurethral and extraurethral apparatus |
| US7976915B2 (en) | 2007-05-23 | 2011-07-12 | Boston Scientific Scimed, Inc. | Endoprosthesis with select ceramic morphology |
| US8002823B2 (en) * | 2007-07-11 | 2011-08-23 | Boston Scientific Scimed, Inc. | Endoprosthesis coating |
| US7942926B2 (en) | 2007-07-11 | 2011-05-17 | Boston Scientific Scimed, Inc. | Endoprosthesis coating |
| JP5591696B2 (en) | 2007-07-12 | 2014-09-17 | ボード オブ リージェンツ オブ ザ ユニバーシティ オブ ネブラスカ | Biopsy elements, arm devices, and medical devices |
| EP2178541A4 (en) * | 2007-07-17 | 2012-11-14 | Medivas Llc | Bioabsorbable elastomeric arterial support device and methods of use |
| WO2009012353A2 (en) | 2007-07-19 | 2009-01-22 | Boston Scientific Limited | Endoprosthesis having a non-fouling surface |
| US8815273B2 (en) | 2007-07-27 | 2014-08-26 | Boston Scientific Scimed, Inc. | Drug eluting medical devices having porous layers |
| US7931683B2 (en) | 2007-07-27 | 2011-04-26 | Boston Scientific Scimed, Inc. | Articles having ceramic coated surfaces |
| EP2182892B1 (en) * | 2007-07-30 | 2020-08-12 | Audubon Technologies, LLC | Device for maintaining patent paranasal sinus ostia |
| US8221822B2 (en) | 2007-07-31 | 2012-07-17 | Boston Scientific Scimed, Inc. | Medical device coating by laser cladding |
| WO2009020520A1 (en) | 2007-08-03 | 2009-02-12 | Boston Scientific Scimed, Inc. | Coating for medical device having increased surface area |
| US20100159011A1 (en) | 2007-08-03 | 2010-06-24 | University Of Massachusetts Medical School | Compositions For Biomedical Applications |
| US20090043276A1 (en) * | 2007-08-09 | 2009-02-12 | Boston Scientific Scimed, Inc. | Drug delivery device, compositions and methods relating thereto |
| US20090076536A1 (en) | 2007-08-15 | 2009-03-19 | Board Of Regents Of The University Of Nebraska | Medical inflation, attachment, and delivery devices and related methods |
| US8052745B2 (en) | 2007-09-13 | 2011-11-08 | Boston Scientific Scimed, Inc. | Endoprosthesis |
| US8337451B2 (en) * | 2007-10-19 | 2012-12-25 | Angio Dynamics, Inc. | Recirculation minimizing catheter |
| US7938855B2 (en) | 2007-11-02 | 2011-05-10 | Boston Scientific Scimed, Inc. | Deformable underlayer for stent |
| US20090118812A1 (en) * | 2007-11-02 | 2009-05-07 | Boston Scientific Scimed, Inc. | Endoprosthesis coating |
| US8216632B2 (en) * | 2007-11-02 | 2012-07-10 | Boston Scientific Scimed, Inc. | Endoprosthesis coating |
| US8029554B2 (en) | 2007-11-02 | 2011-10-04 | Boston Scientific Scimed, Inc. | Stent with embedded material |
| US20090143760A1 (en) * | 2007-11-30 | 2009-06-04 | Jacques Van Dam | Methods, Devices, Kits and Systems for Defunctionalizing the Gallbladder |
| US20110027379A1 (en) * | 2007-12-06 | 2011-02-03 | Cornell University | Oligo-Ethylene Glycol-Based Polymer Compositions and Methods of Use |
| US7972373B2 (en) * | 2007-12-19 | 2011-07-05 | Advanced Technologies And Regenerative Medicine, Llc | Balloon expandable bioabsorbable stent with a single stress concentration region interconnecting adjacent struts |
| WO2009131911A2 (en) | 2008-04-22 | 2009-10-29 | Boston Scientific Scimed, Inc. | Medical devices having a coating of inorganic material |
| WO2009132176A2 (en) | 2008-04-24 | 2009-10-29 | Boston Scientific Scimed, Inc. | Medical devices having inorganic particle layers |
| US7998192B2 (en) | 2008-05-09 | 2011-08-16 | Boston Scientific Scimed, Inc. | Endoprostheses |
| JP2011521716A (en) * | 2008-05-29 | 2011-07-28 | ボストン サイエンティフィック サイムド,インコーポレイテッド | Prosthetic coating |
| US8236046B2 (en) | 2008-06-10 | 2012-08-07 | Boston Scientific Scimed, Inc. | Bioerodible endoprosthesis |
| EP2303350A2 (en) * | 2008-06-18 | 2011-04-06 | Boston Scientific Scimed, Inc. | Endoprosthesis coating |
| US7985252B2 (en) | 2008-07-30 | 2011-07-26 | Boston Scientific Scimed, Inc. | Bioerodible endoprosthesis |
| US8642063B2 (en) | 2008-08-22 | 2014-02-04 | Cook Medical Technologies Llc | Implantable medical device coatings with biodegradable elastomer and releasable taxane agent |
| US20100057188A1 (en) * | 2008-08-28 | 2010-03-04 | Boston Scientific Scimed, Inc. | Endoprostheses with porous regions and non-polymeric coating |
| US8114153B2 (en) * | 2008-09-05 | 2012-02-14 | Boston Scientific Scimed, Inc. | Endoprostheses |
| US8382824B2 (en) | 2008-10-03 | 2013-02-26 | Boston Scientific Scimed, Inc. | Medical implant having NANO-crystal grains with barrier layers of metal nitrides or fluorides |
| WO2010048052A1 (en) | 2008-10-22 | 2010-04-29 | Boston Scientific Scimed, Inc. | Shape memory tubular stent with grooves |
| US8231980B2 (en) | 2008-12-03 | 2012-07-31 | Boston Scientific Scimed, Inc. | Medical implants including iridium oxide |
| WO2010101901A2 (en) | 2009-03-02 | 2010-09-10 | Boston Scientific Scimed, Inc. | Self-buffering medical implants |
| US8071156B2 (en) | 2009-03-04 | 2011-12-06 | Boston Scientific Scimed, Inc. | Endoprostheses |
| US20100244304A1 (en) * | 2009-03-31 | 2010-09-30 | Yunbing Wang | Stents fabricated from a sheet with increased strength, modulus and fracture toughness |
| US20100256546A1 (en) * | 2009-04-03 | 2010-10-07 | Davis Scott A | Polycarbonate Polyurethane Venous Access Devices Having Enhanced Strength |
| US8287937B2 (en) | 2009-04-24 | 2012-10-16 | Boston Scientific Scimed, Inc. | Endoprosthese |
| US9265633B2 (en) | 2009-05-20 | 2016-02-23 | 480 Biomedical, Inc. | Drug-eluting medical implants |
| US8992601B2 (en) * | 2009-05-20 | 2015-03-31 | 480 Biomedical, Inc. | Medical implants |
| US9901347B2 (en) * | 2009-05-29 | 2018-02-27 | Terus Medical, Inc. | Biliary shunts, delivery systems, and methods of using the same |
| US20110022162A1 (en) * | 2009-07-23 | 2011-01-27 | Boston Scientific Scimed, Inc. | Endoprostheses |
| US20110071500A1 (en) * | 2009-09-21 | 2011-03-24 | Navilyst Medical, Inc. | Branched catheter tip |
| US8740970B2 (en) | 2009-12-02 | 2014-06-03 | Castlewood Surgical, Inc. | System and method for attaching a vessel in a vascular environment |
| EP2512754A4 (en) | 2009-12-17 | 2016-11-30 | Univ Nebraska | Modular and cooperative medical devices and related systems and methods |
| US8328760B2 (en) * | 2010-01-11 | 2012-12-11 | Angiodynamics, Inc. | Occlusion resistant catheter |
| US8568471B2 (en) * | 2010-01-30 | 2013-10-29 | Abbott Cardiovascular Systems Inc. | Crush recoverable polymer scaffolds |
| WO2011119573A1 (en) | 2010-03-23 | 2011-09-29 | Boston Scientific Scimed, Inc. | Surface treated bioerodible metal endoprostheses |
| US20110238153A1 (en) * | 2010-03-26 | 2011-09-29 | Boston Scientific Scimed, Inc. | Endoprostheses |
| WO2011119430A1 (en) | 2010-03-26 | 2011-09-29 | Boston Scientific Scimed, Inc. | Endoprosthesis |
| EP2555811B1 (en) | 2010-04-06 | 2017-09-27 | Boston Scientific Scimed, Inc. | Endoprosthesis |
| US8920490B2 (en) | 2010-05-13 | 2014-12-30 | Boston Scientific Scimed, Inc. | Endoprostheses |
| EP2600758A1 (en) | 2010-08-06 | 2013-06-12 | Board of Regents of the University of Nebraska | Methods and systems for handling or delivering materials for natural orifice surgery |
| US20120059451A1 (en) * | 2010-09-08 | 2012-03-08 | Qiang Zhang | Method of Manufacturing a Polymeric Stent Having Reduced Recoil |
| WO2012032128A1 (en) * | 2010-09-08 | 2012-03-15 | Windmöller & Hölscher Kg | Calibrating device for a blown film tube |
| ES2571735T3 (en) | 2010-10-20 | 2016-05-26 | Dsm Ip Assets Bv | Biodegradable compositions that support hanging hydrophilic group and related devices |
| US9999746B2 (en) | 2011-03-22 | 2018-06-19 | Angiodynamics, Inc. | High flow catheters |
| US9050435B2 (en) | 2011-03-22 | 2015-06-09 | Angiodynamics, Inc. | High flow catheters |
| EP3714821A1 (en) | 2011-06-10 | 2020-09-30 | Board of Regents of the University of Nebraska | Surgical end effector |
| US9873765B2 (en) | 2011-06-23 | 2018-01-23 | Dsm Ip Assets, B.V. | Biodegradable polyesteramide copolymers for drug delivery |
| CA2839526A1 (en) | 2011-06-23 | 2012-12-27 | Dsm Ip Assets B.V. | Micro- or nanoparticles comprising a biodegradable polyesteramide copolymer for use in the delivery of bioactive agents |
| EP2726015B1 (en) * | 2011-06-30 | 2019-06-19 | Elixir Medical Corporation | Biodegradable endoprostheses and methods for their fabrication |
| US20130142835A1 (en) * | 2011-07-06 | 2013-06-06 | Alexander M. Seifalian | Synthetic scaffolds and organ and tissue transplantation |
| US9089353B2 (en) | 2011-07-11 | 2015-07-28 | Board Of Regents Of The University Of Nebraska | Robotic surgical devices, systems, and related methods |
| GB201114059D0 (en) * | 2011-08-16 | 2011-09-28 | Depuy Ireland | Attachment mechanism |
| US10582973B2 (en) | 2012-08-08 | 2020-03-10 | Virtual Incision Corporation | Robotic surgical devices, systems, and related methods |
| US9204966B2 (en) * | 2011-11-21 | 2015-12-08 | Northwestern University | Penile prosthesis |
| US20140058205A1 (en) | 2012-01-10 | 2014-02-27 | Board Of Regents Of The University Of Nebraska | Methods, Systems, and Devices for Surgical Access and Insertion |
| US9707339B2 (en) | 2012-03-28 | 2017-07-18 | Angiodynamics, Inc. | High flow rate dual reservoir port system |
| US9713704B2 (en) | 2012-03-29 | 2017-07-25 | Bradley D. Chartrand | Port reservoir cleaning system and method |
| EP4357083A2 (en) | 2012-05-01 | 2024-04-24 | Board of Regents of the University of Nebraska | Single site robotic device and related systems and methods |
| EP3957276A1 (en) | 2012-05-31 | 2022-02-23 | Javelin Medical Ltd. | Device for embolic protection |
| CN104334123B (en) | 2012-06-07 | 2019-02-12 | 意比图密医疗有限公司 | Expansion device |
| EP3189948B1 (en) | 2012-06-22 | 2018-10-17 | Board of Regents of the University of Nebraska | Local control robotic surgical devices |
| US9770305B2 (en) | 2012-08-08 | 2017-09-26 | Board Of Regents Of The University Of Nebraska | Robotic surgical devices, systems, and related methods |
| US9422393B2 (en) * | 2012-11-13 | 2016-08-23 | Syracuse University | Water-triggered shape memory of PCL-PEG multiblock TPUs |
| EP2938388B1 (en) | 2012-12-31 | 2019-08-07 | Clearstream Technologies Limited | Balloon catheter with transient radiopaque marking |
| ES2895968T3 (en) | 2013-01-18 | 2022-02-23 | Javelin Medical Ltd | Monofilament implants and systems for supplying the same |
| CA2906672C (en) | 2013-03-14 | 2022-03-15 | Board Of Regents Of The University Of Nebraska | Methods, systems, and devices relating to force control surgical systems |
| US9743987B2 (en) | 2013-03-14 | 2017-08-29 | Board Of Regents Of The University Of Nebraska | Methods, systems, and devices relating to robotic surgical devices, end effectors, and controllers |
| EP2996545B1 (en) | 2013-03-15 | 2021-10-20 | Board of Regents of the University of Nebraska | Robotic surgical systems |
| US10966700B2 (en) | 2013-07-17 | 2021-04-06 | Virtual Incision Corporation | Robotic surgical devices, systems and related methods |
| US20150025608A1 (en) | 2013-07-22 | 2015-01-22 | Cardiac Pacemakers, Inc. | Lubricious, biocompatible hydrophilic thermoset coating using interpenetrating hydrogel networks |
| TWI478962B (en) * | 2013-10-09 | 2015-04-01 | Univ Nat Taiwan | Manufacturing method of enhancing ordered structure of block copolymers |
| CN105899198A (en) | 2013-12-05 | 2016-08-24 | 图利普医疗有限公司 | Retentive devices and systems for in-situ release of pharmaceutical active agents |
| US9592110B1 (en) | 2013-12-06 | 2017-03-14 | Javelin Medical, Ltd. | Systems and methods for implant delivery |
| US10166321B2 (en) | 2014-01-09 | 2019-01-01 | Angiodynamics, Inc. | High-flow port and infusion needle systems |
| AU2015229323B2 (en) * | 2014-03-14 | 2019-11-07 | The Board Of Trustees Of The Leland Stanford Junior University | Indwelling body lumen expander |
| US9913649B2 (en) | 2014-05-28 | 2018-03-13 | Boston Scientific Scimed, Inc. | Catheter with radiofrequency cutting tip and heated balloon |
| CN103992631B (en) * | 2014-05-29 | 2016-09-21 | 理大产学研基地(深圳)有限公司 | Polymeric material with bidirectional shape memory and preparation method thereof |
| US9730819B2 (en) | 2014-08-15 | 2017-08-15 | Elixir Medical Corporation | Biodegradable endoprostheses and methods of their fabrication |
| US9480588B2 (en) | 2014-08-15 | 2016-11-01 | Elixir Medical Corporation | Biodegradable endoprostheses and methods of their fabrication |
| US9855156B2 (en) | 2014-08-15 | 2018-01-02 | Elixir Medical Corporation | Biodegradable endoprostheses and methods of their fabrication |
| US9259339B1 (en) | 2014-08-15 | 2016-02-16 | Elixir Medical Corporation | Biodegradable endoprostheses and methods of their fabrication |
| US10342561B2 (en) | 2014-09-12 | 2019-07-09 | Board Of Regents Of The University Of Nebraska | Quick-release end effectors and related systems and methods |
| US9527234B2 (en) | 2014-10-02 | 2016-12-27 | Inspiremd, Ltd. | Stent thermoforming apparatus and methods |
| WO2016077478A1 (en) | 2014-11-11 | 2016-05-19 | Board Of Regents Of The University Of Nebraska | Robotic device with compact joint design and related systems and methods |
| CA2969171C (en) | 2014-12-18 | 2023-12-12 | Dsm Ip Assets B.V. | Drug delivery system for delivery of acid sensitive drugs |
| ES2905366T3 (en) * | 2015-02-24 | 2022-04-08 | Univ Freiburg Albert Ludwigs | Phase Segregated Block Copolymers with Tunable Properties |
| CN113318278A (en) * | 2015-05-14 | 2021-08-31 | 用于组织工程和细胞为基础的技术和疗法促进协会 | Ureteral stent, method and application thereof |
| EP3325084B1 (en) | 2015-07-25 | 2019-08-21 | Cardiac Pacemakers, Inc. | Medical electrical lead with biostable pvdf-based materials |
| JP6961146B2 (en) | 2015-08-03 | 2021-11-05 | バーチャル インシジョン コーポレイションVirtual Incision Corporation | Robotic surgical devices, systems and related methods |
| CN106995519B (en) * | 2016-01-25 | 2019-09-20 | 万华化学集团股份有限公司 | A kind of thermoplastic polyurethane and its preparation method and application |
| US11622872B2 (en) | 2016-05-16 | 2023-04-11 | Elixir Medical Corporation | Uncaging stent |
| EP3457985B1 (en) | 2016-05-16 | 2021-02-17 | Elixir Medical Corporation | Uncaging stent |
| CA3024623A1 (en) | 2016-05-18 | 2017-11-23 | Virtual Incision Corporation | Robotic surgical devices, systems and related methods |
| CN116269696A (en) | 2016-08-25 | 2023-06-23 | 内布拉斯加大学董事会 | Quick release tool coupler and related systems and methods |
| CN114872081A (en) | 2016-08-30 | 2022-08-09 | 内布拉斯加大学董事会 | Robotic devices with compact joint design and additional degrees of freedom and related systems and methods |
| JP7222886B2 (en) | 2016-10-21 | 2023-02-15 | ジャベリン メディカル リミテッド | Systems, methods, and devices for embolic protection |
| EP3544539A4 (en) | 2016-11-22 | 2020-08-05 | Board of Regents of the University of Nebraska | Improved gross positioning device and related systems and methods |
| WO2018102430A1 (en) | 2016-11-29 | 2018-06-07 | Virtual Incision Corporation | User controller with user presence detection and related systems and methods |
| US10623846B2 (en) * | 2016-12-06 | 2020-04-14 | Bose Corporation | Earpieces employing viscoelastic materials |
| US10722319B2 (en) | 2016-12-14 | 2020-07-28 | Virtual Incision Corporation | Releasable attachment device for coupling to medical devices and related systems and methods |
| CN110325224B (en) | 2016-12-27 | 2023-01-31 | 波士顿科学国际有限公司 | Degradable scaffold for electrospinning |
| CA3076625A1 (en) | 2017-09-27 | 2019-04-04 | Virtual Incision Corporation | Robotic surgical devices with tracking camera technology and related systems and methods |
| CN107811727B (en) * | 2017-09-29 | 2020-05-01 | 云南靖创液态金属热控技术研发有限公司 | Recyclable heart support |
| EP3735341A4 (en) | 2018-01-05 | 2021-10-06 | Board of Regents of the University of Nebraska | Single-arm robotic device with compact joint design and related systems and methods |
| CN111220290A (en) * | 2018-11-27 | 2020-06-02 | 宁波方太厨具有限公司 | Device for measuring heating rate in equipment cavity |
| JP2022516937A (en) | 2019-01-07 | 2022-03-03 | バーチャル インシジョン コーポレイション | Equipment and methods related to robot-assisted surgery systems |
| US11712329B2 (en) * | 2019-04-04 | 2023-08-01 | Children's Medical Center Corporation | Airway stents |
| CN110330774A (en) * | 2019-07-29 | 2019-10-15 | 中国科学院成都有机化学有限公司 | Composite material and modified degradable ureter rack tube for ureter rack tube |
| EP3795196B1 (en) * | 2019-09-18 | 2022-05-11 | Heraeus Medical GmbH | Device for temporary local application of fluids |
| MX2022010409A (en) * | 2020-02-24 | 2022-09-07 | Pmc Organometallix Inc | Alkyl-bridged tin-based thermal stabilizers for halogenated resins and synthesis and uses therof. |
| CN115089257A (en) * | 2022-08-24 | 2022-09-23 | 北京久事神康医疗科技有限公司 | Embolism device for aneurysm treatment |
Citations (166)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3383336A (en) | 1966-06-27 | 1968-05-14 | Kuyama Hiroshi | Resinous insoluble reaction products |
| US3459725A (en) | 1964-01-17 | 1969-08-05 | Montedison Spa | High molecular weight unsaturated hydrocarbon homopolymers and process for preparing them |
| US3563973A (en) | 1965-03-04 | 1971-02-16 | Radiation Applic Inc | Articles with polymeric memory and method of constructing same |
| US4080357A (en) | 1976-06-07 | 1978-03-21 | Shell Oil Company | Multicomponent halogenated thermoplastic-block copolymer-polymer blends |
| US4503569A (en) | 1983-03-03 | 1985-03-12 | Dotter Charles T | Transluminally placed expandable graft prosthesis |
| US4531933A (en) | 1982-12-07 | 1985-07-30 | C. R. Bard, Inc. | Helical ureteral stent |
| EP0183372A1 (en) | 1984-10-19 | 1986-06-04 | RAYCHEM CORPORATION (a Delaware corporation) | Prosthetic stent |
| EP0183272A2 (en) | 1984-11-30 | 1986-06-04 | Suntory Limited | Vector capable of efficient gene expression and utilization of such vector |
| US4612241A (en) | 1985-06-12 | 1986-09-16 | E. I. Du Pont De Nemours And Company | Impact resistant composites with elastomeric fibers |
| EP0277816A1 (en) | 1987-02-04 | 1988-08-10 | Chisso Corporation | A polysiloxane containing hydroxyl groups and a silicone-modified polyurethane using the same |
| EP0324946A2 (en) | 1988-01-20 | 1989-07-26 | Dainichiseika Color & Chemicals Mfg. Co. Ltd. | Heat-sensitive recording medium |
| EP0343442A2 (en) | 1988-05-25 | 1989-11-29 | Bayer Ag | Mixtures based on metastable amorphous semicrystalline thermoplastics |
| US4893623A (en) | 1986-12-09 | 1990-01-16 | Advanced Surgical Intervention, Inc. | Method and apparatus for treating hypertrophy of the prostate gland |
| EP0368274A2 (en) | 1988-11-11 | 1990-05-16 | Nippon Zeon Co., Ltd. | Structural material and its application |
| US4950258A (en) | 1988-01-28 | 1990-08-21 | Japan Medical Supply Co., Ltd. | Plastic molded articles with shape memory property |
| EP0385443A2 (en) | 1989-02-28 | 1990-09-05 | Daikin Industries, Limited | Polymer material having shape memory characteristics |
| US4969890A (en) | 1987-07-10 | 1990-11-13 | Nippon Zeon Co., Ltd. | Catheter |
| EP0404004A2 (en) | 1989-06-19 | 1990-12-27 | Nippon Unicar Company Limited | A shape memory elastic body |
| US4990155A (en) * | 1989-05-19 | 1991-02-05 | Wilkoff Howard M | Surgical stent method and apparatus |
| US5007926A (en) | 1989-02-24 | 1991-04-16 | The Trustees Of The University Of Pennsylvania | Expandable transluminally implantable tubular prosthesis |
| US5037427A (en) | 1987-03-25 | 1991-08-06 | Terumo Kabushiki Kaisha | Method of implanting a stent within a tubular organ of a living body and of removing same |
| US5062829A (en) | 1989-03-17 | 1991-11-05 | Carter Holt Harvey Plastic Products Group Limited | Relates to devices for administering a substance such as a drug or chemical or the like |
| US5064435A (en) | 1990-06-28 | 1991-11-12 | Schneider (Usa) Inc. | Self-expanding prosthesis having stable axial length |
| US5089005A (en) | 1987-08-13 | 1992-02-18 | Terumo Kabushiki Kaisha | Catheter for the introduction of an expandable member |
| US5100429A (en) | 1989-04-28 | 1992-03-31 | C. R. Bard, Inc. | Endovascular stent and delivery system |
| US5145935A (en) | 1988-09-30 | 1992-09-08 | Mitsubishi Jukogyo Kabushiki Kaisha | Shape memory polyurethane elastomer molded article |
| US5147385A (en) | 1989-11-01 | 1992-09-15 | Schneider (Europe) A.G. | Stent and catheter for the introduction of the stent |
| US5163952A (en) | 1990-09-14 | 1992-11-17 | Michael Froix | Expandable polymeric stent with memory and delivery apparatus and method |
| US5189110A (en) | 1988-12-23 | 1993-02-23 | Asahi Kasei Kogyo Kabushiki Kaisha | Shape memory polymer resin, composition and the shape memorizing molded product thereof |
| US5258020A (en) | 1990-09-14 | 1993-11-02 | Michael Froix | Method of using expandable polymeric stent with memory |
| US5278237A (en) | 1991-08-09 | 1994-01-11 | Daicel-Huls Ltd. | Sculpturing resin composition and process for sculpturing a sculptured product |
| US5282854A (en) | 1989-10-11 | 1994-02-01 | Daikin Industries Ltd. | Fluorine-containing block copolymer and artificial lens comprising the same |
| US5395882A (en) | 1992-09-24 | 1995-03-07 | Rohm Gmbh Chemische Fabrik | Light-scattering polystyrene molding compounds, and molded articles produced therefrom |
| US5421955A (en) | 1991-10-28 | 1995-06-06 | Advanced Cardiovascular Systems, Inc. | Expandable stents and method for making same |
| EP0422693B1 (en) | 1985-01-04 | 1995-06-21 | Thoratec Laboratories Corporation | Method for making an article with shape-memory properties and some of the thus obtained articles |
| US5461114A (en) | 1989-12-28 | 1995-10-24 | Daicel-Huls Ltd. | Composition for use in sculpturing and a process for manufacturing a sculptured product |
| US5466242A (en) | 1994-02-02 | 1995-11-14 | Mori; Katsushi | Stent for biliary, urinary or vascular system |
| US5496311A (en) | 1988-10-28 | 1996-03-05 | Boston Scientific Corporation | Physiologic low stress angioplasty |
| US5506300A (en) | 1985-01-04 | 1996-04-09 | Thoratec Laboratories Corporation | Compositions that soften at predetermined temperatures and the method of making same |
| US5603722A (en) | 1995-06-06 | 1997-02-18 | Quanam Medical Corporation | Intravascular stent |
| US5645559A (en) | 1992-05-08 | 1997-07-08 | Schneider (Usa) Inc | Multiple layer stent |
| US5665822A (en) | 1991-10-07 | 1997-09-09 | Landec Corporation | Thermoplastic Elastomers |
| US5670161A (en) * | 1996-05-28 | 1997-09-23 | Healy; Kevin E. | Biodegradable stent |
| US5674241A (en) | 1995-02-22 | 1997-10-07 | Menlo Care, Inc. | Covered expanding mesh stent |
| US5674242A (en) * | 1995-06-06 | 1997-10-07 | Quanam Medical Corporation | Endoprosthetic device with therapeutic compound |
| US5741333A (en) | 1995-04-12 | 1998-04-21 | Corvita Corporation | Self-expanding stent for a medical device to be introduced into a cavity of a body |
| US5755769A (en) | 1992-03-12 | 1998-05-26 | Laboratoire Perouse Implant | Expansible endoprosthesis for a human or animal tubular organ, and fitting tool for use thereof |
| US5762625A (en) | 1992-09-08 | 1998-06-09 | Kabushikikaisha Igaki Iryo Sekkei | Luminal stent and device for inserting luminal stent |
| US5766188A (en) | 1995-05-08 | 1998-06-16 | Kabushikikaisha Igaki Iryo Sekkei | Medical suturing material |
| US5769883A (en) | 1991-10-04 | 1998-06-23 | Scimed Life Systems, Inc. | Biodegradable drug delivery vascular stent |
| US5776162A (en) | 1997-01-03 | 1998-07-07 | Nitinol Medical Technologies, Inc. | Vessel implantable shape memory appliance with superelastic hinged joint |
| US5800516A (en) | 1996-08-08 | 1998-09-01 | Cordis Corporation | Deployable and retrievable shape memory stent/tube and method |
| US5817100A (en) | 1994-02-07 | 1998-10-06 | Kabushikikaisya Igaki Iryo Sekkei | Stent device and stent supplying system |
| US5830179A (en) | 1996-04-09 | 1998-11-03 | Endocare, Inc. | Urological stent therapy system and method |
| US5833651A (en) * | 1996-11-08 | 1998-11-10 | Medtronic, Inc. | Therapeutic intraluminal stents |
| US5843096A (en) | 1995-05-08 | 1998-12-01 | Igaki; Keiji | Medical suturing material |
| US5849037A (en) | 1995-04-12 | 1998-12-15 | Corvita Corporation | Self-expanding stent for a medical device to be introduced into a cavity of a body, and method for its preparation |
| US5876432A (en) | 1994-04-01 | 1999-03-02 | Gore Enterprise Holdings, Inc. | Self-expandable helical intravascular stent and stent-graft |
| US5880240A (en) | 1996-04-03 | 1999-03-09 | Arakawa Chemical Industries, Ltd. | Alkyl-containing porous resin, process for its preparation and its use |
| US5889118A (en) | 1996-06-03 | 1999-03-30 | Minnesota Mining And Manufacturing Company | Thermomorphic "smart" pressure sensitive adhesives |
| US5900246A (en) | 1993-03-18 | 1999-05-04 | Cedars-Sinai Medical Center | Drug incorporating and releasing polymeric coating for bioprosthesis |
| US5908918A (en) | 1996-07-17 | 1999-06-01 | Chronopol, Inc. | Impact modified polylactide |
| US5910357A (en) | 1996-07-12 | 1999-06-08 | Nitto Denko Corporation | Separation membrane and method of producing the same, and shape memory polymer composition |
| US5935506A (en) | 1995-10-24 | 1999-08-10 | Biotronik Meβ- und Therapiegerate GmbH & Co. Ingenieurburo Berlin | Method for the manufacture of intraluminal stents of bioresorbable polymeric material |
| US5955559A (en) | 1996-09-17 | 1999-09-21 | Shell Oil Company | Cast polyurethane elastomers containing low polarity amine curing agents |
| US5954744A (en) | 1995-06-06 | 1999-09-21 | Quanam Medical Corporation | Intravascular stent |
| US5961547A (en) | 1995-06-22 | 1999-10-05 | Ali Razavi | Temporary stent |
| US5964744A (en) | 1993-01-04 | 1999-10-12 | Menlo Care, Inc. | Polymeric medical device systems having shape memory |
| US5997563A (en) | 1998-09-28 | 1999-12-07 | Medtronic, Inc. | Implantable stent having variable diameter |
| US6024764A (en) | 1997-08-19 | 2000-02-15 | Intermedics, Inc. | Apparatus for imparting physician-determined shapes to implantable tubular devices |
| US6033413A (en) | 1998-04-20 | 2000-03-07 | Endocare, Inc. | Stent delivery system |
| US6045568A (en) | 1991-03-08 | 2000-04-04 | Igaki; Keiji | Luminal stent, holding structure therefor and device for attaching luminal stent |
| JP2000119465A (en) | 1998-10-13 | 2000-04-25 | Tokai Rubber Ind Ltd | Shape-memory resin composition and shape-memory active made thereof |
| US6056844A (en) | 1997-06-06 | 2000-05-02 | Triton Systems, Inc. | Temperature-controlled induction heating of polymeric materials |
| EP1016424A1 (en) | 1998-12-31 | 2000-07-05 | Ethicon, Inc. | Radiopaque polymer coating |
| US6086204A (en) | 1999-09-20 | 2000-07-11 | Magnante; Peter C. | Methods and devices to design and fabricate surfaces on contact lenses and on corneal tissue that correct the eye's optical aberrations |
| US6086610A (en) | 1996-10-22 | 2000-07-11 | Nitinol Devices & Components | Composite self expanding stent device having a restraining element |
| JP3068611B1 (en) | 1999-06-15 | 2000-07-24 | 三菱重工業株式会社 | Ink supply device and method of mounting surface cover for preventing contamination on ink tray of ink supply device |
| JP3068610B1 (en) | 1999-06-10 | 2000-07-24 | 多摩川精機株式会社 | Concentric shaft motor |
| JP2000319423A (en) | 1999-05-12 | 2000-11-21 | Showa Highpolymer Co Ltd | Flexible material |
| WO2000071554A2 (en) | 1999-05-24 | 2000-11-30 | California Institute Of Technology | Imidazolidine-based metal carbene metathesis catalysts |
| US6156842A (en) | 1998-03-11 | 2000-12-05 | The Dow Chemical Company | Structures and fabricated articles having shape memory made from α-olefin/vinyl or vinylidene aromatic and/or hindered aliphatic vinyl or vinylidene interpolymers |
| US6160084A (en) | 1998-02-23 | 2000-12-12 | Massachusetts Institute Of Technology | Biodegradable shape memory polymers |
| WO2000078246A2 (en) | 1999-06-21 | 2000-12-28 | Endocare, Inc. | Stent insertion device |
| WO2001007499A1 (en) | 1999-07-20 | 2001-02-01 | Aortech Biomaterials Pty Ltd. | Shape memory polyurethane or polyurethane-urea polymers |
| US6183248B1 (en) | 1998-11-30 | 2001-02-06 | Muhammad Chishti | System and method for releasing tooth positioning appliances |
| US6200335B1 (en) | 1997-03-31 | 2001-03-13 | Kabushikikaisha Igaki Iryo Sekkei | Stent for vessel |
| US6217609B1 (en) | 1998-06-30 | 2001-04-17 | Schneider (Usa) Inc | Implantable endoprosthesis with patterned terminated ends and methods for making same |
| US6241691B1 (en) | 1997-12-05 | 2001-06-05 | Micrus Corporation | Coated superelastic stent |
| US6248129B1 (en) * | 1990-09-14 | 2001-06-19 | Quanam Medical Corporation | Expandable polymeric stent with memory and delivery apparatus and method |
| US6283992B1 (en) | 1995-11-27 | 2001-09-04 | Schneider (Europe) Gmbh | Conical stent |
| US6287326B1 (en) | 1999-08-02 | 2001-09-11 | Alsius Corporation | Catheter with coiled multi-lumen heat transfer extension |
| US20010029398A1 (en) | 1999-06-03 | 2001-10-11 | Jadhav Balkrishna S. | Bioresorbable stent |
| WO2001080936A1 (en) | 2000-04-20 | 2001-11-01 | Kawasumi Laboratories, Inc. | Stent |
| US6315791B1 (en) | 1996-12-03 | 2001-11-13 | Atrium Medical Corporation | Self-expanding prothesis |
| US6323459B1 (en) | 1996-09-05 | 2001-11-27 | Medtronic, Inc. | Selectively activated shape memory device |
| WO2001091822A1 (en) | 2000-05-31 | 2001-12-06 | Mnemoscience Gmbh | Shape memory thermoplastics and polymer networks for tissue engineering |
| WO2001093783A2 (en) | 2000-06-07 | 2001-12-13 | Advanced Cardiovascular Systems, Inc. | Variable stiffness stent |
| US20020007222A1 (en) | 2000-04-11 | 2002-01-17 | Ashvin Desai | Method and apparatus for supporting a body organ |
| US20020015519A1 (en) | 1998-12-11 | 2002-02-07 | Lord Corporation | Fiber substrate adhesion and coatings by contact metathesis polymerization |
| US6348065B1 (en) | 1995-03-01 | 2002-02-19 | Scimed Life Systems, Inc. | Longitudinally flexible expandable stent |
| WO2002015951A2 (en) | 2000-08-23 | 2002-02-28 | Thoratec Corporation | Coated vascular grafts and methods of use |
| US6364904B1 (en) | 1999-07-02 | 2002-04-02 | Scimed Life Systems, Inc. | Helically formed stent/graft assembly |
| US20020055787A1 (en) | 1998-10-15 | 2002-05-09 | Charles D. Lennox | Treating urinary retention |
| US6388043B1 (en) | 1998-02-23 | 2002-05-14 | Mnemoscience Gmbh | Shape memory polymers |
| WO2002039875A2 (en) | 2000-11-17 | 2002-05-23 | B. Braun Medical Inc. | Method for deploying a thermo-mechanically expandable stent |
| US6406493B1 (en) | 2000-06-02 | 2002-06-18 | Hosheng Tu | Expandable annuloplasty ring and methods of use |
| US6413273B1 (en) | 1998-11-25 | 2002-07-02 | Israel Aircraft Industries Ltd. | Method and system for temporarily supporting a tubular organ |
| US20020137864A1 (en) | 2001-01-24 | 2002-09-26 | Tong Tat Hung | Shape memory styrene copolymer |
| US20020151967A1 (en) | 1996-04-09 | 2002-10-17 | Mikus Paul W. | Urological stent therapy system and method |
| US20020156523A1 (en) | 1994-08-31 | 2002-10-24 | Lilip Lau | Exterior supported self-expanding stent-graft |
| WO2002083786A1 (en) | 2001-04-11 | 2002-10-24 | Cheong Seok Hong | Shape memory rubber composition |
| US6478773B1 (en) | 1998-12-21 | 2002-11-12 | Micrus Corporation | Apparatus for deployment of micro-coil using a catheter |
| US6485507B1 (en) | 1999-07-28 | 2002-11-26 | Scimed Life Systems | Multi-property nitinol by heat treatment |
| US20020198584A1 (en) | 1998-08-21 | 2002-12-26 | Unsworth John Duncan | Shape memory tubular stent |
| US6500204B1 (en) | 1998-09-08 | 2002-12-31 | Kabushikikaisha Igaki Iryo Sekkei | Stent for vessels |
| US6517569B2 (en) | 1998-09-14 | 2003-02-11 | Endocare, Inc. | Insertion device for stents and methods for use |
| US6517570B1 (en) | 1994-08-31 | 2003-02-11 | Gore Enterprise Holdings, Inc. | Exterior supported self-expanding stent-graft |
| US20030033001A1 (en) | 2001-02-27 | 2003-02-13 | Keiji Igaki | Stent holding member and stent feeding system |
| US20030040803A1 (en) | 2001-08-23 | 2003-02-27 | Rioux Robert F. | Maintaining an open passageway through a body lumen |
| US6530950B1 (en) | 1999-01-12 | 2003-03-11 | Quanam Medical Corporation | Intraluminal stent having coaxial polymer member |
| US6538089B1 (en) | 1999-02-05 | 2003-03-25 | Forskarpatent I Syd Ab | Gels with a shape memory |
| US20030060530A1 (en) | 2001-07-24 | 2003-03-27 | Topolkaraev Vasily A. | Methods of making humidity activated materials having shape-memory |
| US20030060793A1 (en) | 2001-07-24 | 2003-03-27 | Topolkaraev Vasily A. | Disposable products having humidity activated materials with shape-memory |
| WO2003035743A1 (en) | 2001-10-25 | 2003-05-01 | Ticona Gmbh | Method for reducing the toughness of shaped plastic parts to be mechanically worked and the use thereof |
| US20030114777A1 (en) * | 2001-12-18 | 2003-06-19 | Scimed Life Systems, Inc. | Super elastic guidewire with shape retention tip |
| US6596818B1 (en) | 1996-10-08 | 2003-07-22 | Alan M. Zamore | Irradiation conversion of thermoplastic to thermoset polymers |
| US20030147046A1 (en) | 2002-02-06 | 2003-08-07 | Shadduck John H. | Adaptive optic lens system and method of use |
| US20030173702A1 (en) | 2001-04-18 | 2003-09-18 | Keiji Igaki | Melting and spinning device and melting and spinning method |
| US20030191276A1 (en) | 2002-02-26 | 2003-10-09 | Mnemoscience Gmbh | Polymeric networks |
| WO2003084491A1 (en) | 2002-04-10 | 2003-10-16 | Mnemoscience Gmbh | Method for treating hair with form memory polymers |
| WO2003084490A1 (en) | 2002-04-10 | 2003-10-16 | Mnemoscience Gmbh | Method for the generation of memory effects on hair |
| WO2003088818A2 (en) | 2002-04-18 | 2003-10-30 | Mnemoscience Gmbh | Biodegradable shape memory polymeric sutures |
| WO2003015840A3 (en) | 2001-08-17 | 2003-12-04 | Advanced Bio Prosthetic Surfac | Embolic protection device |
| US6679605B2 (en) | 2000-05-22 | 2004-01-20 | Medennium, Inc. | Crystalline polymeric compositions for ophthalmic devices |
| US20040014929A1 (en) | 2002-04-18 | 2004-01-22 | Mnemoscience Gmbh | Polyester urethanes |
| US20040015261A1 (en) | 2000-08-28 | 2004-01-22 | Hofmann Gregory J. | Shape memory polymer or alloy ophthalmic lens mold and methods of forming ophthalmic products |
| DE10228120A1 (en) | 2002-06-24 | 2004-01-22 | Mnemoscience Gmbh | Permanent deforation of hair comprises applying a shape-memory composition to the hair before, during or after imparting a predetermined shape to the hair and then fixing the shape |
| WO2004006885A2 (en) | 2002-07-10 | 2004-01-22 | Mnemoscience Gmbh | Systems for releasing active ingredients, based on biodegradable or biocompatible polymers with a shape memory effect |
| US20040024143A1 (en) | 2002-04-18 | 2004-02-05 | Mnemoscience Gmbh | Interpenetrating networks |
| US20040030062A1 (en) | 2002-05-02 | 2004-02-12 | Mather Patrick T. | Castable shape memory polymers |
| WO2004032799A2 (en) | 2002-10-11 | 2004-04-22 | Boston Scientific Limited | Implantable medical devices |
| WO2004033553A1 (en) | 2002-10-11 | 2004-04-22 | University Of Connecticut | Crosslinked polycyclooctene |
| WO2004033539A1 (en) | 2002-10-11 | 2004-04-22 | University Of Connecticut | Blends of amorphous and semicrystalline polymers having shape memory properties |
| US20040153025A1 (en) | 2003-02-03 | 2004-08-05 | Seifert Paul S. | Systems and methods of de-endothelialization |
| WO2004073690A1 (en) | 2003-02-19 | 2004-09-02 | Mnemoscience Gmbh | Self-expanding device for the gastrointestinal or urogenital area |
| US20040199242A1 (en) * | 2001-12-27 | 2004-10-07 | James Hong | Hybrid intravascular stent |
| WO2004090042A1 (en) | 2003-04-10 | 2004-10-21 | Mnemoscience Gmbh | Blends with shape memory characteristics |
| USRE38653E1 (en) | 1991-03-08 | 2004-11-16 | Kabushikikaisha Igaki Iryo Sekkei | Luminal stent, holding structure therefor and device for attaching luminal stent |
| WO2004110515A1 (en) | 2003-06-13 | 2004-12-23 | Mnemoscience Gmbh | Biodegradable stents |
| US6837901B2 (en) | 2001-04-27 | 2005-01-04 | Intek Technology L.L.C. | Methods for delivering, repositioning and/or retrieving self-expanding stents |
| WO2005009523A1 (en) | 2003-07-18 | 2005-02-03 | Boston Scientific Limited | Medical devices |
| USRE38711E1 (en) | 1991-03-08 | 2005-03-15 | Kabushikikaisha Igaki Iryo Sekkei | Luminal stent, holding structure therefor and device for attaching luminal stent |
| WO2005070988A1 (en) | 2004-01-20 | 2005-08-04 | Ucl Biomedica Plc | Polymer for use in conduits, medical devices and biomedical surface modification |
| US20050245719A1 (en) | 2002-10-11 | 2005-11-03 | Mather Patrick T | Shape memory polymers based on semicrystalline thermoplastic polyurethanes bearing nanostructured hard segments |
| US20050251249A1 (en) | 2002-10-11 | 2005-11-10 | Sahatjian Ronald A | Endoprostheses |
| US7037288B2 (en) | 2002-01-14 | 2006-05-02 | Codman & Shurtleff, Inc. | Anti-block catheter |
| US7070615B1 (en) | 2000-03-13 | 2006-07-04 | Keiji Igaki | Linear material for blood vessel stent and blood vessel stent utilizing same |
| US20060235501A1 (en) | 2003-05-23 | 2006-10-19 | Keiji Igaki | Stent supplying device |
| US20070260302A1 (en) | 2003-10-15 | 2007-11-08 | Kabushikikaisha Igaki Iryo Sekkei | Device for delivery of stent for vessel |
| US7367990B2 (en) | 2002-09-25 | 2008-05-06 | Kabushikikaisha Igaki Iryo Sekkei | Thread for vascular stent and vascular stent using the thread |
| JP4100831B2 (en) | 1999-07-16 | 2008-06-11 | 大成建設株式会社 | Gap covering device for buildings with seismic isolation structure |
| US7410498B2 (en) | 2000-06-29 | 2008-08-12 | Pentech Medical Devices Ltd. | Polymeric stents and other surgical articles |
| US7473273B2 (en) | 2002-01-22 | 2009-01-06 | Medtronic Vascular, Inc. | Stent assembly with therapeutic agent exterior banding |
| US7498042B2 (en) | 2000-11-30 | 2009-03-03 | Kyoto Medical Planning Co., Ltd. | Stent for blood vessel and material for stent for blood vessel |
| US8070793B2 (en) | 2004-11-12 | 2011-12-06 | Kabushikikaisha Igaki Iryo Sekkei | Stent for vessel |
Family Cites Families (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4820302A (en) * | 1982-04-22 | 1989-04-11 | Sterling Drug Inc. | Bio compatible and blood compatible materials and methods |
| DE8223248U1 (en) | 1982-08-18 | 1983-08-25 | Huelsta Werke Huels Kg, 4424 Stadtlohn | MATTRESS |
| JPS62192440U (en) | 1986-05-23 | 1987-12-07 | ||
| JPS63145325A (en) | 1986-12-08 | 1988-06-17 | Daiseru Hiyurusu Kk | Resin material for shaping of model |
| JPS63145325U (en) | 1987-03-13 | 1988-09-26 | ||
| JPS63179955U (en) | 1987-05-15 | 1988-11-21 | ||
| JPH0832766B2 (en) | 1988-12-07 | 1996-03-29 | 帝人株式会社 | Shape memory cross-linked polymer molded article and method for producing the same |
| JP2656343B2 (en) | 1989-03-30 | 1997-09-24 | 株式会社クラレ | Trans polyisoprene resin composition |
| JP2906614B2 (en) | 1990-08-29 | 1999-06-21 | 株式会社浜松プラテック | Temperature-sensitive transparent-opaque conversion composition and temperature sensor using temperature-sensitive transparent-opaque conversion composition |
| EP0674683B1 (en) | 1992-12-21 | 1997-10-15 | Raychem Corporation | Polymeric blends |
| NL9400519A (en) | 1994-03-31 | 1995-11-01 | Rijksuniversiteit | Intravascular polymeric stent. |
| JPH07292040A (en) | 1994-04-28 | 1995-11-07 | Yoshihito Osada | Heat-sensitive shape-memory gel |
| US6099562A (en) | 1996-06-13 | 2000-08-08 | Schneider (Usa) Inc. | Drug coating with topcoat |
| CN1052915C (en) * | 1995-11-27 | 2000-05-31 | 中国医学科学院生物医学工程研究所 | Medical carrier of protein coat for carrying gene and its prodn. method |
| JP3669529B2 (en) | 1996-03-19 | 2005-07-06 | 三井化学株式会社 | Shape memory vulcanized rubber molding |
| JPH101545A (en) | 1996-04-19 | 1998-01-06 | Pilot Ink Co Ltd | Thermoplastic resin composition and its molded product capable of temperature-dependent deformation and shaping |
| DE19746653C1 (en) * | 1997-10-22 | 1998-11-12 | Pfaff Ag G M | Sewing machine thread cutting |
| JPH11154420A (en) | 1997-11-21 | 1999-06-08 | Hitachi Chem Co Ltd | Resin composition for electrical insulation and electric appliance electrically insulated |
| GB9803770D0 (en) | 1998-02-23 | 1998-04-15 | Unilever Plc | Detergent compositions |
| JPH11302493A (en) | 1998-04-23 | 1999-11-02 | Tokai Rubber Ind Ltd | Shape memory resin composition and shape memory article using the same |
| GB9819764D0 (en) | 1998-09-10 | 1998-11-04 | Oxagen Limited | Method |
| US6336937B1 (en) | 1998-12-09 | 2002-01-08 | Gore Enterprise Holdings, Inc. | Multi-stage expandable stent-graft |
| US6958212B1 (en) | 1999-02-01 | 2005-10-25 | Eidgenossische Technische Hochschule Zurich | Conjugate addition reactions for the controlled delivery of pharmaceutically active compounds |
| US6908624B2 (en) | 1999-12-23 | 2005-06-21 | Advanced Cardiovascular Systems, Inc. | Coating for implantable devices and a method of forming the same |
| WO2001056641A1 (en) | 2000-02-04 | 2001-08-09 | C. R. Bard, Inc. | Triple lumen stone balloon catheter and method |
| DE60118790T2 (en) * | 2000-12-19 | 2006-08-31 | Bausch & Lomb Incorporated | POLYMERIC BIOMATERIALS CONTAINING SILSESQUIOXAN MONOMERS |
| US6550480B2 (en) | 2001-01-31 | 2003-04-22 | Numed/Tech Llc | Lumen occluders made from thermodynamic materials |
| WO2002064014A2 (en) * | 2001-02-09 | 2002-08-22 | Endoluminal Therapeutics, Inc. | Endomural therapy |
| US6679906B2 (en) * | 2001-07-13 | 2004-01-20 | Radiant Medical, Inc. | Catheter system with on-board temperature probe |
| JP2005503865A (en) * | 2001-09-28 | 2005-02-10 | ボストン サイエンティフィック リミテッド | Medical device comprising nanomaterial and treatment method using the same |
| US7008979B2 (en) * | 2002-04-30 | 2006-03-07 | Hydromer, Inc. | Coating composition for multiple hydrophilic applications |
| WO2004011525A1 (en) | 2002-07-30 | 2004-02-05 | University Of Connecticut | Nonionic telechelic polymers incorporating polyhedral oligosilsesquioxane (poss) and uses thereof |
| US7067606B2 (en) * | 2002-07-30 | 2006-06-27 | University Of Connecticut | Nonionic telechelic polymers incorporating polyhedral oligosilsesquioxane (POSS) and uses thereof |
| JP4379044B2 (en) | 2003-08-22 | 2009-12-09 | 株式会社カネカ | Indwelling stent |
-
2004
- 2004-10-05 US US10/958,435 patent/US7794494B2/en not_active Expired - Fee Related
-
2005
- 2005-10-05 WO PCT/US2005/035444 patent/WO2006041767A2/en active Application Filing
- 2005-10-05 CA CA002583191A patent/CA2583191A1/en not_active Abandoned
- 2005-10-05 AU AU2005294569A patent/AU2005294569A1/en not_active Abandoned
- 2005-10-05 EP EP05810344A patent/EP1824412A2/en not_active Withdrawn
-
2010
- 2010-09-13 US US12/880,483 patent/US20140135889A9/en not_active Abandoned
- 2010-11-05 US US12/940,388 patent/US8784465B2/en not_active Expired - Fee Related
-
2012
- 2012-06-11 US US13/493,532 patent/US9115245B2/en not_active Expired - Fee Related
Patent Citations (192)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3459725A (en) | 1964-01-17 | 1969-08-05 | Montedison Spa | High molecular weight unsaturated hydrocarbon homopolymers and process for preparing them |
| US3563973A (en) | 1965-03-04 | 1971-02-16 | Radiation Applic Inc | Articles with polymeric memory and method of constructing same |
| US3383336A (en) | 1966-06-27 | 1968-05-14 | Kuyama Hiroshi | Resinous insoluble reaction products |
| US4080357A (en) | 1976-06-07 | 1978-03-21 | Shell Oil Company | Multicomponent halogenated thermoplastic-block copolymer-polymer blends |
| US4531933A (en) | 1982-12-07 | 1985-07-30 | C. R. Bard, Inc. | Helical ureteral stent |
| US4503569A (en) | 1983-03-03 | 1985-03-12 | Dotter Charles T | Transluminally placed expandable graft prosthesis |
| EP0183372A1 (en) | 1984-10-19 | 1986-06-04 | RAYCHEM CORPORATION (a Delaware corporation) | Prosthetic stent |
| EP0183272A2 (en) | 1984-11-30 | 1986-06-04 | Suntory Limited | Vector capable of efficient gene expression and utilization of such vector |
| US5506300A (en) | 1985-01-04 | 1996-04-09 | Thoratec Laboratories Corporation | Compositions that soften at predetermined temperatures and the method of making same |
| EP0422693B1 (en) | 1985-01-04 | 1995-06-21 | Thoratec Laboratories Corporation | Method for making an article with shape-memory properties and some of the thus obtained articles |
| US4612241A (en) | 1985-06-12 | 1986-09-16 | E. I. Du Pont De Nemours And Company | Impact resistant composites with elastomeric fibers |
| US4893623A (en) | 1986-12-09 | 1990-01-16 | Advanced Surgical Intervention, Inc. | Method and apparatus for treating hypertrophy of the prostate gland |
| EP0277816A1 (en) | 1987-02-04 | 1988-08-10 | Chisso Corporation | A polysiloxane containing hydroxyl groups and a silicone-modified polyurethane using the same |
| US5037427A (en) | 1987-03-25 | 1991-08-06 | Terumo Kabushiki Kaisha | Method of implanting a stent within a tubular organ of a living body and of removing same |
| US4969890A (en) | 1987-07-10 | 1990-11-13 | Nippon Zeon Co., Ltd. | Catheter |
| US5089005A (en) | 1987-08-13 | 1992-02-18 | Terumo Kabushiki Kaisha | Catheter for the introduction of an expandable member |
| EP0324946A2 (en) | 1988-01-20 | 1989-07-26 | Dainichiseika Color & Chemicals Mfg. Co. Ltd. | Heat-sensitive recording medium |
| US4950258A (en) | 1988-01-28 | 1990-08-21 | Japan Medical Supply Co., Ltd. | Plastic molded articles with shape memory property |
| EP0343442A2 (en) | 1988-05-25 | 1989-11-29 | Bayer Ag | Mixtures based on metastable amorphous semicrystalline thermoplastics |
| US5145935A (en) | 1988-09-30 | 1992-09-08 | Mitsubishi Jukogyo Kabushiki Kaisha | Shape memory polyurethane elastomer molded article |
| US5496311A (en) | 1988-10-28 | 1996-03-05 | Boston Scientific Corporation | Physiologic low stress angioplasty |
| EP0368274A2 (en) | 1988-11-11 | 1990-05-16 | Nippon Zeon Co., Ltd. | Structural material and its application |
| US5189110A (en) | 1988-12-23 | 1993-02-23 | Asahi Kasei Kogyo Kabushiki Kaisha | Shape memory polymer resin, composition and the shape memorizing molded product thereof |
| US5007926A (en) | 1989-02-24 | 1991-04-16 | The Trustees Of The University Of Pennsylvania | Expandable transluminally implantable tubular prosthesis |
| EP0385443A2 (en) | 1989-02-28 | 1990-09-05 | Daikin Industries, Limited | Polymer material having shape memory characteristics |
| US5062829A (en) | 1989-03-17 | 1991-11-05 | Carter Holt Harvey Plastic Products Group Limited | Relates to devices for administering a substance such as a drug or chemical or the like |
| US5100429A (en) | 1989-04-28 | 1992-03-31 | C. R. Bard, Inc. | Endovascular stent and delivery system |
| US4990155A (en) * | 1989-05-19 | 1991-02-05 | Wilkoff Howard M | Surgical stent method and apparatus |
| EP0404004A2 (en) | 1989-06-19 | 1990-12-27 | Nippon Unicar Company Limited | A shape memory elastic body |
| US5282854A (en) | 1989-10-11 | 1994-02-01 | Daikin Industries Ltd. | Fluorine-containing block copolymer and artificial lens comprising the same |
| US5147385A (en) | 1989-11-01 | 1992-09-15 | Schneider (Europe) A.G. | Stent and catheter for the introduction of the stent |
| US5461114A (en) | 1989-12-28 | 1995-10-24 | Daicel-Huls Ltd. | Composition for use in sculpturing and a process for manufacturing a sculptured product |
| US5064435A (en) | 1990-06-28 | 1991-11-12 | Schneider (Usa) Inc. | Self-expanding prosthesis having stable axial length |
| US6248129B1 (en) * | 1990-09-14 | 2001-06-19 | Quanam Medical Corporation | Expandable polymeric stent with memory and delivery apparatus and method |
| US5607467A (en) | 1990-09-14 | 1997-03-04 | Froix; Michael | Expandable polymeric stent with memory and delivery apparatus and method |
| US5258020A (en) | 1990-09-14 | 1993-11-02 | Michael Froix | Method of using expandable polymeric stent with memory |
| US5163952A (en) | 1990-09-14 | 1992-11-17 | Michael Froix | Expandable polymeric stent with memory and delivery apparatus and method |
| US6080177A (en) | 1991-03-08 | 2000-06-27 | Igaki; Keiji | Luminal stent, holding structure therefor and device for attaching luminal stent |
| US6045568A (en) | 1991-03-08 | 2000-04-04 | Igaki; Keiji | Luminal stent, holding structure therefor and device for attaching luminal stent |
| USRE38653E1 (en) | 1991-03-08 | 2004-11-16 | Kabushikikaisha Igaki Iryo Sekkei | Luminal stent, holding structure therefor and device for attaching luminal stent |
| USRE38711E1 (en) | 1991-03-08 | 2005-03-15 | Kabushikikaisha Igaki Iryo Sekkei | Luminal stent, holding structure therefor and device for attaching luminal stent |
| US5278237A (en) | 1991-08-09 | 1994-01-11 | Daicel-Huls Ltd. | Sculpturing resin composition and process for sculpturing a sculptured product |
| US5769883A (en) | 1991-10-04 | 1998-06-23 | Scimed Life Systems, Inc. | Biodegradable drug delivery vascular stent |
| US5665822A (en) | 1991-10-07 | 1997-09-09 | Landec Corporation | Thermoplastic Elastomers |
| US5421955B1 (en) | 1991-10-28 | 1998-01-20 | Advanced Cardiovascular System | Expandable stents and method for making same |
| US5421955A (en) | 1991-10-28 | 1995-06-06 | Advanced Cardiovascular Systems, Inc. | Expandable stents and method for making same |
| US5755769A (en) | 1992-03-12 | 1998-05-26 | Laboratoire Perouse Implant | Expansible endoprosthesis for a human or animal tubular organ, and fitting tool for use thereof |
| US5645559A (en) | 1992-05-08 | 1997-07-08 | Schneider (Usa) Inc | Multiple layer stent |
| US5762625A (en) | 1992-09-08 | 1998-06-09 | Kabushikikaisha Igaki Iryo Sekkei | Luminal stent and device for inserting luminal stent |
| US5395882A (en) | 1992-09-24 | 1995-03-07 | Rohm Gmbh Chemische Fabrik | Light-scattering polystyrene molding compounds, and molded articles produced therefrom |
| US5964744A (en) | 1993-01-04 | 1999-10-12 | Menlo Care, Inc. | Polymeric medical device systems having shape memory |
| US5900246A (en) | 1993-03-18 | 1999-05-04 | Cedars-Sinai Medical Center | Drug incorporating and releasing polymeric coating for bioprosthesis |
| US5466242A (en) | 1994-02-02 | 1995-11-14 | Mori; Katsushi | Stent for biliary, urinary or vascular system |
| US5817100A (en) | 1994-02-07 | 1998-10-06 | Kabushikikaisya Igaki Iryo Sekkei | Stent device and stent supplying system |
| US5876432A (en) | 1994-04-01 | 1999-03-02 | Gore Enterprise Holdings, Inc. | Self-expandable helical intravascular stent and stent-graft |
| US20020156523A1 (en) | 1994-08-31 | 2002-10-24 | Lilip Lau | Exterior supported self-expanding stent-graft |
| US6517570B1 (en) | 1994-08-31 | 2003-02-11 | Gore Enterprise Holdings, Inc. | Exterior supported self-expanding stent-graft |
| US5968070A (en) | 1995-02-22 | 1999-10-19 | Cordis Corporation | Covered expanding mesh stent |
| US5674241A (en) | 1995-02-22 | 1997-10-07 | Menlo Care, Inc. | Covered expanding mesh stent |
| US6348065B1 (en) | 1995-03-01 | 2002-02-19 | Scimed Life Systems, Inc. | Longitudinally flexible expandable stent |
| US5849037A (en) | 1995-04-12 | 1998-12-15 | Corvita Corporation | Self-expanding stent for a medical device to be introduced into a cavity of a body, and method for its preparation |
| US5741333A (en) | 1995-04-12 | 1998-04-21 | Corvita Corporation | Self-expanding stent for a medical device to be introduced into a cavity of a body |
| US5843096A (en) | 1995-05-08 | 1998-12-01 | Igaki; Keiji | Medical suturing material |
| US5766188A (en) | 1995-05-08 | 1998-06-16 | Kabushikikaisha Igaki Iryo Sekkei | Medical suturing material |
| US5674242A (en) * | 1995-06-06 | 1997-10-07 | Quanam Medical Corporation | Endoprosthetic device with therapeutic compound |
| US5603722A (en) | 1995-06-06 | 1997-02-18 | Quanam Medical Corporation | Intravascular stent |
| US5954744A (en) | 1995-06-06 | 1999-09-21 | Quanam Medical Corporation | Intravascular stent |
| US5961547A (en) | 1995-06-22 | 1999-10-05 | Ali Razavi | Temporary stent |
| US5935506A (en) | 1995-10-24 | 1999-08-10 | Biotronik Meβ- und Therapiegerate GmbH & Co. Ingenieurburo Berlin | Method for the manufacture of intraluminal stents of bioresorbable polymeric material |
| US6283992B1 (en) | 1995-11-27 | 2001-09-04 | Schneider (Europe) Gmbh | Conical stent |
| US5880240A (en) | 1996-04-03 | 1999-03-09 | Arakawa Chemical Industries, Ltd. | Alkyl-containing porous resin, process for its preparation and its use |
| US6174305B1 (en) | 1996-04-09 | 2001-01-16 | Endocare, Inc. | Urological stent therapy system and method |
| US5830179A (en) | 1996-04-09 | 1998-11-03 | Endocare, Inc. | Urological stent therapy system and method |
| US5928217A (en) | 1996-04-09 | 1999-07-27 | Endocare, Inc. | Urological stent therapy system and method |
| US20020151967A1 (en) | 1996-04-09 | 2002-10-17 | Mikus Paul W. | Urological stent therapy system and method |
| US6416545B1 (en) | 1996-04-09 | 2002-07-09 | Endocare, Inc. | Urological stent therapy system and method |
| US6139536A (en) | 1996-04-09 | 2000-10-31 | Endocare, Inc. | Urological stent therapy system and method |
| US5670161A (en) * | 1996-05-28 | 1997-09-23 | Healy; Kevin E. | Biodegradable stent |
| US5889118A (en) | 1996-06-03 | 1999-03-30 | Minnesota Mining And Manufacturing Company | Thermomorphic "smart" pressure sensitive adhesives |
| US5910357A (en) | 1996-07-12 | 1999-06-08 | Nitto Denko Corporation | Separation membrane and method of producing the same, and shape memory polymer composition |
| US5908918A (en) | 1996-07-17 | 1999-06-01 | Chronopol, Inc. | Impact modified polylactide |
| US5800516A (en) | 1996-08-08 | 1998-09-01 | Cordis Corporation | Deployable and retrievable shape memory stent/tube and method |
| US6323459B1 (en) | 1996-09-05 | 2001-11-27 | Medtronic, Inc. | Selectively activated shape memory device |
| US5955559A (en) | 1996-09-17 | 1999-09-21 | Shell Oil Company | Cast polyurethane elastomers containing low polarity amine curing agents |
| US6596818B1 (en) | 1996-10-08 | 2003-07-22 | Alan M. Zamore | Irradiation conversion of thermoplastic to thermoset polymers |
| US6086610A (en) | 1996-10-22 | 2000-07-11 | Nitinol Devices & Components | Composite self expanding stent device having a restraining element |
| US6179878B1 (en) | 1996-10-22 | 2001-01-30 | Thomas Duerig | Composite self expanding stent device having a restraining element |
| US5833651A (en) * | 1996-11-08 | 1998-11-10 | Medtronic, Inc. | Therapeutic intraluminal stents |
| US6315791B1 (en) | 1996-12-03 | 2001-11-13 | Atrium Medical Corporation | Self-expanding prothesis |
| US5776162A (en) | 1997-01-03 | 1998-07-07 | Nitinol Medical Technologies, Inc. | Vessel implantable shape memory appliance with superelastic hinged joint |
| US6413272B1 (en) | 1997-03-31 | 2002-07-02 | Kabushikikaisha Igaki Iryo Sekkei | Stent for vessel |
| US6632242B2 (en) | 1997-03-31 | 2003-10-14 | Kabushikikaisha Igaki Iryo Sekkei | Stent for vessel |
| US6200335B1 (en) | 1997-03-31 | 2001-03-13 | Kabushikikaisha Igaki Iryo Sekkei | Stent for vessel |
| US6056844A (en) | 1997-06-06 | 2000-05-02 | Triton Systems, Inc. | Temperature-controlled induction heating of polymeric materials |
| US6395038B1 (en) | 1997-08-19 | 2002-05-28 | Intermedics Inc. | Apparatus for imparting physician-determined shapes to implantable tubular devices |
| US6024764A (en) | 1997-08-19 | 2000-02-15 | Intermedics, Inc. | Apparatus for imparting physician-determined shapes to implantable tubular devices |
| US6241691B1 (en) | 1997-12-05 | 2001-06-05 | Micrus Corporation | Coated superelastic stent |
| US6720402B2 (en) | 1998-02-23 | 2004-04-13 | Mnemoscience Gmbh | Shape memory polymers |
| US6388043B1 (en) | 1998-02-23 | 2002-05-14 | Mnemoscience Gmbh | Shape memory polymers |
| US6160084A (en) | 1998-02-23 | 2000-12-12 | Massachusetts Institute Of Technology | Biodegradable shape memory polymers |
| US6156842A (en) | 1998-03-11 | 2000-12-05 | The Dow Chemical Company | Structures and fabricated articles having shape memory made from α-olefin/vinyl or vinylidene aromatic and/or hindered aliphatic vinyl or vinylidene interpolymers |
| US6033413A (en) | 1998-04-20 | 2000-03-07 | Endocare, Inc. | Stent delivery system |
| US6217609B1 (en) | 1998-06-30 | 2001-04-17 | Schneider (Usa) Inc | Implantable endoprosthesis with patterned terminated ends and methods for making same |
| US20020198584A1 (en) | 1998-08-21 | 2002-12-26 | Unsworth John Duncan | Shape memory tubular stent |
| US6500204B1 (en) | 1998-09-08 | 2002-12-31 | Kabushikikaisha Igaki Iryo Sekkei | Stent for vessels |
| US7335226B2 (en) | 1998-09-08 | 2008-02-26 | Kabushikikaisha Igaki Iryo Sekkei | Yarn for vessel stent |
| US7331988B2 (en) | 1998-09-08 | 2008-02-19 | Kabushikikaisha Igaki Iryo Sekkei | Stent for vessel |
| US7066952B2 (en) | 1998-09-08 | 2006-06-27 | Kabushikikaisha Igaki Iryo Sekkei | Method for manufacturing yarn for vessel stent |
| US6517569B2 (en) | 1998-09-14 | 2003-02-11 | Endocare, Inc. | Insertion device for stents and methods for use |
| US5997563A (en) | 1998-09-28 | 1999-12-07 | Medtronic, Inc. | Implantable stent having variable diameter |
| JP2000119465A (en) | 1998-10-13 | 2000-04-25 | Tokai Rubber Ind Ltd | Shape-memory resin composition and shape-memory active made thereof |
| US20020055787A1 (en) | 1998-10-15 | 2002-05-09 | Charles D. Lennox | Treating urinary retention |
| US6413273B1 (en) | 1998-11-25 | 2002-07-02 | Israel Aircraft Industries Ltd. | Method and system for temporarily supporting a tubular organ |
| US6183248B1 (en) | 1998-11-30 | 2001-02-06 | Muhammad Chishti | System and method for releasing tooth positioning appliances |
| US20020015519A1 (en) | 1998-12-11 | 2002-02-07 | Lord Corporation | Fiber substrate adhesion and coatings by contact metathesis polymerization |
| US6478773B1 (en) | 1998-12-21 | 2002-11-12 | Micrus Corporation | Apparatus for deployment of micro-coil using a catheter |
| EP1016424A1 (en) | 1998-12-31 | 2000-07-05 | Ethicon, Inc. | Radiopaque polymer coating |
| US6530950B1 (en) | 1999-01-12 | 2003-03-11 | Quanam Medical Corporation | Intraluminal stent having coaxial polymer member |
| US6538089B1 (en) | 1999-02-05 | 2003-03-25 | Forskarpatent I Syd Ab | Gels with a shape memory |
| JP2000319423A (en) | 1999-05-12 | 2000-11-21 | Showa Highpolymer Co Ltd | Flexible material |
| WO2000071554A2 (en) | 1999-05-24 | 2000-11-30 | California Institute Of Technology | Imidazolidine-based metal carbene metathesis catalysts |
| US20010029398A1 (en) | 1999-06-03 | 2001-10-11 | Jadhav Balkrishna S. | Bioresorbable stent |
| US6368346B1 (en) | 1999-06-03 | 2002-04-09 | American Medical Systems, Inc. | Bioresorbable stent |
| JP3068610B1 (en) | 1999-06-10 | 2000-07-24 | 多摩川精機株式会社 | Concentric shaft motor |
| JP3068611B1 (en) | 1999-06-15 | 2000-07-24 | 三菱重工業株式会社 | Ink supply device and method of mounting surface cover for preventing contamination on ink tray of ink supply device |
| WO2000078246A2 (en) | 1999-06-21 | 2000-12-28 | Endocare, Inc. | Stent insertion device |
| US6364904B1 (en) | 1999-07-02 | 2002-04-02 | Scimed Life Systems, Inc. | Helically formed stent/graft assembly |
| JP4100831B2 (en) | 1999-07-16 | 2008-06-11 | 大成建設株式会社 | Gap covering device for buildings with seismic isolation structure |
| US6858680B2 (en) | 1999-07-20 | 2005-02-22 | Aortech Biomaterials Pty Ltd | Shape memory polyurethane or polyurethane-urea polymers |
| WO2001007499A1 (en) | 1999-07-20 | 2001-02-01 | Aortech Biomaterials Pty Ltd. | Shape memory polyurethane or polyurethane-urea polymers |
| US6485507B1 (en) | 1999-07-28 | 2002-11-26 | Scimed Life Systems | Multi-property nitinol by heat treatment |
| US6287326B1 (en) | 1999-08-02 | 2001-09-11 | Alsius Corporation | Catheter with coiled multi-lumen heat transfer extension |
| US6086204A (en) | 1999-09-20 | 2000-07-11 | Magnante; Peter C. | Methods and devices to design and fabricate surfaces on contact lenses and on corneal tissue that correct the eye's optical aberrations |
| US7070615B1 (en) | 2000-03-13 | 2006-07-04 | Keiji Igaki | Linear material for blood vessel stent and blood vessel stent utilizing same |
| US20020007222A1 (en) | 2000-04-11 | 2002-01-17 | Ashvin Desai | Method and apparatus for supporting a body organ |
| WO2001080936A1 (en) | 2000-04-20 | 2001-11-01 | Kawasumi Laboratories, Inc. | Stent |
| US6679605B2 (en) | 2000-05-22 | 2004-01-20 | Medennium, Inc. | Crystalline polymeric compositions for ophthalmic devices |
| WO2001091822A1 (en) | 2000-05-31 | 2001-12-06 | Mnemoscience Gmbh | Shape memory thermoplastics and polymer networks for tissue engineering |
| US6406493B1 (en) | 2000-06-02 | 2002-06-18 | Hosheng Tu | Expandable annuloplasty ring and methods of use |
| WO2001093783A2 (en) | 2000-06-07 | 2001-12-13 | Advanced Cardiovascular Systems, Inc. | Variable stiffness stent |
| US7410498B2 (en) | 2000-06-29 | 2008-08-12 | Pentech Medical Devices Ltd. | Polymeric stents and other surgical articles |
| WO2002015951A2 (en) | 2000-08-23 | 2002-02-28 | Thoratec Corporation | Coated vascular grafts and methods of use |
| US20040015261A1 (en) | 2000-08-28 | 2004-01-22 | Hofmann Gregory J. | Shape memory polymer or alloy ophthalmic lens mold and methods of forming ophthalmic products |
| WO2002039875A2 (en) | 2000-11-17 | 2002-05-23 | B. Braun Medical Inc. | Method for deploying a thermo-mechanically expandable stent |
| US7498042B2 (en) | 2000-11-30 | 2009-03-03 | Kyoto Medical Planning Co., Ltd. | Stent for blood vessel and material for stent for blood vessel |
| US20020137864A1 (en) | 2001-01-24 | 2002-09-26 | Tong Tat Hung | Shape memory styrene copolymer |
| US20030033001A1 (en) | 2001-02-27 | 2003-02-13 | Keiji Igaki | Stent holding member and stent feeding system |
| WO2002083786A1 (en) | 2001-04-11 | 2002-10-24 | Cheong Seok Hong | Shape memory rubber composition |
| US20030173702A1 (en) | 2001-04-18 | 2003-09-18 | Keiji Igaki | Melting and spinning device and melting and spinning method |
| US6837901B2 (en) | 2001-04-27 | 2005-01-04 | Intek Technology L.L.C. | Methods for delivering, repositioning and/or retrieving self-expanding stents |
| US20030060530A1 (en) | 2001-07-24 | 2003-03-27 | Topolkaraev Vasily A. | Methods of making humidity activated materials having shape-memory |
| US20030060793A1 (en) | 2001-07-24 | 2003-03-27 | Topolkaraev Vasily A. | Disposable products having humidity activated materials with shape-memory |
| WO2003015840A3 (en) | 2001-08-17 | 2003-12-04 | Advanced Bio Prosthetic Surfac | Embolic protection device |
| US20030040803A1 (en) | 2001-08-23 | 2003-02-27 | Rioux Robert F. | Maintaining an open passageway through a body lumen |
| WO2003035743A1 (en) | 2001-10-25 | 2003-05-01 | Ticona Gmbh | Method for reducing the toughness of shaped plastic parts to be mechanically worked and the use thereof |
| US20030114777A1 (en) * | 2001-12-18 | 2003-06-19 | Scimed Life Systems, Inc. | Super elastic guidewire with shape retention tip |
| US7670302B2 (en) | 2001-12-18 | 2010-03-02 | Boston Scientific Scimed, Inc. | Super elastic guidewire with shape retention tip |
| US20040199242A1 (en) * | 2001-12-27 | 2004-10-07 | James Hong | Hybrid intravascular stent |
| US7037288B2 (en) | 2002-01-14 | 2006-05-02 | Codman & Shurtleff, Inc. | Anti-block catheter |
| US7473273B2 (en) | 2002-01-22 | 2009-01-06 | Medtronic Vascular, Inc. | Stent assembly with therapeutic agent exterior banding |
| US20030147046A1 (en) | 2002-02-06 | 2003-08-07 | Shadduck John H. | Adaptive optic lens system and method of use |
| US20030191276A1 (en) | 2002-02-26 | 2003-10-09 | Mnemoscience Gmbh | Polymeric networks |
| WO2003084491A1 (en) | 2002-04-10 | 2003-10-16 | Mnemoscience Gmbh | Method for treating hair with form memory polymers |
| WO2003084490A1 (en) | 2002-04-10 | 2003-10-16 | Mnemoscience Gmbh | Method for the generation of memory effects on hair |
| WO2003088818A2 (en) | 2002-04-18 | 2003-10-30 | Mnemoscience Gmbh | Biodegradable shape memory polymeric sutures |
| US20040015187A1 (en) | 2002-04-18 | 2004-01-22 | Mnemoscience Corporation | Biodegradable shape memory polymeric sutures |
| US20040014929A1 (en) | 2002-04-18 | 2004-01-22 | Mnemoscience Gmbh | Polyester urethanes |
| US6852825B2 (en) | 2002-04-18 | 2005-02-08 | Mnemoscience Gmbh | Polyester urethanes |
| US20040024143A1 (en) | 2002-04-18 | 2004-02-05 | Mnemoscience Gmbh | Interpenetrating networks |
| US20040030062A1 (en) | 2002-05-02 | 2004-02-12 | Mather Patrick T. | Castable shape memory polymers |
| DE10228120A1 (en) | 2002-06-24 | 2004-01-22 | Mnemoscience Gmbh | Permanent deforation of hair comprises applying a shape-memory composition to the hair before, during or after imparting a predetermined shape to the hair and then fixing the shape |
| WO2004006885A2 (en) | 2002-07-10 | 2004-01-22 | Mnemoscience Gmbh | Systems for releasing active ingredients, based on biodegradable or biocompatible polymers with a shape memory effect |
| US7367990B2 (en) | 2002-09-25 | 2008-05-06 | Kabushikikaisha Igaki Iryo Sekkei | Thread for vascular stent and vascular stent using the thread |
| WO2004033553A1 (en) | 2002-10-11 | 2004-04-22 | University Of Connecticut | Crosslinked polycyclooctene |
| US20040116641A1 (en) | 2002-10-11 | 2004-06-17 | Mather Patrick T. | Shape memory polymers based on semicrystalline thermoplastic polyurethanes bearing nanostructured hard segments |
| US20050245719A1 (en) | 2002-10-11 | 2005-11-03 | Mather Patrick T | Shape memory polymers based on semicrystalline thermoplastic polyurethanes bearing nanostructured hard segments |
| US20050251249A1 (en) | 2002-10-11 | 2005-11-10 | Sahatjian Ronald A | Endoprostheses |
| US20040122184A1 (en) | 2002-10-11 | 2004-06-24 | Mather Patrick T. | Crosslinked polycyclooctene |
| WO2004032799A2 (en) | 2002-10-11 | 2004-04-22 | Boston Scientific Limited | Implantable medical devices |
| WO2004033515A2 (en) | 2002-10-11 | 2004-04-22 | University Of Connecticut | Shape memory polymers based on semicrystalline thermoplastic polyurethanes bearing nanostructured hard segments |
| US7091297B2 (en) | 2002-10-11 | 2006-08-15 | The University Of Connecticut | Shape memory polymers based on semicrystalline thermoplastic polyurethanes bearing nanostructured hard segments |
| US20050010275A1 (en) | 2002-10-11 | 2005-01-13 | Sahatjian Ronald A. | Implantable medical devices |
| WO2004033539A1 (en) | 2002-10-11 | 2004-04-22 | University Of Connecticut | Blends of amorphous and semicrystalline polymers having shape memory properties |
| US20040153025A1 (en) | 2003-02-03 | 2004-08-05 | Seifert Paul S. | Systems and methods of de-endothelialization |
| WO2004073690A1 (en) | 2003-02-19 | 2004-09-02 | Mnemoscience Gmbh | Self-expanding device for the gastrointestinal or urogenital area |
| WO2004090042A1 (en) | 2003-04-10 | 2004-10-21 | Mnemoscience Gmbh | Blends with shape memory characteristics |
| US20060235501A1 (en) | 2003-05-23 | 2006-10-19 | Keiji Igaki | Stent supplying device |
| WO2004110515A1 (en) | 2003-06-13 | 2004-12-23 | Mnemoscience Gmbh | Biodegradable stents |
| WO2005009523A1 (en) | 2003-07-18 | 2005-02-03 | Boston Scientific Limited | Medical devices |
| US20070260302A1 (en) | 2003-10-15 | 2007-11-08 | Kabushikikaisha Igaki Iryo Sekkei | Device for delivery of stent for vessel |
| WO2005070988A1 (en) | 2004-01-20 | 2005-08-04 | Ucl Biomedica Plc | Polymer for use in conduits, medical devices and biomedical surface modification |
| US8070793B2 (en) | 2004-11-12 | 2011-12-06 | Kabushikikaisha Igaki Iryo Sekkei | Stent for vessel |
Non-Patent Citations (72)
| Title |
|---|
| "Cross-link" definition, Wikipedia (http://en.wikipedia.org/wiki/Cross-link), 6 pages, Nov. 20, 2013. |
| "Hydroxyl Terminated Polybutadiene Resins and Derivtives-Poly bd® and Krasol®", Sartomer Product Bulletin, Sep. 2004. |
| "Hydroxyl Terminated Polybutadiene Resins and Derivtives—Poly bd® and Krasol®", Sartomer Product Bulletin, Sep. 2004. |
| "POSS® (Polyhedral Oligomeric Silsesquioxane) Molecules from READE," READE-Your Higher Technology Specialty Chemicals Resource, READ SuperSite Search, (http://www.reade.com/products/37-polymeric-powders/646-polyhedral-oligomeric-silsequioxane-molecular-silica-poss-molecules-poss-molecular-silicas-poss-chemicals-poss-monomers-poss-silanols-heatabrasion-resistant-paints-and-coatings-mechanical-property-m), published Feb. 1, 2001, 3 pages. |
| "POSS® (Polyhedral Oligomeric Silsesquioxane) Molecules from READE," READE—Your Higher Technology Specialty Chemicals Resource, READ SuperSite Search, (http://www.reade.com/products/37-polymeric-powders/646-polyhedral-oligomeric-silsequioxane-molecular-silica-poss-molecules-poss-molecular-silicas-poss-chemicals-poss-monomers-poss-silanols-heatabrasion-resistant-paints-and-coatings-mechanical-property-m), published Feb. 1, 2001, 3 pages. |
| "Silsesquioxanes, Bridging the Gap Between Polymers & Ceramics" ChemFiles vol. 1, No. 6, 2001. |
| "Suite of Shape-Memory Polymers", Chemical & Engineering, Feb. 5, 2001. |
| Bassi et al., "The Monoclinic Structure of Even Trans-Polyalkenamers", European Polymer Journal, vol. 3, pp. 339-352, 1967. |
| Bassi et al., "The Triclinic Structure of trans-Polyoctenamer", European Polymer Journal, vol. 4, pp. 123-132, 1968. |
| Bielawski et al., "Highly Efficient Ring-Opening Metathesis Polymerization (ROMP) Using New Ruthenium Catalysts Containing N-Heterocyclic Carbene Ligands", Angew. Chem. Int. Ed., 39, No. 16, pp. 2903-2906, 2000. |
| Bielawski et al., "Highly efficient syntheses of acetoxy- and hyroxy-terminated telechelic poly(butadiene)s using ruthenium catalysts containing N-heterocyclic ligands", Polymer, 42 , pp. 4939-4945, 2001. |
| Boochathum et al., "Vulcanization of Cis- and Trans-Polyisoprene and Their Blends: Cure Characteristics and Crosslink Distribution", European Polymer Journal 37, pp. 417-427, 2001. |
| Brochure, Degussa High Performance Polymers, The Rubber with Unique Properties, Vestenamer ©, Undated. |
| Calderon et al., "Melting Temperature of trans-Polyoctenamer", Journal of Polymer Science: Part A-2, vol. 5, pp. 1283-1292, 1967. |
| Chun et al., "Enhanced Dynamic Mechanical and Shape-Memory Properties of a Poly(ethylene terephthalate)-Poly(ethylene glycol) Copolymer Crosslinked by Maleic Anhydride", Journal of Applied Polymer Science, vol. 83, pp. 27-37, 2002. |
| Dinger, et al., "High Turnover Numbers with Ruthenium-Based Metathesis Catalysts" Advanced Synthesis Catalysis, vol. 344, pp. 671-677, 2002. |
| Du Prez et al., "Segmented Networks by Cationic Polymerization: Design and Applications" NATO Sci. Ser., Ser. E, pp. 75-98, 1999. |
| Fan et al., "Synthesis and Properties of Polyurethane Modified with Aminoethylaminopropyl Poly(Dimethyl siloxane)", Journal of Applied Polymer Science, vol. 74, pp. 2552-2558, 1999. |
| Fu et al., "Nanoscale Reinforcement of Polyhedral Oligomeric Silsesquioxane (POSS) in Polyurethane Elastomer", Polymer Int. 49, pp. 437-440, 2000. |
| Fu et al., "Structural Development During Deformation of Polyurethane Containing Polyhedral Oligomeric Silsesquioxanes (POSS) Molecules", Polymer 42, pp. 599-611, 2001. |
| Gajria et al., "Miscibility and Biodegradability of Blends of Poly(Lactic Acid) and Poly(Vinyl Acetate)", Polymer, vol. 37, pp. 437-444, 1996. |
| Ge et al., "Synthesis of Thermoplastic Polyurethanes Bearing Nanostructured Hard Segments: New Shape Memory Polymers" Polymer Program, Institute of Materials Science and Department of Engineering, UCONN, (Abstract, 2 pp.), Jul. 2003. |
| Goethals et al. "Poly(Vinyl Ethers) as Building Blocks for New Materials" Macromol. Symp., 132, pp. 57-64, 1998. |
| Gordon, "Applications of Shape Memory Polyurethanes", Proceedings of the First International Conference on Shape Memory and Superelastic Technologies, SMST International Committee, pp. 115-199, 1994. |
| Gupta et al., "Effect of solvent exposure on the Properties of hyroxy-terminated polybutadiene-based polyurethanes", Polym. Int. 52, pp. 938-948, 2003. |
| Irie, Shape Memory Polymers, Cambridge University Press: Cambridge, UK, pp. 203-219, 1998. |
| Ishii, "Shape Memory Resins", Trans-polyisoprene-based Shape Memory Resins, Zairyo Gijutsu, 7(6), Abstract only, 1 page, 1989. |
| Jeon et al., "Shape Memory and Nanostructure in Poly(norbornyl-POSS) Copolymers", Polymer International, 49, pp. 453-457, 2000. |
| Jeong et al., "Miscibility and Shape Memory Effect of Thermoplastic Polyurethane Blends with Phenoxy Resin", European Polymer Journal , 37, pp. 2245-2252, 2001. |
| Jeong et al., "Miscibility and Shape Memory Property of Poly(vinyl chloride)/Thermoplastic Polyurethane Blends", Journal of Materials Science, 36, pp. 5457-5463, 2001. |
| Kagami et al., "Shape Memory Behaviors of Crosslinked Copolymers Containing Stearyl Acrylate" Macromol. Rapid. Commun., 17(8), pp. 539-543, 1996. |
| Kaneko et al., "Shape Memory Gels with Multi-Stimuli-Responses", Proc. SPIE-Int. Soc. Opt. Eng., 3669, pp. 199-208, 1999. |
| Kannan et al., "Polyhedral Oligomeric Silisesquixoane Nanocomposites: The Next Generation Material for Biomedical Applications", Acc. Chem. Res., vol. 38, No. 11, pp. 879-884, 2005. |
| Kim et al., "Polyurethane Ionomers Having Shape Memory Effects", Polymer , vol. 39, No. 13, pp. 2803-2808, 1998. |
| Lendlein et al., "AB-Polymer Networks Based on Oligo(epsilon-caprolactone) Segments Showing Shape-Memory Properties" Proc. Natl. Acad. Sci., USA, 98(3), pp. 842-847, 2001. |
| Lendlein et al., "AB-Polymer Networks Based on Oligo(ε-caprolactone) Segments Showing Shape-Memory Properties" Proc. Natl. Acad. Sci., USA, 98(3), pp. 842-847, 2001. |
| Lendlein et al., "Biodegradable, Elastic Shape-Memory Polymers for Potential Biomedical Applications", Science, vol. 296, pp. 1673-1676, May 31, 2002. |
| Lendlein et al., "Shape-Memory Polymers", Angew. Chem. Int. Ed. 41, pp. 2034-2057, 2002. |
| Lin et al., "Study on Shape-Memory Behavior of Polyether-Based Polyurethanes. I. Influence of the Hard-Segment Content", Journal of Applied Polymer Science, vol. 69, pp. 1563-1574, 1998. |
| Lin et al., "Study on Shape-Memory Behavior of Polyether-Based Polyurethanes. II. Influence of Soft-Segment Molecular Weight", Journal of Applied Polymer Science, vol. 69, pp. 1575-1586, 1998. |
| Liu et al., "Shape Memory of Hydrogen-Bonded Polymer Network/Poly(ethylene glycol) Complexes", Chengdu Institute of Organic Chemistry, Chinese Academy of Sciences, 2003. |
| Liu et al., "Themomechanical Characterization of a Novel Series of Shape Memory Polymers", SPE ANTEC Proceedings, 5 pages, 2002. |
| Liu, et al., "Chemically Cross-Linked Polycylooctene: Synthesis, Characterization and Shape Memory Behavior" Macromolecules, 35, pp. 9868-9874, 2002. |
| Mather et al., "Strain Recovery in Drawn POSS Hybrid Thermoplastics," XIIIth International Congress on Rheology, Cambridge, UK, 4, pp. 53-55, 2000. |
| Mather et al., "Strain Recovery in POSS Hybrid Thermoplastics," Polymer Preprints 41(1), pp. 528-529, 2000. |
| Mauler et al., "Functional Group Determination in Hydroxilated Polymer", Eur. Polym. J., vol. 31, No. 1, pp. 51-55, 1995. |
| Mauler et al., "Liquid-crystalline polyacrylate crosslinked with alpha, omega polyisoprene diacrylate segments", Polymer Bulletin, 41, pp. 291-297, 1998. |
| Mauler et al., "Liquid-crystalline polyacrylate crosslinked with α, ω polyisoprene diacrylate segments", Polymer Bulletin, 41, pp. 291-297, 1998. |
| Nakayama, K., "Properties and Applications of Shape-Memory Polymers", International Polymer Science and Technology, 18, T/43-48, 1991. |
| Oh et al., "Dynamic Mechanical Properties of Carbon Black Filled Trans-polyoctenamer Vulcanizates", Abstract Only, (Oct. 19, 1985). |
| Paul Starck, "Dynamic Mechanical Thermal Analysis on Ziegler-Natta and Metallocene Type Ethylene Copolymers", Eur. Poly. J. vol. 33, No. 3, pp. 339-348, 1997. |
| Ramanathan et al., "Polyurethane elastomers", Polymer Data Handbook, pp. 874-877, 1999. |
| Ramanathan et al., "Polyurethane urea", Polymer Data Handbook, pp. 878-881, 1999. |
| Ramanathan et al., "Polyurethane", Polymer Data Handbook, pp. 870-873, 1999. |
| Reyntjens et al., "Polymer Networks Containing Crystallizable Poly(octadecyl vinyl ether) Segments for Shape-Memory Materials", Macromol. Rapid. Commun., 20(5), pp. 251-255, 1999. |
| Rousseau et al., "Shape Memory Effect Exhibited by Smectic-C Liquid Crystalline Elastomers" J. Am. Chem. Soc., 125, pp. 15300-15301, 2003. |
| Sarbu et al., "Synthesis of Hydroxy-Telechelic Poly(methyl acrylate) and Polystyrene by Atom Transfer Radical Coupling", Macromolecules, 37, pp. 9694-9700, 2004. |
| Schneider et al., "Crystallinity of trans-Polyoctenamer: Characterization and Influence of Sample History", Journal of Molecular Catalysis, 46, pp. 395-403, 1988. |
| Schwab et al., "Hybrid Nanoreinforced Polyurethanes Based on Polyhedral Oligomeric Silsesquioxanes(Poss)", ACS Polymeric Materials Science and Engineering, Fall Meeting, 1997, vol. 77, pp. 549-550. |
| Schwab et al., "Synthesis and Applications of RuC12(=CHR')(PR3)2: The Influence of the Alkylidene Moiety on Metathesis Activity", J. Am. Chem. Soc., 118, pp. 100-110, 1996. |
| Schwabb et al., "Polyhedral Oligomeric Sil silsesquioxanes (POSS): Silicon Based Monomers and Their Use in the Preparation of Hybrid Polyurethanes", Mat. Res. Soc. Symp. Proc., vol. 519, pp. 21-27, 1998. |
| Sung et al., "Properties of Segmented Poly(urethaneureas) Based on 2,4-Toluene Diisocyanate. 1. Thermal Transitions, X-ray Studies, and Comparison with Segmented Poly(urethanes)", Macromolecules, 13, pp. 111-116, 1980. |
| Valimaa et al., "Viscoelastic memory and self-expansion of self-reinforced bioabsorbable stents", Biomaterials, 23, pp. 3575-3582, 2002. |
| Van Humbeeck, "Shape Memory Alloys: A Material and a Technology", Advanced Engineering Materials, vol. 3, No. 11, pp. 837-850, 2001. |
| Wache et al., "Development of a polymer stent with shape memory effect as a drug delivery system", Journal of Materials Science: Materials in Medicine, 14, 109-112, (2003). |
| Wei et al., "Shape-Memory Materials and Hybrid Composites for Smart Systems", Journal of Materials Science 33, pp. 3743-3762, 1998. |
| Woojin Lee, "Polymer Gel Based Actuator: Dynamic model of gel for real time control", Massachuetts Institute of Technology, Department of Mechanical Engineering, May 3, 1996. |
| Yeh et al., "Radiation-Induced Crosslinking: Effect on Structure of Polyethylene", Colloid & Polymer Sci. 263, pp. 109-115, 1985. |
| Yoshida et al., "Development and Application of Shape-Memory Polymer Gel (Part 1)-Synthesis and Processing of Shape-Memory Polymer Gel", Hokkaidoritsu Kogyo Shikenjo Hokoku, 298 Abstract Only, 1 page, 1999. |
| Yoshida et al., "Development and Application of Shape-Memory Polymer Gel (Part 1)—Synthesis and Processing of Shape-Memory Polymer Gel", Hokkaidoritsu Kogyo Shikenjo Hokoku, 298 Abstract Only, 1 page, 1999. |
| Zhu et al., "Shape-Memory Effects of Radiation Crosslinked Poly(epsilon-caprolactone)", Journal of Applied Polymer Science, vol. 90, pp. 1589-1595, 2003. |
| Zhu et al., "Shape-Memory Effects of Radiation Crosslinked Poly(ε-caprolactone)", Journal of Applied Polymer Science, vol. 90, pp. 1589-1595, 2003. |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20150114143A1 (en) * | 2013-10-24 | 2015-04-30 | Spirit Aerosystems, Inc. | Remoldable contour sensor holder |
| US9646599B2 (en) * | 2013-10-24 | 2017-05-09 | Spirit Aerosystems, Inc. | Remoldable contour sensor holder |
| US11096774B2 (en) | 2016-12-09 | 2021-08-24 | Zenflow, Inc. | Systems, devices, and methods for the accurate deployment of an implant in the prostatic urethra |
| US11903859B1 (en) | 2016-12-09 | 2024-02-20 | Zenflow, Inc. | Methods for deployment of an implant |
| US11890213B2 (en) | 2019-11-19 | 2024-02-06 | Zenflow, Inc. | Systems, devices, and methods for the accurate deployment and imaging of an implant in the prostatic urethra |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1824412A2 (en) | 2007-08-29 |
| US9115245B2 (en) | 2015-08-25 |
| US20050216074A1 (en) | 2005-09-29 |
| AU2005294569A1 (en) | 2006-04-20 |
| WO2006041767A3 (en) | 2007-01-25 |
| US20120253451A1 (en) | 2012-10-04 |
| US20100331954A1 (en) | 2010-12-30 |
| WO2006041767A2 (en) | 2006-04-20 |
| US20140135889A9 (en) | 2014-05-15 |
| US7794494B2 (en) | 2010-09-14 |
| US20110054591A1 (en) | 2011-03-03 |
| US20140067039A9 (en) | 2014-03-06 |
| CA2583191A1 (en) | 2006-04-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8784465B2 (en) | Implantable medical devices | |
| AU2010200568B2 (en) | Implantable medical devices | |
| US7919162B2 (en) | Intraluminal prostheses having polymeric material with selectively modified crystallinity and methods of making same | |
| CA2498180C (en) | Carbon dioxide-assisted methods of providing biocompatible intraluminal prostheses | |
| CA2591276A1 (en) | Implantable endoprosthesis and methods of delivering | |
| KR20190028419A (en) | Medical equipment and plasticized nylon materials |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: BOSTON SCIENTIFIC SCIMED, INC., MINNESOTA Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:SAHATJIAN, RONALD A.;TAN, FRANCISCA;REEL/FRAME:030044/0195 Effective date: 20041101 Owner name: UNIVERSITY OF CONNECTICUT, CONNECTICUT Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:MATHER, PATRICK T.;LIU, CHANGDENG;CAMPO, CHERYL J.;SIGNING DATES FROM 20050603 TO 20050615;REEL/FRAME:030044/0259 |
|
| AS | Assignment |
Owner name: SCIMED LIFE SYSTEMS, INC., MINNESOTA Free format text: CORRECTIVE ASSIGNMENT TO CORRECT THE NAME OF THE ASSIGNEE PREVIOUSLY RECORDED ON REEL 030044 FRAME 0195. ASSIGNOR(S) HEREBY CONFIRMS THE THE NAME OF THE ASSIGNEE IS "SCIMED LIFE SYSTEMS, INC.";ASSIGNORS:SAHATJIAN, RONALD A.;TAN, FRANCISCA;REEL/FRAME:030091/0875 Effective date: 20041101 |
|
| AS | Assignment |
Owner name: BOSTON SCIENTIFIC SCIMED, INC., MINNESOTA Free format text: CHANGE OF NAME;ASSIGNOR:SCIMED LIFE SYSTEMS, INC.;REEL/FRAME:030143/0399 Effective date: 20041222 |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 4TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1551) Year of fee payment: 4 |
|
| FEPP | Fee payment procedure |
Free format text: MAINTENANCE FEE REMINDER MAILED (ORIGINAL EVENT CODE: REM.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| LAPS | Lapse for failure to pay maintenance fees |
Free format text: PATENT EXPIRED FOR FAILURE TO PAY MAINTENANCE FEES (ORIGINAL EVENT CODE: EXP.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| STCH | Information on status: patent discontinuation |
Free format text: PATENT EXPIRED DUE TO NONPAYMENT OF MAINTENANCE FEES UNDER 37 CFR 1.362 |
|
| FP | Lapsed due to failure to pay maintenance fee |
Effective date: 20220722 |