1. A device, comprising a sensor and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
2. The device of claim 1 wherein the agent inhibits cell regeneration.
3. The device of claim 1 wherein the agent inhibits angiogenesis.
4. The device of claim 1 wherein the agent inhibits fibroblast migration.
5. The device of claim 1 wherein the agent inhibits fibroblast proliferation.
6. The device of claim 1 wherein the agent inhibits deposition of extracellular matrix.
7. The device of claim 1 wherein the agent inhibits tissue remodeling.
8. The device of claim f wherein the agent is an angiogenesis inhibitor.
9. The device of claim 1 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
10. The device of claim 1 wherein the agent is a chemokine receptor antagonist.
11. The device of claim 1 wherein the agent is a cell cycle inhibitor.
12. The device of claim 1 wherein the agent is a taxane.
13. The device of claim 1 wherein the agent is an antimicrotubule agent.
14. The device of claim 1 wherein the agent is paclitaxel.
15. The device of claim 1 wherein the agent is not paclitaxel.
16. The device of claim 1 wherein the agent is an analogue or derivative of paclitaxel.
17. The device of claim 1 wherein the agent is a vinca alkaloid.
18. The device of claim 1 wherein the agent is camptothecin or an analogue or derivative thereof.
19. The device of claim 1 wherein the agent is a podophyllotoxin.
20. The device of claim 1 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
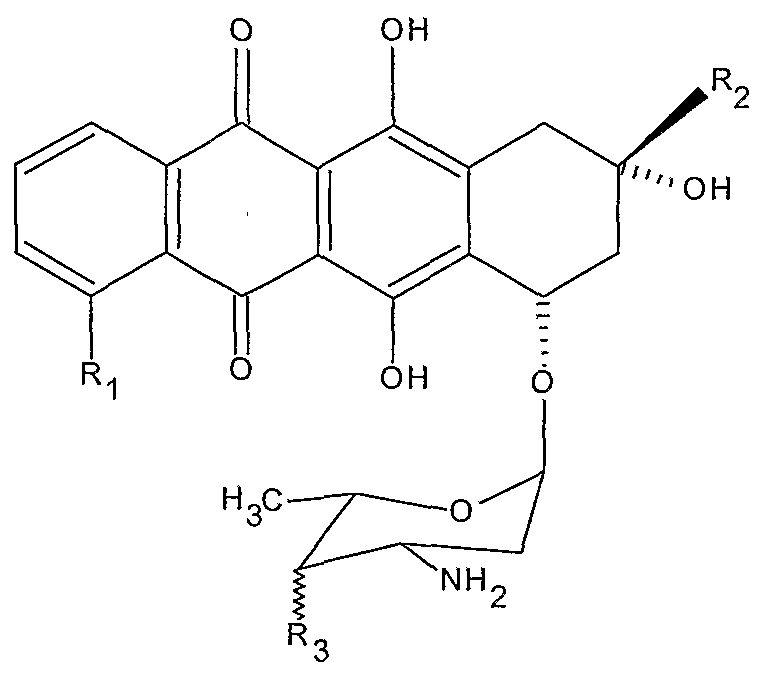
21. The device of claim 1 wherein the agent is an anthracycline.
22. The device of claim 1 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
23. The device of claim 1 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
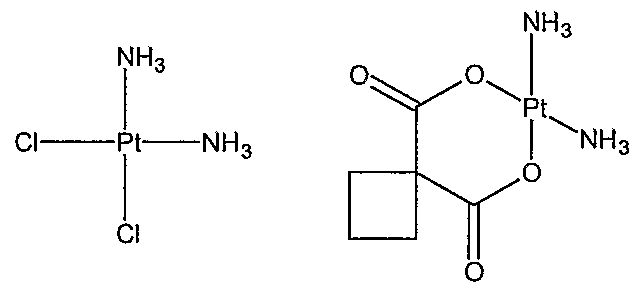
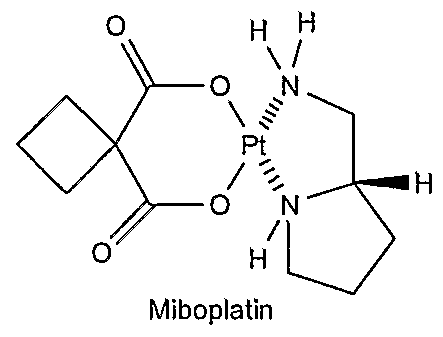
24. The device of claim 1 wherein the agent is a platinum compound.
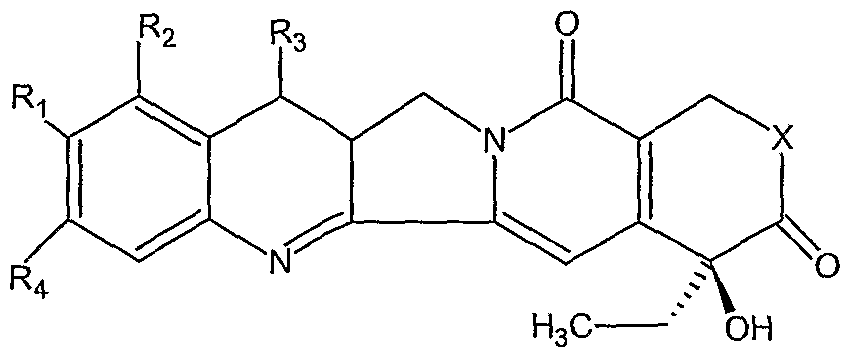
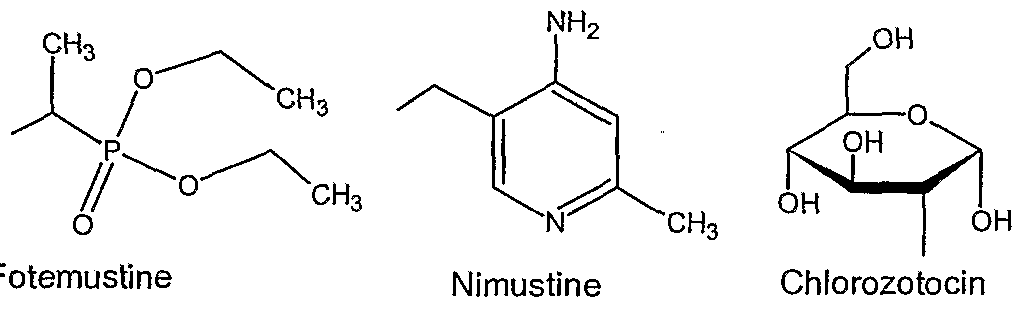
25. The device of claim 1 wherein the agent is a nitrosourea.
26. The device of claim 1 wherein the agent is a nitroimidazole.
27. The device of claim 1 wherein the agent is a folic acid antagonist.
28. The device of claim 1 wherein the agent is a cytidine analogue.
29. The device of claim 1 wherein the agent is a pyrimidine analogue.
30. The device of claim 1 wherein the agent is a fluoropyrimidine analogue.
31. The device of claim 1 wherein the agent is a purine analogue.
32. The device of claim 1 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
33. The device of claim 1 wherein the agent is a hydroxyurea.
34. The device of claim 1 wherein the agent is a mytomicin or an analogue or derivative thereof.
35. The device of claim 1 wherein the agent is an alkyl sulfonate.
36. The device of claim 1 wherein the agent is a benzamide or an analogue or derivative thereof.
37. The device of claim 1 wherein the agent is a nicotinamide or an analogue or derivative thereof.
38. The device of claim 1 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
39. The device of claim 1 wherein the agent is a DNA alkylating agent.
40. The device of claim 1 wherein the agent is an antimicrotubule agent.
41. The device of claim 1 wherein the agent is a topoisomerase inhibitor.
42. The device of claim 1 wherein the agent is a DNA cleaving agent.
43. The device of claim 1 wherein the agent is an antimetabolite.
44. The device of claim 1 wherein the agent inhibits adenosine deaminase.
45. The device of claim 1 wherein the agent inhibits purine ring synthesis.
46. The device of claim 1 wherein the agent is a nucleotide interconversion inhibitor.
47. The device of claim 1 wherein the agent inhibits dihydrofolate reduction.
48. The device of claim 1 wherein the agent blocks thymidine monophosphate.
49. The device of claim 1 wherein the agent causes DNA damage.
50. The device of claim 1 wherein the agent is a DNA intercalation agent.
51. The device of claim 1 wherein the agent is a RNA synthesis inhibitor.
52. The device of claim 1 wherein the agent is a pyrimidine synthesis inhibitor.
53. The device of claim 1 wherein the agent inhibits ribonucleotide synthesis or function.
54. The device of claim 1 wherein the agent inhibits thymidine monophosphate synthesis or function.
55. The device of claim 1 wherein the agent inhibits DNA synthesis.
56. The device of claim 1 wherein the agent causes DNA adduct formation.
57. The device of claim 1 wherein the agent inhibits protein synthesis.
58. The device of claim 1 wherein the agent inhibits microtubule function.
59. The device of claim 1 wherein the agent is a cyclin dependent protein kinase inhibitor.
60. The device of claim 1 wherein the agent is an epidermal growth factor kinase inhibitor.
61. The device of claim 1 wherein the agent is an elastase inhibitor.
62. The device of claim 1 wherein the agent is a factor Xa inhibitor.
63. The device of claim 1 wherein the agent is a farnesyltransferase inhibitor.
64. The device of claim 1 wherein the agent is a fibrinogen antagonist.
65. The device of claim 1 wherein the agent is a guanylate cyclase stimulant.
66. The device of claim 1 wherein the agent is a heat shock protein 90 antagonist.
67. The device of claim 1 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
68. The device of claim 1 wherein the agent is a guanylate cyclase stimulant.
69. The device of claim 1 wherein the agent is a HMGCoA reductase inhibitor.
70. The device of claim 1 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
71. The device of claim 1 wherein the agent is a hydroorotate dehydrogenase inhibitor.
72. The device of claim 1 wherein the agent is an IKK2 inhibitor.
73. The device of claim 1 wherein the agent is an IL-1 antagonist.
74. The device of claim 1 wherein the agent is an ICE antagonist.
75. The device of claim 1 wherein the agent is an IRAK antagonist.
76. The device of claim 1 wherein the agent is an IL-4 agonist.
77. The device of claim 1 wherein the agent is an immunomodulatory agent.
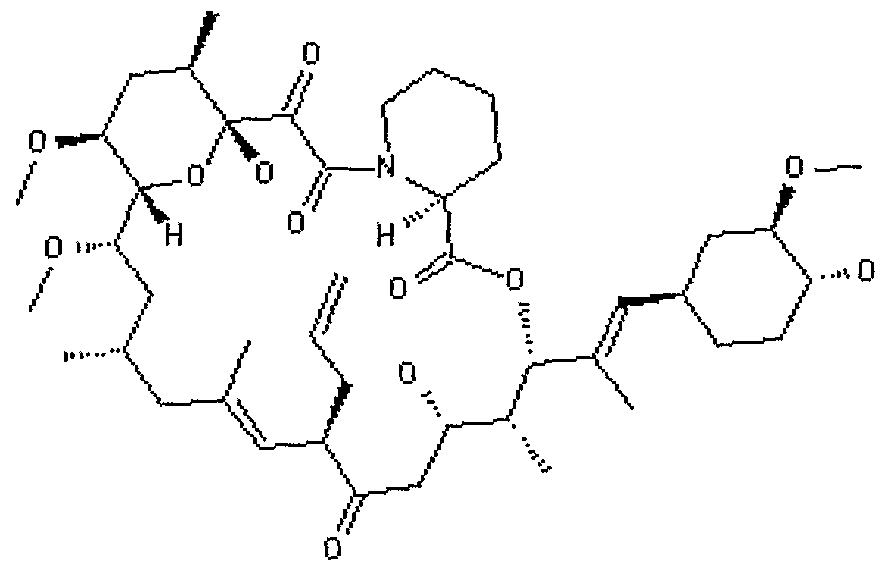
78. The device of claim 1 wherein the agent is sirolimus or an analogue or derivative thereof.
79. The device of claim 1 wherein the agent is not sirolimus.
80. The device of claim 1 wherein the agent is everolimus or an analogue or derivative thereof.
81. The device of claim 1 wherein the agent is tacrolimus or an analogue or derivative thereof.
82. The device of claim 1 wherein the agent is not tacrolimus.
83. The device of claim 1 wherein the agent is biolmus or an analogue or derivative thereof.
84. The device of claim 1 wherein the agent is tresperimus or an analogue or derivative thereof.
85. The device of claim 1 wherein the agent is auranofin or an analogue or derivative thereof.
86. The device of claim 1 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
87. The device of claim 1 wherein the agent is gusperimus or an analogue or derivative thereof.
88. The device of claim 1 wherein the agent is pimecrolimus or an analogue or derivative thereof.
89. The device of claim 1 wherein the agent is ABT-578 or an analogue or derivative thereof.
90. The device of claim 1 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
91. The device of claim 1 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
92. The device of claim 1 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
93. The device of claim 1 wherein the agent is a leukotriene inhibitor.
94. The device of claim 1 wherein the agent is a MCP-1 antagonist.
95. The device of claim 1 wherein the agent is a MMP inhibitor.
96. The device of claim 1 wherein the agent is an NF kappa B inhibitor.
97. The device of claim 1 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
98. The device of claim 1 wherein the agent is an NO antagonist.
99. The device of claim 1 wherein the agent is a p38 MAP kinase inhibitor.
100. The device of claim 1 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
101. The device of claim 1 wherein the agent is a phosphodiesterase inhibitor.
102. The device of claim 1 wherein the agent is a TGF beta inhibitor.
103. The device of claim 1 wherein the agent is a thromboxane A2 antagonist.
104. The device of claim 1 wherein the agent is a TNF alpha antagonist
105. The device of claim 1 wherein the agent is a TACE inhibitor.
106. The device of claim 1 wherein the agent is a tyrosine kinase inhibitor.
107. The device of claim 1 wherein the agent is a vitronectin inhibitor.
108. The device of claim 1 wherein the agent is a fibroblast growth factor inhibitor.
109. The device of claim 1 wherein the agent is a protein kinase inhibitor.
110. The device of claim 1 wherein the agent is a PDGF receptor kinase inhibitor.
111. The device of claim 1 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
112. The device of claim 1 wherein the agent is a retinoic acid receptor antagonist.
113. The device of claim 1 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
114. The device of claim 1 wherein the agent is a fibrinogen antagonist.
115. The device of claim 1 wherein the agent is an antimycotic agent.
116. The device of claim 1 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
117. The device of claim 1 wherein the agent is a bisphosphonate.
118. The device of claim 1 wherein the agent is a phospholipase A1 inhibitor.
119. The device of claim 1 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
120. The device of claim 1 wherein the agent is a macrolide antibiotic.
121. The device of claim 1 wherein the agent is a GPIIb/llla receptor antagonist.
122. The device of claim 1 wherein the agent is an endothelin receptor antagonist
123. The device of claim 1 wherein the agent is a peroxisome proliferator-activated receptor agonist.
124. The device of claim 1 wherein the agent is an estrogen receptor agent.
125. The device of claim 1 wherein the agent is a somastostatin analogue.
126. The device of claim 1 wherein the agent is a neurokinin 1 antagonist.
127. The device of claim 1 wherein the agent is a neurokinin 3 antagonist
128. The device of claim 1 wherein the agent is a VLA-4 antagonist.
129. The device of claim 1 wherein the agent is an osteoclast inhibitor.
130. The device of claim 1 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
131. The device of claim 1 wherein the agent is an angiotensin I converting enzyme inhibitor.
132. The device of claim 1 wherein the agent is an angiotensin II antagonist.
133. The device of claim 1 wherein the agent is an enkephalinase inhibitor.
134. The device of claim 1 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
135. The device of claim 1 wherein the agent is a protein kinase C inhibitor.
136. The device of claim 1 wherein the agent is a ROCK (rho- associated kinase) inhibitor.
137. The device of claim 1 wherein the agent is a CXCR3 inhibitor.
138. The device of claim 1 wherein the agent is an Itk inhibitor.
139. The device of claim 1 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
140. The device of claim 1 wherein the agent is a PPAR agonist.
141. The device of claim 1 wherein the agent is an immunosuppressant.
142. The device of claim 1 wherein the agent is an Erb inhibitor.
143. The device of claim 1 wherein the agent is an apoptosis agonist.
144. The device of claim 1 wherein the agent is a lipocortin agonist.
145. The device of claim 1 wherein the agent is a VCAM-1 antagonist.
146. The device of claim 1 wherein the agent is a collagen antagonist.
147. The device of claim 1 wherein the agent is an alpha 2 integrin antagonist.
148. The device of claim 1 wherein the agent is a TNF alpha inhibitor.
149. The device of claim 1 wherein the agent is a nitric oxide inhibitor
150. The device of claim 1 wherein the agent is a cathepsin inhibitor.
151. The device of claim 1 wherein the agent is not an anti- inflammatory agent.
152. The device of claim 1 wherein the agent is not a steroid.
153. The device of claim 1 wherein the agent is not a glucocorticosteroid.
154. The device of claim 1 wherein the agent is not dexamethasone.
155. The device of claim 1 wherein the agent is not an anti- infective agent.
156. The device of claim 1 wherein the agent is not an antibiotic.
157. The device of claim 1 wherein the agent is not an anti-fugal agent.
158. The device of claim 1 wherein the agent is not beclomethasone.
159. The device of claim 1 wherein the agent is not dipropionate.
160. The device of claim 1 , further comprising a coating, wherein the coating comprises the anti-scarring agent and a polymer.
161. The device of claim 1 , further comprising a coating, wherein the coating comprises the anti-scarring agent.
162. The device of claim 1 , further comprising a coating, wherein the coating is disposed on a surface of the device.
163. The device of claim 1 , further comprising a coating, wherein the coating directly contacts the device.
164. The device of claim 1 , further comprising a coating, wherein the coating indirectly contacts the device.
165. The device of claim 1 , further comprising a coating, wherein the coating partially covers the device.
166. The device of claim 1 , further comprising a coating, wherein the coating completely covers the device.
167. The device of claim 1 , further comprising a coating, wherein the coating is a uniform coating.
168. The device of claim 1 , further comprising a coating, wherein the coating is a non-uniform coating.
169. The device of claim 1 , further comprising a coating, wherein the coating is a discontinuous coating.
170. The device of claim 1 , further comprising a coating, wherein the coating is a patterned coating.
171. The device of claim 1 , further comprising a coating, wherein the coating has a thickness of 100 μm or less.
172. The device of claim 1 , further comprising a coating, wherein the coating has a thickness of 10 μm or less.
173. The device of claim 1 , further comprising a coating, wherein the coating adheres to the surface of the device upon deployment of the device.
174. The device of claim 1 , further comprising a coating, wherein the coating is stable at room temperature for a period of 1 year.
175. The device of claim 1 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1% by weight.
176. The device of claim 1 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
177. The device of claim 1 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
178. The device of claim 1 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight
179. The device of claim 1 , further comprising a coating, wherein the coating further comprises a polymer.
180. The device of claim 1 , further comprising a first coating having a first composition and the second coating having a second composition.
181. The device of claim 1 , further comprising a first coating having a first composition and the second coating having a second composition, wherein the first composition and the second composition are different.
182. The device of claim 1 , further comprising a polymer.
183. The device of claim 1 , further comprising a polymeric carrier.
184. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a copolymer.
185. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a block copolymer.
186. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a random copolymer.
187. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a biodegradable polymer.
188. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-biodegradable polymer.
189. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophilic polymer.
190. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophobic polymer.
191. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophilic domains.
192. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophobic domains.
193. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-conductive polymer.
194. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises an elastomer.
195. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrogel.
196. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a silicone polymer.
197. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrocarbon polymer.
198. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a styrene-derived polymer.
199. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a butadiene polymer.
200. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a macromer.
201. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a poly( ethylene glycol) polymer.
202. The device of claim 1 , further comprising a polymeric carrier, wherein the polymeric carrier comprises an amorphous polymer.
203. The device of claim 1 , further comprising a lubricious coating.
204. The device of claim 1 wherein the anti-scarring agent is located within pores or holes of the device.
205. The device of claim 1 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
206. The device of claim 1 , further comprising a second I pharmaceutically active agent.
207. The device of claim 1 , further comprising an anti- inflammatory agent.
208. The device of claim 1 , further comprising an agent that inhibits infection.
209. The device of claim 1 , further comprising an agent that inhibits infection, wherein the agent is an anthracycline.
210. The device of claim 1 , further comprising an agent that inhibits infection, wherein the agent is doxorubicin.
211. The device of claim 1 , further comprising an agent that inhibits infection, wherein the agent is mitoxantrone.
212. The device of claim 1 , further comprising an agent that inhibits infection, wherein the agent is a fluoropyrimidine.
213. The device of claim 1 , further comprising an agent that inhibits infection, wherein the agent is 5-fluorouracil (5-FU).
214. The device of claim 1 , further comprising an agent that inhibits infection, wherein the agent is a folic acid antagonist.
215. The device of claim 1 , further comprising an agent that inhibits infection, wherein the agent is methotrexate.
216. The device of claim 1 , further comprising an agent that inhibits infection, wherein the agent is a podophylotoxin.
217. The device of claim 1 , further comprising an agent that inhibits infection, wherein the agent is etoposide.
218. The device of claim 1 , further comprising an agent that inhibits infection, wherein the agent is a camptothecin.
219. The device of claim 1 , further comprising an agent that inhibits infection, wherein the agent is a hydroxyurea.
220. The device of claim 1 , further comprising an agent that inhibits infection, wherein the agent is a platinum complex.
221. The device of claim 1 , further comprising an agent that inhibits infection, wherein the agent is cisplatin.
222. The device of claim 1 , further comprising an anti-thrombotic agent.
223. The device of claim 1 , further comprising a visualization agent.
224. The device of claim 1 , further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
225. The device of claim 1 , further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises barium, tantalum, or technetium.
226. The device of claim 1 , further comprising a visualization agent, wherein the visualization agent is a MRI responsive material.
227. The device of claim 1 , further comprising a visualization agent, wherein the visualization agent comprises a gadolinium chelate.
228. The device of claim 1 , further comprising a visualization agent, wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
229. The device of claim 1 , further comprising a visualization agent, wherein the visualization agent comprises an iron oxide compound.
230. The device of claim 1 , further comprising a visualization agent, wherein the visualization agent comprises a dye, pigment, or colorant.
231. The device of claim 1 , further comprising an echogenic material.
232. The device of claim 1 , further comprising an echogenic material, wherein the echogenic material is in the form of a coating.
233. The device of claim 1 wherein the device is sterile.
234. The device of claim 1 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
235. The device of claim 1 wherein the device is adapted for delivering the anti-scarring agent locally to tissue proximate to the device.
236. The device of claim 1 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
237. The device of claim 1 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is connective tissue.
238. The device of claim 1 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is muscle tissue.
239. The device of claim 1 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is nerve tissue.
240. The device of claim 1 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is epithelium tissue.
241. The device of claim 1 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
242. The device of claim 1 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
243. The device of claim 1 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
244. The device of claim 1 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
245. The device of claim 1 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
246. The device of claim 1 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
247. The device of claim 1 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
248. The device of claim 1 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
249. The device of claim 1 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent
250. The device of claim 1 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
251. The device of claim 1 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
252. The device of claim 1 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent
253. The device of claim 1 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent
254. The device of claim 1 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
255. The device of claim 1 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
256. The device of claim 1 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
257. The device of claim 1 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
258. The device of claim 1 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
259. The device of claim 1 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
260. The device of claim 1 wherein the agent or the composition is affixed to the sensor.
261. The device of claim 1 wherein the agent or the composition is covalently attached to the sensor.
262. The device of claim 1 wherein the agent or the composition is non-covalently attached to the sensor.
263. The device of claim 1 further comprising a coating that absorbs the agent or the composition.
264. The device of claim 1 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
265. The device of claim 1 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
266. The device of claim 1 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
267. The device of claim 1 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
268. The device of claim 1 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
269. The device of claim 1 further comprising a pump that is linked to the sensor.
270. The device of any one of claims 1-269 wherein the sensor is a blood or tissue glucose monitor.
271. The device of any one of claims 1 -269 wherein the sensoryte sensor.
272. The device of any one of claims 1-269 wherein the sensornstituent sensor.
273. The device of any one of claims 1-269 wherein the sensorure sensor.
274. The device of any one of claims 1 -269 wherein the sensor r.
275. The device of any one of claims 1-269 wherein the sensor sensor.
276. The device of any one of claims 1 -269 wherein the sensormetric sensor.
277. The device of any one of claims 1-269 wherein the sensor sensor.
278. The device of any one of claims 1-269 wherein the sensor r.
279. The device of any one of claims 1-269 wherein the sensor transponder.
280. The device of any one of claims 1 -269 wherein the sensornsor.
281. The device of any one of claims 1-269 wherein the sensorresistive sensor.
282. The device of any one of claims 1 -269 wherein the sensor is a cardiac sensor.
283. The device of any one of claims 1 -269 wherein the sensor is a respiratory sensor.
284. The device of any one of claims 1-269 wherein the sensor is an auditory sensor.
285. The device of any one of claims 1-269 wherein the sensor is a metabolite sensor.
286. The device of any one of claims 1 -269 wherein the sensor detects mechanical changes.
287. The device of any one of claims 1 -269 wherein the sensor detects physical changes.
288. The device of any one of claims 1-269 wherein the sensor detects electrochemical changes.
289. The device of any one of claims 1-269 wherein the sensor detects magnetic changes.
290. The device of any one of claims 1-269 wherein the sensor detects acceleration changes.
291. The device of any one of claims 1 -269 wherein the sensor detects ionizing radiation changes.
292. The device of any one of claims 1 -269 wherein the sensor detects acoustic wave changes.
293. The device of any one of claims 1-269 wherein the sensor detects chemical changes.
294. The device of any one of claims 1-269 wherein the sensor detects drug concentration changes.
295. The device of any one of claims 1 -269 wherein the sensor detects hormone changes.
296. The device of any one of claims 1-269 wherein the sensor detects barometric changes.
297. A device, comprising a blood or tissue glucose monitor (i.e., a sensor) and an anti-scarring agent or a composition comprising an anti- scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
298. The device of claim 297 wherein the agent inhibits cell regeneration.
299. The device of claim 297 wherein the agent inhibits angiogenesis.
300. The device of claim 297 wherein the agent inhibits fibroblast migration.
301. The device of claim 297 wherein the agent inhibits fibroblast proliferation.
302. The device of claim 297 wherein the agent inhibits deposition of extracellular matrix.
303. The device of claim 297 wherein the agent inhibits tissue remodeling.
304. The device of claim 297 wherein the agent is an angiogenesis inhibitor.
305. The device of claim 297 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
306. The device of claim 297 wherein the agent is a chemokine receptor antagonist. ^
307. The device of claim 297 wherein the agent is a cell cycle inhibitor.
308. The device of claim 297 wherein the agent is a taxane.
309. The device of claim 297 wherein the agent is an antimicrotubule agent.
310. The device of claim 297 wherein the agent is paclitaxel.
311. The device of claim 297 wherein the agent is not paclitaxel.
312. The device of claim 297 wherein the agent is an analogue or derivative of paclitaxel.
313. The device of claim 297 wherein the agent is a vinca alkaloid.
314. The device of claim 297 wherein the agent is camptothecin or an analogue or derivative thereof.
315. The device of claim 297 wherein the agent is a podophyllotoxin.
316. The device of claim 297 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
317. The device of claim 297 wherein the agent is an anthracycline.
318. The device of claim 297 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
319. The device of claim 297 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
320. The device of claim 297 wherein the agent is a platinum compound.
321. The device of claim 297 wherein the agent is a nitrosourea.
322. The device of claim 297 wherein the agent is a nitroimidazole.
323. The device of claim 297 wherein the agent is a folic acid antagonist.
324. The device of claim 297 wherein the agent is a cytidine analogue.
325. The device of claim 297 wherein the agent is a pyrimidine analogue.
326. The device of claim 297 wherein the agent is a fluoropyrimidine analogue.
327. The device of claim 297 wherein the agent is a purine analogue.
328. The device of claim 297 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
329. The device of claim 297 wherein the agent is a hydroxyurea.
330. The device of claim 297 wherein the agent is a mytomicin or an analogue or derivative thereof.
331. The device of claim 297 wherein the agent is an alkyl sulfonate.
332. The device of claim 297 wherein the agent is a benzamide or an analogue or derivative thereof.
333. The device of claim 297 wherein the agent is a nicotinamide or an analogue or derivative thereof.
334. The device of claim 297 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
335. The device of claim 297 wherein the agent is a DNA alkylating agent.
336. The device of claim 297 wherein the agent is an antimicrotubule agent.
337. The device of claim 297 wherein the agent is a topoisomerase inhibitor.
338. The device of claim 297 wherein the agent is a DNA cleaving agent.
339. The device of claim 297 wherein the agent is an antimetabolite.
340. The device of claim 297 wherein the agent inhibits adenosine deaminase.
341. The device of claim 297 wherein the agent inhibits purine ring synthesis.
342. The device of claim 297 wherein the agent is a nucleotide interconversion inhibitor.
343. The device of claim 297 wherein the agent inhibits dihydrofolate reduction.
344. The device of claim 297 wherein the agent blocks thymidine monophosphate.
345. The device of claim 297 wherein the agent causes DNA damage.
346. The device of claim 297 wherein the agent is a DNA intercalation agent.
347. The device of claim 297 wherein the agent is a RNA synthesis inhibitor.
348. The device of claim 297 wherein the agent is a pyrimidine synthesis inhibitor.
349. The device of claim 297 wherein the agent inhibits ribonucleotide synthesis or function.
350. The device of claim 297 wherein the agent inhibits thymidine monophosphate synthesis or function.
351. The device of claim 297 wherein the agent inhibits DNA synthesis.
352. The device of claim 297 wherein the agent causes DNA adduct formation.
353. The device of claim 297 wherein the agent inhibits protein synthesis.
354. The device of claim 297 wherein the agent inhibits microtubule function.
355. The device of claim 297 wherein the agent is a cyclin dependent protein kinase inhibitor.
356. The device of claim 297 wherein the agent is an epidermal growth factor kinase inhibitor.
357. The device of claim 297 wherein the agent is an elastase inhibitor.
358. The device of claim 297 wherein the agent is a factor Xa inhibitor.
359. The device of claim 297 wherein the agent is a farnesyltransferase inhibitor.
360. The device of claim 297 wherein the agent is a fibrinogen antagonist.
361. The device of claim 297 wherein the agent is a guanylate cyclase stimulant.
362. The device of claim 297 wherein the agent is a heat shock protein 90 antagonist.
363. The device of claim 297 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
364. The device of claim 297 wherein the agent is a guanylate cyclase stimulant.
365. The device of claim 297 wherein the agent is a HMGCoA reductase inhibitor.
366. The device of claim 297 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
367. The device of claim 297 wherein the agent is a hydroorotate dehydrogenase inhibitor.
368. The device of claim 297 wherein the agent is an IKK2 inhibitor.
369. The device of claim 297 wherein the agent is an IL-1 antagonist
370. The device of claim 297 wherein the agent is an ICE antagonist.
371. The device of claim 297 wherein the agent is an IRAK antagonist.
372. The device of claim 297 wherein the agent is an IL-4 agonist.
373. The device of claim 297 wherein the agent is an immunomodulatory agent.
374. The device of claim 297 wherein the agent is sirolimus or an analogue or derivative thereof.
375. The device of claim 297 wherein the agent is not sirolimus.
376. The device of claim 297 wherein the agent is everolimus or an analogue or derivative thereof.
377. The device of claim 297 wherein the agent is tacrolimus or an analogue or derivative thereof.
378. The device of claim 297 wherein the agent is not tacrolimus.
379. The device of claim 297 wherein the agent is biolmus or an analogue or derivative thereof.
380. The device of claim 297 wherein the agent is tresperimus or an analogue or derivative thereof.
381. The device of claim 297 wherein the agent is auranofin or an analogue or derivative thereof.
382. The device of claim 297 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
383. The device of claim 297 wherein the agent is gusperimus or an analogue or derivative thereof.
384. The device of claim 297 wherein the agent is pimecrolimus or an analogue or derivative thereof.
385. The device of claim 297 wherein the agent is ABT-578 or an analogue or derivative thereof.
386. The device of claim 297 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
387. The device of claim 297 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
388. The device of claim 297 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
389. The device of claim 297 wherein the agent is a leukotriene inhibitor.
390. The device of claim 297 wherein the agent is a MCP-1 antagonist.
391. The device of claim 297 wherein the agent is a MMP inhibitor.
392. The device of claim 297 wherein the agent is an NF kappa B inhibitor.
393. The device of claim 297 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
394. The device of claim 297 wherein the agent is an NO antagonist.
395. The device of claim 297 wherein the agent is a p38 MAP kinase inhibitor.
396. The device of claim 297 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
397. The device of claim 297 wherein the agent is a phosphodiesterase inhibitor.
398. The device of claim 297 wherein the agent is a TGF beta inhibitor.
399. The device of claim 297 wherein the agent is a thromboxane A2 antagonist.
400. The device of claim 297 wherein the agent is a TNF alpha antagonist.
401. The device of claim 297 wherein the agent is a TACE inhibitor.
402. The device of claim 297 wherein the agent is a tyrosine kinase inhibitor.
403. The device of claim 297 wherein the agent is a vitronectin inhibitor.
404. The device of claim 297 wherein the agent is a fibroblast growth factor inhibitor.
405. The device of claim 297 wherein the agent is a protein kinase inhibitor.
406. The device of claim 297 wherein the agent is a PDGF receptor kinase inhibitor.
407. The device of claim 297 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
408. The device of claim 297 wherein the agent is a retinoic acid receptor antagonist.
409. The device of claim 297 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
410. The device of claim 297 wherein the agent is a fibrinogen antagonist.
411. The device of claim 297 wherein the agent is an antimycotic agent.
412. The device of claim 297 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
413. The device of claim 297 wherein the agent is a bisphosphonate.
414. The device of claim 297 wherein the agent is a phospholipase A1 inhibitor.
415. The device of claim 297 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
416. The device of claim 297 wherein the agent is a macrolide antibiotic.
417. The device of claim 297 wherein the agent is a GPIIb/llla receptor antagonist.
418. The device of claim 297 wherein the agent is an endothelin receptor antagonist.
419. The device of claim 297 wherein the agent is a peroxisome proliferator-activated receptor agonist.
420. The device of claim 297 wherein the agent is an estrogen receptor agent.
421. The device of claim 297 wherein the agent is a somastostatin analogue.
422. The device of claim 297 wherein the agent is a neurokinin 1 antagonist.
423. The device of claim 297 wherein the agent is a neurokinin 3 antagonist.
424. The device of claim 297 wherein the agent is a VLA-4 antagonist
425. The device of claim 297 wherein the agent is an osteoclast inhibitor.
426. The device of claim 297 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
427. The device of claim 297 wherein the agent is an angiotensin I converting enzyme inhibitor.
428. The device of claim 297 wherein the agent is an angiotensin II antagonist.
429. The device of claim 297 wherein the agent is an enkephalinase inhibitor.
430. The device of claim 297 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
431. The device of claim 297 wherein the agent is a protein kinase C inhibitor.
432. The device of claim 297 wherein the agent is a ROCK (rho- associated kinase) inhibitor.
433. The device of claim 297 wherein the agent is a CXCR3 inhibitor.
434. The device of claim 297 wherein the agent is an Itk inhibitor.
435. The device of claim 297 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
436. The device of claim 297 wherein the agent is a PPAR agonist.
437. The device of claim 297 wherein the agent is an immunosuppressant
438. The device of claim 297 wherein the agent is an Erb inhibitor.
439. The device of claim 297 wherein the agent is an apoptosis agonist.
440. The device of claim 297 wherein the agent is a lipocortin agonist.
441. The device of claim 297 wherein the agent is a VCAM-1 antagonist.
442. The device of claim 297 wherein the agent is a collagen antagonist.
443. The device of claim 297 wherein the agent is an alpha 2 integrin antagonist.
444. The device of claim 297 wherein the agent is a TNF alpha inhibitor.
445. The device of claim 297 wherein the agent is a nitric oxide inhibitor
446. The device of claim 297 wherein the agent is a cathepsin inhibitor.
447. The device of claim 297 wherein the agent is not an anti- inflammatory agent.
448. The device of claim 297 wherein the agent is not a steroid.
449. The device of claim 297 wherein the agent is not a glucocorticosteroid.
450. The device of claim 297 wherein the agent is not dexamethasone.
451. The device of claim 297 wherein the agent is not an anti- infective agent.
452. The device of claim 297 wherein the agent is not an antibiotic.
453. The device of claim 297 wherein the agent is not an anti- fugal agent.
454. The device of claim 297 wherein the agent is not beclomethasone.
455. The device of claim 297 wherein the agent is not dipropionate.
456. The device of claim 297, further comprising a coating, wherein the coating comprises the anti-scarring agent and a polymer.
457. The device of claim 297, further comprising a coating, wherein the coating comprises the anti-scarring agent.
458. The device of claim 297, further comprising a coating, wherein the coating is disposed on a surface of the device.
459. The device of claim 297, further comprising a coating, wherein the coating directly contacts the device.
460. The device of claim 297, further comprising a coating, wherein the coating indirectly contacts the device.
461. The device of claim 297, further comprising a coating, wherein the coating partially covers the device.
462. The device of claim 297, further comprising a coating, wherein the coating completely covers the device.
463. The device of claim 297, further comprising a coating, wherein the coating is a uniform coating.
464. The device of claim 297, further comprising a coating, wherein the coating is a non-uniform coating.
465. The device of claim 297, further comprising a coating, wherein the coating is a discontinuous coating.
466. The device of claim 297, further comprising a coating, wherein the coating is a patterned coating.
467. The device of claim 297, further comprising a coating, wherein the coating has a thickness of 100 μm or less.
468. The device of claim 297, further comprising a coating, wherein the coating has a thickness of 10 μm or less.
469. The device of claim 297, further comprising a coating, wherein the coating adheres to the surface of the device upon deployment of the device.
470. The device of claim 297, further comprising a coating, wherein the coating is stable at room temperature for a period of 1 year.
471. The device of claim 297, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1% by weight.
472. The device of claim 297, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
473. The device of claim 297, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
474. The device of claim 297, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
475. The device of claim 297, further comprising a coating, wherein the coating further comprises a polymer.
476. The device of claim 297, further comprising a first coating having a first composition and the second coating having a second composition.
477. The device of claim 297, further comprising a first coating having a first composition and the second coating having a second composition, wherein the first composition and the second composition are different.
478. The device of claim 297, further comprising a polymer.
479. The device of claim 297, further comprising a polymeric carrier.
480. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a copolymer.
481. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a block copolymer.
482. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a random copolymer.
483. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a biodegradable polymer.
484. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-biodegradable polymer.
485. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophilic polymer.
486. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophobic polymer.
487. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophilic domains.
488. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophobic domains.
489. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-conductive polymer.
490. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises an elastomer.
491. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrogel.
492. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a silicone polymer.
493. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrocarbon polymer.
494. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a styrene-derived polymer.
495. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a butadiene polymer.
496. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a macromer.
497. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises a poly( ethylene glycol) polymer.
498. The device of claim 297, further comprising a polymeric carrier, wherein the polymeric carrier comprises an amorphous polymer.
499. The device of claim 297, further comprising a lubricious coating.
500. The device of claim 297 wherein the anti-scarring agent is located within pores or holes of the device.
501. The device of claim 297 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
502. The device of claim 297, further comprising a second pharmaceutically active agent.
503. The device of claim 297, further comprising an anti- inflammatory agent.
504. The device of claim 297, further comprising an agent that inhibits infection.
505. The device of claim 297, further comprising an agent that inhibits infection, wherein the agent is an anthracycline.
506. The device of claim 297, further comprising an agent that inhibits infection, wherein the agent is doxorubicin.
507. The device of claim 297, further comprising an agent that inhibits infection, wherein the agent is mitoxantrone.
508. The device of claim 297, further comprising an agent that inhibits infection, wherein the agent is a fluoropyrimidine.
509. The device of claim 297, further comprising an agent that inhibits infection, wherein the agent is 5-fluorouracil (5-FU).
510. The device of claim 297, further comprising an agent that inhibits infection, wherein the agent is a folic acid antagonist.
511. The device of claim 297, further comprising an agent that inhibits infection, wherein the agent is methotrexate.
512. The device of claim 297, further comprising an agent that inhibits infection, wherein the agent is a podophylotoxin.
513. The device of claim 297, further comprising an agent that inhibits infection, wherein the agent is etoposide.
514. The device of claim 297, further comprising an agent that inhibits infection, wherein the agent is a camptothecin.
515. The device of claim 297, further comprising an agent that inhibits infection, wherein the agent is a hydroxyurea.
516. The device of claim 297, further comprising an agent that inhibits infection, wherein the agent is a platinum complex.
517. The device of claim 297, further comprising an agent that inhibits infection, wherein the agent is cisplatin.
518. The device of claim 297, further comprising an anti- thrombotic agent.
519. The device of claim 297, further comprising a visualization agent.
520. The device of claim 297, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
521. The device of claim 297, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises barium, tantalum, or technetium.
522. The device of claim 297, further comprising a visualization agent, wherein the visualization agent is a MRI responsive material.
523. The device of claim 297, further comprising a visualization agent, wherein the visualization agent comprises a gadolinium chelate.
524. The device of claim 297, further comprising a visualization agent, wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
525. The device of claim 297, further comprising a visualization agent, wherein the visualization agent comprises an iron oxide compound.
526. The device of claim 297, further comprising a visualization agent, wherein the visualization agent comprises a dye, pigment, or colorant.
527. The device of claim 297, further comprising an echogenic material.
528. The device of claim 297, further comprising an echogenic material, wherein the echogenic material is in the form of a coating.
529. The device of claim 297 wherein the device is sterile.
530. The device of claim 297 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
531. The device of claim 297 wherein the device is adapted for delivering the anti-scarring agent locally to tissue proximate to the device.
532. The device of claim 297 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
533. The device of claim 297 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is connective tissue.
534. The device of claim 297 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is muscle tissue.
535. The device of claim 297 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is nerve tissue.
536. The device of claim 297 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is epithelium tissue.
537. The device of claim 297 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
538. The device of claim 297 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
539. The device of claim 297 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
540. The device of claim 297 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
541. The device of claim 297 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
542. The device of claim 297 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
543. The device of claim 297 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
544. The device of claim 297 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
545. The device of claim 297 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
546. The device of claim 297 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
547. The device of claim 297 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
548. The device of claim 297 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
549. The device of claim 297 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
550. The device of claim 297 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
551. The device of claim 297 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
552. The device of claim 297 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
553. The device of claim 297 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
554. The device of claim 297 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
555. The device of claim 297 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
556. The device of claim 297 wherein the agent or the composition is affixed to the sensor.
557. The device of claim 297 wherein the agent or the composition is covalently attached to the sensor.
558. The device of claim 297 wherein the agent or the composition is non-covalently attached to the sensor.
559. The device of claim 297 further comprising a coating that absorbs the agent or the composition.
560. The device of claim 297 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
561. The device of claim 297 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
562. The device of claim 297 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
563. The device of claim 297 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
564. The device of claim 297 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
565. The device of claim 297 further comprising a pump that is linked to the sensor.
566. The device of any one of claims 297-565 wherein the device is deliverable to the vascular system transluminally using a catheter on a stent platform.
567. The device of any one of claims 297-565 wherein the device is composed of glucose sensitive living cells that monitor blood glucose levels and produce a detectable electrical or optical signal in response to changes in glucose concentrations.
568. The device of any one of claims 297-565 wherein the device is an electrode composed of an analyte responsive enzyme.
569. The device of any one of claims 297-565 wherein the device is a closed loop insulin delivery system that comprises a sensing means that detects the host's blood glucose level and stimulates an insulin pump to supply insulin.
570. The device of any one of claims 297-565 wherein the device is a closed loop insulin delivery system that comprises a sensing means that detects the host's blood glucose level and stimulates the pancreas to supply insulin.
571. A device, comprising a pressure or stress sensor and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
572. The device of claim 571 wherein the agent inhibits cell regeneration.
573. The device of claim 571 wherein the agent inhibits angiogenesis.
574. The device of claim 571 wherein the agent inhibits fibroblast migration.
575. The device of claim 571 wherein the agent inhibits fibroblast proliferation.
576. The device of claim 571 wherein the agent inhibits deposition of extracellular matrix.
577. The device of claim 571 wherein the agent inhibits tissue remodeling.
578. The device of claim 571 wherein the agent is an angiogenesis inhibitor.
579. The device of claim 571 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
580. The device of claim 571 wherein the agent is a chemokine receptor antagonist.
581. The device of claim 571 wherein the agent is a cell cycle inhibitor.
582. The device of claim 571 wherein the agent is a taxane.
583. The device of claim 571 wherein the agent is an antimicrotubule agent.
584. The device of claim 571 wherein the agent is paclitaxel.
585. The device of claim 571 wherein the agent is not paclitaxel.
586. The device of claim 571 wherein the agent is an analogue or derivative of paclitaxel.
587. The device of claim 571 wherein the agent is a vinca alkaloid.
588. The device of claim 571 wherein the agent is camptothecin or an analogue or derivative thereof.
589. The device of claim 571 wherein the agent is a podophyllotoxin.
590. The device of claim 571 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
591. The device of claim 571 wherein the agent is an anthracycline.
592. The device of claim 571 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
593. The device of claim 571 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
594. The device of claim 571 wherein the agent is a platinum compound.
595. The device of claim 571 wherein the agent is a nitrosourea.
596. The device of claim 571 wherein the agent is a nitroimidazole.
597. The device of claim 571 wherein the agent is a folic acid antagonist.
598. The device of claim 571 wherein the agent is a cytidine analogue.
599. The device of claim 571 wherein the agent is a pyrimidine analogue.
600. The device of claim 571 wherein the agent is a fluoropyrimidine analogue.
601. The device of claim 571 wherein the agent is a purine analogue.
602. The device of claim 571 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
603. The device of claim 571 wherein the agent is a hydroxyurea.
604. The device of claim 571 wherein the agent is a mytomicin or an analogue or derivative thereof.
605. The device of claim 571 wherein the agent is an alkyl sulfonate.
606. The device of claim 571 wherein the agent is a benzamide or an analogue or derivative thereof.
607. The device of claim 571 wherein the agent is a nicotinamide or an analogue or derivative thereof.
608. The device of claim 571 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
609. The device of claim 571 wherein the agent is a DNA alkylating agent.
610. The device of claim 571 wherein the agent is an antimicrotubule agent.
611. The device of claim 571 wherein the agent is a topoisomerase inhibitor.
612. The device of claim 571 wherein the agent is a DNA cleaving agent.
613. The device of claim 571 wherein the agent is an antimetabolite.
614. The device of claim 571 wherein the agent inhibits adenosine deaminase.
615. The device of claim 571 wherein the agent inhibits purine ring synthesis.
616. The device of claim 571 wherein the agent is a nucleotide interconversion inhibitor.
617. The device of claim 571 wherein the agent inhibits dihydrofolate reduction.
618. The device of claim 571 wherein the agent blocks thymidine monophosphate.
619. The device of claim 571 wherein the agent causes DNA damage.
620. The device of claim 571 wherein the agent is a DNA intercalation agent.
621. The device of claim 571 wherein the agent is a RNA synthesis inhibitor.
622. The device of claim 571 wherein the agent is a pyrimidine synthesis inhibitor.
623. The device of claim 571 wherein the agent inhibits ribonucleotide synthesis or function.
624. The device of claim 571 wherein the agent inhibits thymidine monophosphate synthesis or function.
625. The device of claim 571 wherein the agent inhibits DNA synthesis.
626. The device of claim 571 wherein the agent causes DNA adduct formation.
627. The device of claim 571 wherein the agent inhibits protein synthesis.
628. The device of claim 571 wherein the agent inhibits microtubule function.
629. The device of claim 571 wherein the agent is a cyclin dependent protein kinase inhibitor.
630. The device of claim 571 wherein the agent is an epidermal growth factor kinase inhibitor.
631. The device of claim 571 wherein the agent is an elastase inhibitor.
632. The device of claim 571 wherein the agent is a factor Xa inhibitor.
633. The device of claim 571 wherein the agent is a farnesyltransferase inhibitor.
634. The device of claim 571 wherein the agent is a fibrinogen antagonist.
635. The device of claim 571 wherein the agent is a guanylate cyclase stimulant.
636. The device of claim 571 wherein the agent is a heat shock protein 90 antagonist.
637. The device of claim 571 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
638. The device of claim 571 wherein the agent is a guanylate cyclase stimulant
639. The device of claim 571 wherein the agent is a HMGCoA reductase inhibitor.
640. The device of claim 571 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
641. The device of claim 571 wherein the agent is a hydroorotate dehydrogenase inhibitor.
642. The device of claim 571 wherein the agent is an IKK2 inhibitor.
643. The device of claim 571 wherein the agent is an IL-1 antagonist.
644. The device of claim 571 wherein the agent is an ICE antagonist.
645. The device of claim 571 wherein the agent is an IRAK antagonist.
646. The device of claim 571 wherein the agent is an IL-4 agonist.
647. The device of claim 571 wherein the agent is an immunomodulatory agent.
648. The device of claim 571 wherein the agent is sirolimus or an analogue or derivative thereof.
649. The device of claim 571 wherein the agent is not sirolimus.
650. The device of claim 571 wherein the agent is everolimus or an analogue or derivative thereof.
651. The device of claim 571 wherein the agent is tacrolimus or an analogue or derivative thereof.
652. The device of claim 571 wherein the agent is not tacrolimus.
653. The device of claim 571 wherein the agent is biolmus or an analogue or derivative thereof.
654. The device of claim 571 wherein the agent is tresperimus or an analogue or derivative thereof. I
655. The device of claim 571 wherein the agent is auranofin or an analogue or derivative thereof.
656. The device of claim 571 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
657. The device of claim 571 wherein the agent is gusperimus or an analogue or derivative thereof.
658. The device of claim 571 wherein the agent is pimecrolimus or an analogue or derivative thereof.
659. The device of claim 571 wherein the agent is ABT-578 or an analogue or derivative thereof.
660. The device of claim 571 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
661. The device of claim 571 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
662. The device of claim 571 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
663. The device of claim 571 wherein the agent is a leukotriene inhibitor.
664. The device of claim 571 wherein the agent is a MCP-1 antagonist.
665. The device of claim 571 wherein the agent is a MMP inhibitor.
666. The device of claim 571 wherein the agent is an NF kappa B inhibitor.
667. The device of claim 571 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
668. The device of claim 571 wherein the agent is an NO antagonist.
669. The device of claim 571 wherein the agent is a p38 MAP kinase inhibitor.
670. The device of claim 571 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
671. The device of claim 571 wherein the agent is a phosphodiesterase inhibitor.
672. The device of claim 571 wherein the agent is a TGF beta inhibitor.
673. The device of claim 571 wherein the agent is a thromboxane A2 antagonist.
674. The device of claim 571 wherein the agent is a TNF alpha antagonist.
675. The device of claim 571 wherein the agent is a TACE inhibitor.
676. The device of claim 571 wherein the agent is a tyrosine kinase inhibitor.
677. The device of claim 571 wherein the agent is a vitronectin inhibitor.
678. The device of claim 571 wherein the agent is a fibroblast growth factor inhibitor.
679. The device of claim 571 wherein the agent is a protein kinase inhibitor.
680. The device of claim 571 wherein the agent is a PDGF receptor kinase inhibitor.
681. The device of claim 571 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
682. The device of claim 571 wherein the agent is a retinoic acid receptor antagonist.
683. The device of claim 571 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
684. The device of claim 571 wherein the agent is a fibrinogen antagonist.
685. The device of claim 571 wherein the agent is an antimycotic agent.
686. The device of claim 571 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
687. The device of claim 571 wherein the agent is a bisphosphonate.
688. The device of claim 571 wherein the agent is a phospholipase A1 inhibitor.
689. The device of claim 571 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
690. The device of claim 571 wherein the agent is a macrolide antibiotic.
691. The device of claim 571 wherein the agent is a GPIIb/llla receptor antagonist.
692. The device of claim 571 wherein the agent is an endothelin receptor antagonist.
693. The device of claim 571 wherein the agent is a peroxisome proliferator-activated receptor agonist.
694. The device of claim 571 wherein the agent is an estrogen receptor agent.
695. The device of claim 571 wherein the agent is a somastostatin analogue.
696. The device of claim 571 wherein the agent is a neurokinin 1 antagonist.
697. The device of claim 571 wherein the agent is a neurokinin 3 antagonist.
698. The device of claim 571 wherein the agent is a VLA-4 antagonist.
699. The device of claim 571 wherein the agent is an osteoclast inhibitor.
700. The device of claim 571 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
701. The device of claim 571 wherein the agent is an angiotensin I converting enzyme inhibitor.
702. The device of claim 571 wherein the agent is an angiotensin II antagonist.
703. The device of claim 571 wherein the agent is an enkephalinase inhibitor.
704. The device of claim 571 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
705. The device of claim 571 wherein the agent is a protein kinase C inhibitor.
706. The device of claim 571 wherein the agent is a ROCK (rho- associated kinase) inhibitor.
707. The device of claim 571 wherein the agent is a CXCR3 inhibitor.
708. The device of claim 571 wherein the agent is an Itk inhibitor.
709. The device of claim 571 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
710. The device of claim 571 wherein the agent is a PPAR agonist.
711. The device of claim 571 wherein the agent is an immunosuppressant.
712. The device of claim 571 wherein the agent is an Erb inhibitor.
713. The device of claim 571 wherein the agent is an apoptosis agonist.
714. The device of claim 571 wherein the agent is a lipocortin agonist.
715. The device of claim 571 wherein the agent is a VCAM-1 antagonist.
716. The device of claim 571 wherein the agent is a collagen antagonist.
717. The device of claim 571 wherein the agent is an alpha 2 integrin antagonist.
718. The device of claim 571 wherein the agent is a TNF alpha inhibitor.
I 719. The device of claim 571 wherein the agent is a nitric oxide inhibitor
720. The device of claim 571 wherein the agent is a cathepsin inhibitor.
721. The device of claim 571 wherein the agent is not an anti- inflammatory agent.
722. The device of claim 571 wherein the agent is not a steroid.
723. The device of claim 571 wherein the agent is not a glucocorticosteroid .
724. The device of claim 571 wherein the agent is not dexamethasone.
725. The device of claim 571 wherein the agent is not an anti- infective agent.
726. The device of claim 571 wherein the agent is not an antibiotic.
727. The device of claim 571 wherein the agent is not an anti- fugal agent.
728. The device of claim 571 wherein the agent is not beclomethasone.
729. The device of claim 571 wherein the agent is not dipropionate.
730. The device of claim 571 , further comprising a coating, wherein the coating comprises the anti-scarring agent and a polymer.
731. The device of claim 571 , further comprising a coating, wherein the coating comprises the anti-scarring agent.
732. The device of claim 571 , further comprising a coating, wherein the coating is disposed on a surface of the device.
733. The device of claim 571 , further comprising a coating, wherein the coating directly contacts the device.
734. The device of claim 571 , further comprising a coating, wherein the coating indirectly contacts the device.
735. The device of claim 571 , further comprising a coating, wherein the coating partially covers the device.
736. The device of claim 571 , further comprising a coating, wherein the coating completely covers the device.
737. The device of claim 571 , further comprising a coating, wherein the coating is a uniform coating.
738. The device of claim 571 , further comprising a coating, wherein the coating is a non-uniform coating.
739. The device of claim 571 , further comprising a coating, wherein the coating is a discontinuous coating.
740. The device of claim 571 , further comprising a coating, wherein the coating is a patterned coating.
741. The device of claim 571 , further comprising a coating, wherein the coating has a thickness of 100 μm or less.
742. The device of claim 571 , further comprising a coating, wherein the coating has a thickness of 10 μm or less.
743. The device of claim 571 , further comprising a coating, wherein the coating adheres to the surface of the device upon deployment of the device.
744. The device of claim 571 , further comprising a coating, wherein the coating is stable at room temperature for a period of 1 year.
745. The device of claim 571 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1% by weight
746. The device of claim 571 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
747. The device of claim 571 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight
748. The device of claim 571 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
749. The device of claim 571 , further comprising a coating, wherein the coating further comprises a polymer.
750. The device of claim 571 , further comprising a first coating having a first composition and the second coating having a second composition.
751. The device of claim 571 , further comprising a first coating having a first composition and the second coating having a second composition, wherein the first composition and the second composition are different.
752. The device of claim 571 , further comprising a polymer.
753. The device of claim 571 , further comprising a polymeric carrier.
754. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a copolymer.
755. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a block copolymer.
756. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a random copolymer.
757. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a biodegradable polymer.
758. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-biodegradable polymer.
759. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophilic polymer.
760. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophobic polymer.
761. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophilic domains.
762. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophobic domains.
763. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-conductive polymer.
764. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises an elastomer.
765. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrogel.
766. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a silicone polymer.
767. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrocarbon polymer.
768. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a styrene-derived polymer.
769. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a butadiene polymer.
770. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a macromer.
771. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
772. The device of claim 571 , further comprising a polymeric carrier, wherein the polymeric carrier comprises an amorphous polymer.
773. The device of claim 571 , further comprising a lubricious coating.
774. The device of claim 571 wherein the anti-scarring agent is located within pores or holes of the device.
775. The device of claim 571 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
776. The device of claim 571 , further comprising a second pharmaceutically active agent.
777. The device of claim 571 , further comprising an anti- inflammatory agent.
778. The device of claim 571 , further comprising an agent that inhibits infection.
779. The device of claim 571 , further comprising an agent that inhibits infection, wherein the agent is an anthracycline.
780. The device of claim 571 , further comprising an agent that inhibits infection, wherein the agent is doxorubicin.
781. The device of claim 571 , further comprising an agent that inhibits infection, wherein the agent is mitoxantrone.
782. The device of claim 571 , further comprising an agent that inhibits infection, wherein the agent is a fluoropyrimidine.
783. The device of claim 571 , further comprising an agent that inhibits infection, wherein the agent is 5-fluorouracil (5-FU).
784. The device of claim 571 , further comprising an agent that inhibits infection, wherein the agent is a folic acid antagonist.
785. The device of claim 571 , further comprising an agent that inhibits infection, wherein the agent is methotrexate.
786. The device of claim 571 , further comprising an agent that inhibits infection, wherein the agent is a podophylotoxin.
787. The device of claim 571 , further comprising an agent that inhibits infection, wherein the agent is etoposide.
788. The device of claim 571 , further comprising an agent that inhibits infection, wherein the agent is a camptothecin.
789. The device of claim 571 , further comprising an agent that inhibits infection, wherein the agent is a hydroxyurea.
790. The device of claim 571 , further comprising an agent that inhibits infection, wherein the agent is a platinum complex.
791. The device of claim 571 , further comprising an agent that inhibits infection, wherein the agent is cisplatin.
792. The device of claim 571 , further comprising an anti- thrombotic agent
793. The device of claim 571 , further comprising a visualization agent.
794. The device of claim 571 , further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
795. The device of claim 571 , further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises barium, tantalum, or technetium.
796. The device of claim 571 , further comprising a visualization agent, wherein the visualization agent is a MRI responsive material.
797. The device of claim 571 , further comprising a visualization agent, wherein the visualization agent comprises a gadolinium chelate.
798. The device of claim 571 , further comprising a visualization agent, wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
799. The device of claim 571 , further comprising a visualization agent, wherein the visualization agent comprises an iron oxide compound.
800. The device of claim 571 , further comprising a visualization agent, wherein the visualization agent comprises a dye, pigment, or colorant.
801. The device of claim 571 , further comprising an echogenic material.
802. The device of claim 571 , further comprising an echogenic material, wherein the echogenic material is in the form of a coating.
803. The device of claim 571 wherein the device is sterile.
804. The device of claim 571 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
805. The device of claim 571 wherein the device is adapted for delivering the anti-scarring agent locally to tissue proximate to the device.
806. The device of claim 571 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
807. The device of claim 571 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is connective tissue.
808. The device of claim 571 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is muscle tissue.
809. The device of claim 571 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is nerve tissue.
810. The device of claim 571 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is epithelium tissue.
811. The device of claim 571 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
812. The device of claim 571 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
813. The device of claim 571 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
814. The device of claim 571 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
815. The device of claim 571 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
816. The device of claim 571 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
817. The device of claim 571 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
818. The device of claim 571 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
819. The device of claim 571 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent
820. The device of claim 571 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
821. The device of claim 571 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
822. The device of claim 571 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
823. The device of claim 571 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
824. The device of claim 571 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
825. The device of claim 571 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
826. The device of claim 571 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
827. The device of claim 571 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
828. The device of claim 571 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
829. The device of claim 571 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
830. The device of claim 571 wherein the agent or the composition is affixed to the sensor.
831. The device of claim 571 wherein the agent or the composition is covalently attached to the sensor.
832. The device of claim 571 wherein the agent or the composition is non-covalently attached to the sensor.
833. The device of claim 571 further comprising a coating that absorbs the agent or the composition.
834. The device of claim 571 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
835. The device of claim 571 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
836. The device of claim 571 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
837. The device of claim 571 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
838. The device of claim 571 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
839. The device of claim 571 further comprising a pump that is linked to the sensor.
840. The device of any one of claims 571 -839 wherein the device monitors blood pressure.
841. The device of any one of claims 571 -839 wherein the device monitors fluid flow.
842. The device of any one of claims 571 -839 wherein the device monitors pressure within an aneurysm sac.
843. The device of any one of claims 571 -839 wherein the device monitors intracranial pressure.
844. The device of any one of claims 571 -839 wherein the device monitors mechanical pressure associated with a bone fracture.
845. The device of any one of claims 571 -839 wherein the device monitors barometric pressure.
846. The device of any one of claims 571 -839 wherein the device monitors eye tremors.
847. The device of any one of claims 571 -839 wherein the device monitors the depth of a corneal implant.
848. The device of any one of claims 571 -839 wherein the device monitors intraocular pressure.
849. The device of any one of claims 571-839 wherein the device is a passive sensor with an inductor-capacitor circuit.
850. The device of any one of claims 571-839 wherein the device is a self-powered strain sensing system.
851. The device of any one of claims 571-839 wherein the sensor comprises a lead, a sensor module, a sensor circuit and means for providing voltage.
852. A device, comprising a cardiac sensor and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
853. The device of claim 852 wherein the agent inhibits cell regeneration.
854. The device of claim 852 wherein the agent inhibits angiogenesis.
855. The device of claim 852 wherein the agent inhibits fibroblast migration.
856. The device of claim 852 wherein the agent inhibits fibroblast proliferation.
857. The device of claim 852 wherein the agent inhibits deposition of extracellular matrix.
858. The device of claim 852 wherein the agent inhibits tissue remodeling.
859. The device of claim 852 wherein the agent is an angiogenesis inhibitor.
860. The device of claim 852 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
861. The device of claim 852 wherein the agent is a chemokine receptor antagonist.
862. The device of claim 852 wherein the agent is a cell cycle inhibitor.
863. The device of claim 852 wherein the agent is a taxane.
864. The device of claim 852 wherein the agent is an antimicrotubule agent.
865. The device of claim 852 wherein the agent is paclitaxel.
866. The device of claim 852 wherein the agent is not paclitaxel.
867. The device of claim 852 wherein the agent is an analogue or derivative of paclitaxel.
868. The device of claim 852 wherein the agent is a vinca alkaloid.
869. The device of claim 852 wherein the agent is camptothecin or an analogue or derivative thereof.
870. The device of claim 852 wherein the agent is a podophyllotoxin.
871. The device of claim 852 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
872. The device of claim 852 wherein the agent is an anthracycline.
873. The device of claim 852 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
874. The device of claim 852 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
875. The device of claim 852 wherein the agent is a platinum compound.
876. The device of claim 852 wherein the agent is a nitrosourea.
877. The device of claim 852 wherein the agent is a nitroimidazole.
878. The device of claim 852 wherein the agent is a folic acid antagonist.
879. The device of claim 852 wherein the agent is a cytidine analogue.
880. The device of claim 852 wherein the agent is a pyrimidine analogue.
881. The device of claim 852 wherein the agent is a fluoropyrimidine analogue.
882. The device of claim 852 wherein the agent is a purine analogue.
883. The device of claim 852 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
884. The device of claim 852 wherein the agent is a hydroxyurea.
885. The device of claim 852 wherein the agent is a mytomicin or an analogue or derivative thereof.
886. The device of claim 852 wherein the agent is an alkyl sulfonate.
887. The device of claim 852 wherein the agent is a benzamide or an analogue or derivative thereof.
888. The device of claim 852 wherein the agent is a nicotinamide or an analogue or derivative thereof.
889. The device of claim 852 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
890. The device of claim 852 wherein the agent is a DNA alkylating agent.
891. The device of claim 852 wherein the agent is an antimicrotubule agent.
892. The device of claim 852 wherein the agent is a topoisomerase inhibitor.
893. The device of claim 852 wherein the agent is a DNA cleaving agent.
894. The device of claim 852 wherein the agent is an antimetabolite.
895. The device of claim 852 wherein the agent inhibits adenosine deaminase.
896. The device of claim 852 wherein the agent inhibits purine ring synthesis.
897. The device of claim 852 wherein the agent is a nucleotide interconversion inhibitor.
898. The device of claim 852 wherein the agent inhibits dihydrofolate reduction.
899. The device of claim 852 wherein the agent blocks thymidine monophosphate.
900. The device of claim 852 wherein the agent causes DNA damage.
901. The device of claim 852 wherein the agent is a DNA intercalation agent.
902. The device of claim 852 wherein the agent is a RNA synthesis inhibitor.
903. The device of claim 852 wherein the agent is a pyrimidine synthesis inhibitor.
904. The device of claim 852 wherein the agent inhibits ribonucleotide synthesis or function.
905. The device of claim 852 wherein the agent inhibits thymidine monophosphate synthesis or function.
906. The device of claim 852 wherein the agent inhibits DNA synthesis.
907. The device of claim 852 wherein the agent causes DNA adduct formation.
908. The device of claim 852 wherein the agent inhibits protein synthesis.
909. The device of claim 852 wherein the agent inhibits microtubule function.
910. The device of claim 852 wherein the agent is a cyclin dependent protein kinase inhibitor.
911. The device of claim 852 wherein the agent is an epidermal growth factor kinase inhibitor.
912. The device of claim 852 wherein the agent is an elastase inhibitor.
913. The device of claim 852 wherein the agent is a factor Xa inhibitor.
914. The device of claim 852 wherein the agent is a farnesyltransferase inhibitor.
915. The device of claim 852 wherein the agent is a fibrinogen antagonist.
916. The device of claim 852 wherein the agent is a guanylate cyclase stimulant
917. The device of claim 852 wherein the agent is a heat shock protein 90 antagonist.
918. The device of claim 852 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
919. The device of claim 852 wherein the agent is a guanylate cyclase stimulant.
920. The device of claim 852 wherein the agent is a HMGCoA reductase inhibitor.
921. The device of claim 852 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
922. The device of claim 852 wherein the agent is a hydroorotate dehydrogenase inhibitor.
923. The device of claim 852 wherein the agent is an IKK2 inhibitor.
924. The device of claim 852 wherein the agent is an IL-1 antagonist.
925. The device of claim 852 wherein the agent is an ICE antagonist.
926. The device of claim 852 wherein the agent is an IRAK antagonist.
927. The device of claim 852 wherein the agent is an IL-4 agonist.
928. The device of claim 852 wherein the agent is an immunomodulatory agent.
929. The device of claim 852 wherein the agent is sirolimus or an analogue or derivative thereof.
930. The device of claim 852 wherein the agent is not sirolimus.
931. The device of claim 852 wherein the agent is everolimus or an analogue or derivative thereof.
932. The device of claim 852 wherein the agent is tacrolimus or an analogue or derivative thereof.
933. The device of claim 852 wherein the agent is not tacrolimus.
934. The device of claim 852 wherein the agent is biolmus or an analogue or derivative thereof.
935. The device of claim 852 wherein the agent is tresperimus or an analogue or derivative thereof.
936. The device of claim 852 wherein the agent is auranofin or an analogue or derivative thereof.
937. The device of claim 852 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
938. The device of claim 852 wherein the agent is gusperimus or an analogue or derivative thereof.
939. The device of claim 852 wherein the agent is pimecrolimus or an analogue or derivative thereof.
940. The device of claim 852 wherein the agent is ABT-578 or an analogue or derivative thereof.
941. The device of claim 852 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
942. The device of claim 852 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
943. The device of claim 852 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
944. The device of claim 852 wherein the agent is a leukotriene inhibitor.
945. The device of claim 852 wherein the agent is a MCP-1 antagonist.
946. The device of claim 852 wherein the agent is a MMP inhibitor.
947. The device of claim 852 wherein the agent is an NF kappa B inhibitor.
948. The device of claim 852 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
949. The device of claim 852 wherein the agent is an NO antagonist.
950. The device of claim 852 wherein the agent is a p38 MAP kinase inhibitor.
951. The device of claim 852 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
952. The device of claim 852 wherein the agent is a phosphodiesterase inhibitor.
953. The device of claim 852 wherein the agent is a TGF beta inhibitor.
954. The device of claim 852 wherein the agent is a thromboxane A2 antagonist.
955. The device of claim 852 wherein the agent is a TNF alpha antagonist.
956. The device of claim 852 wherein the agent is a TACE inhibitor.
957. The device of claim 852 wherein the agent is a tyrosine kinase inhibitor.
958. The device of claim 852 wherein the agent is a vitronectin inhibitor.
959. The device of claim 852 wherein the agent is a fibroblast growth factor inhibitor.
960. The device of claim 852 wherein the agent is a protein kinase inhibitor.
961. The device of claim 852 wherein the agent is a PDGF receptor kinase inhibitor.
962. The device of claim 852 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
963. The device of claim 852 wherein the agent is a retinoic acid receptor antagonist.
964. The device of claim 852 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
965. The device of claim 852 wherein the agent is a fibrinogen antagonist.
966. The device of claim 852 wherein the agent is an antimycotic agent.
967. The device of claim 852 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
968. The device of claim 852 wherein the agent is a bisphosphonate.
969. The device of claim 852 wherein the agent is a phospholipase A1 inhibitor.
970. The device of claim 852 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
971. The device of claim 852 wherein the agent is a macrolide antibiotic.
972. The device of claim 852 wherein the agent is a GPIIb/llla receptor antagonist.
973. The device of claim 852 wherein the agent is an endothelin receptor antagonist.
974. The device of claim 852 wherein the agent is a peroxisome proliferator-activated receptor agonist.
975. The device of claim 852 wherein the agent is an estrogen receptor agent.
976. The device of claim 852 wherein the agent is a somastostatin analogue.
977. The device of claim 852 wherein the agent is a neurokinin 1 antagonist.
978. The device of claim 852 wherein the agent is a neurokinin 3 antagonist.
979. The device of claim 852 wherein the agent is a VLA-4 antagonist.
980. The device of claim 852 wherein the agent is an osteoclast inhibitor.
981. The device of claim 852 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
982. The device of claim 852 wherein the agent is an angiotensin I converting enzyme inhibitor.
983. The device of claim 852 wherein the agent is an angiotensin II antagonist.
984. The device of claim 852 wherein the agent is an enkephalinase inhibitor.
985. The device of claim 852 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
986. The device of claim 852 wherein the agent is a protein kinase C inhibitor.
987. The device of claim 852 wherein the agent is a ROCK (rho- associated kinase) inhibitor.
988. The device of claim 852 wherein the agent is a CXCR3 inhibitor.
989. The device of claim 852 wherein the agent is an Itk inhibitor.
990. The device of claim 852 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
991. The device of claim 852 wherein the agent is a PPAR agonist
992. The device of claim 852 wherein the agent is an immunosuppressant.
993. The device of claim 852 wherein the agent is an Erb inhibitor.
994. The device of claim 852 wherein the agent is an apoptosis agonist.
995. The device of claim 852 wherein the agent is a lipocortin agonist.
996. The device of claim 852 wherein the agent is a VCAM-1 antagonist.
997. The device of claim 852 wherein the agent is a collagen antagonist.
998. The device of claim 852 wherein the agent is an alpha 2 integrin antagonist.
999. The device of claim 852 wherein the agent is a TNF alpha inhibitor.
1000. The device of claim 852 wherein the agent is a nitric oxide inhibitor
1001. The device of claim 852 wherein the agent is a cathepsin inhibitor.
1002. The device of claim 852 wherein the agent is not an anti- inflammatory agent.
1003. The device of claim 852 wherein the agent is not a steroid.
1004. The device of claim 852 wherein the agent is not a glucocorticosteroid.
1005. The device of claim 852 wherein the agent is not dexamethasone.
1006. The device of claim 852 wherein the agent is not an anti- infective agent.
1007. The device of claim 852 wherein the agent is not an antibiotic.
1008. The device of claim 852 wherein the agent is not an anti- fugal agent
1009. The device of claim 852 wherein the agent is not beclomethasone.
1010. The device of claim 852 wherein the agent is not dipropionate.
1011. The device of claim 852, further comprising a coating, wherein the coating comprises the anti-scarring agent and a polymer.
1012. The device of claim 852, further comprising a coating, wherein the coating comprises the anti-scarring agent.
1013. The device of claim 852, further comprising a coating, wherein the coating is disposed on a surface of the device.
1014. The device of claim 852, further comprising a coating, wherein the coating directly contacts the device.
1015. The device of claim 852, further comprising a coating, wherein the coating indirectly contacts the device.
1016. The device of claim 852, further comprising a coating, wherein the coating partially covers the device.
1017. The device of claim 852, further comprising a coating, wherein the coating completely covers the device.
1018. The device of claim 852, further comprising a coating, wherein the coating is a uniform coating.
1019. The device of claim 852, further comprising a coating, wherein the coating is a non-uniform coating.
1020. The device of claim 852, further comprising a coating, wherein the coating is a discontinuous coating.
1021. The device of claim 852, further comprising a coating, wherein the coating is a patterned coating.
1022. The device of claim 852, further comprising a coating, wherein the coating has a thickness of 100 μm or less.
1023. The device of claim 852, further comprising a coating, wherein the coating has a thickness of 10 μm or less.
1024. The device of claim 852, further comprising a coating, wherein the coating adheres to the surface of the device upon deployment of the device.
1025. The device of claim 852, further comprising a coating, wherein the coating is stable at room temperature for a period of 1 year.
1026. The device of claim 852, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001 % to about 1% by weight.
1027. The device of claim 852, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight
1028. The device of claim 852, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight
1029. The device of claim 852, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
1030. The device of claim 852, further comprising a coating, wherein the coating further comprises a polymer.
1031. The device of claim 852, further comprising a first coating having a first composition and the second coating having a second composition.
1032. The device of claim 852, further comprising a first coating having a first composition and the second coajing having a second composition, wherein the first composition and the second composition are different.
1033. The device of claim 852, further comprising a polymer.
1034. The device of claim 852, further comprising a polymeric carrier.
1035. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a copolymer.
1036. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a block copolymer.
1037. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a random copolymer.
1038. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a biodegradable polymer.
1039. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-biodegradable polymer.
1040. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophilic polymer.
1041. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophobic polymer.
1042. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophilic domains.
1043. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophobic domains.
1044. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-conductive polymer.
1045. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises an elastomer.
1046. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrogel.
1047. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a silicone polymer.
1048. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrocarbon polymer.
1049. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a styrene-derived polymer.
1050. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a butadiene polymer.
1051. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a macromer.
1052. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
1053. The device of claim 852, further comprising a polymeric carrier, wherein the polymeric carrier comprises an amorphous polymer.
1054. The device of claim 852, further comprising a lubricious coating.
1055. The device of claim 852 wherein the anti-scarring agent is located within pores or holes of the device.
1056. The device of claim 852 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
1057. The device of claim 852, further comprising a second pharmaceutically active agent.
1058. The device of claim 852, further comprising an anti- inflammatory agent.
1059. The device of claim 852, further comprising an agent that inhibits infection.
1060. The device of claim 852, further comprising an agent that inhibits infection, wherein the agent is an anthracycline.
1061. The device of claim 852, further comprising an agent that inhibits infection, wherein the agent is doxorubicin.
1062. The device of claim 852, further comprising an agent that inhibits infection, wherein the agent is mitoxantrone.
1063. The device of claim 852, further comprising an agent that inhibits infection, wherein the agent is a fluoropyrimidine.
1064. The device of claim 852, further comprising an agent that inhibits infection, wherein the agent is 5-fluorouracil (5-FU).
1065. The device of claim 852, further comprising an agent that inhibits infection, wherein the agent is a folic acid antagonist.
1066. The device of claim 852, further comprising an agent that inhibits infection, wherein the agent is methotrexate.
1067. The device of claim 852, further comprising an agent that inhibits infection, wherein the agent is a podophylotoxin.
1068. The device of claim 852, further comprising an agent that inhibits infection, wherein the agent is etoposide.
1069. The device of claim 852, further comprising an agent that inhibits infection, wherein the agent is a camptothecin.
1070. The device of claim 852, further comprising an agent that inhibits infection, wherein the agent is a hydroxyurea.
1071. The device of claim 852, further comprising an agent that inhibits infection, wherein the agent is a platinum complex.
1072. The device of claim 852, further comprising an agent that inhibits infection, wherein the agent is cisplatin.
1073. The device of claim 852, further comprising an anti- thrombotic agent
1074. The device of claim 852, further comprising a visualization agent.
1075. The device of claim 852, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
1076. The device of claim 852, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises barium, tantalum, or technetium.
1077. The device of claim 852, further comprising a visualization agent, wherein the visualization agent is a MRI responsive material.
1078. The device of claim 852, further comprising a visualization agent, wherein the visualization agent comprises a gadolinium chelate.
1079. The device of claim 852, further comprising a visualization agent, wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
1080. The device of claim 852, further comprising a visualization agent, wherein the visualization agent comprises an iron oxide compound.
1081. The device of claim 852, further comprising a visualization agent, wherein the visualization agent comprises a dye, pigment, or colorant.
1082. The device of claim 852, further comprising an echogenic material.
1083. The device of claim 852, further comprising an echogenic material, wherein the echogenic material is in the form of a coating.
1084. The device of claim 852 wherein the device is sterile.
1085. The device of claim 852 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
1086. The device of claim 852 wherein the device is adapted for delivering the anti-scarring agent locally to tissue proximate to the device.
1087. The device of claim 852 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
1088. The device of claim 852 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is connective tissue.
1089. The device of claim 852 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is muscle tissue.
1090. The device of claim 852 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is nerve tissue.
1091. The device of claim 852 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is epithelium tissue.
1092. The device of claim 852 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
1093. The device of claim 852 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
1094. The device of claim 852 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
1095. The device of claim 852 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
1096. The device of claim 852 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
1097. The device of claim 852 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
1098. The device of claim 852 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
1099. The device of claim 852 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
1100. The device of claim 852 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
1101. The device of claim 852 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
1102. The device of claim 852 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent
1103. The device of claim 852 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
1104. The device of claim 852 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
1105. The device of claim 852 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1106. The device of claim 852 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1107. The device of claim 852 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1108. The device of claim 852 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1109. The device of claim 852 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1110. The device of claim 852 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1111. The device of claim 852 wherein the agent or the composition is affixed to the sensor.
1112. The device of claim 852 wherein the agent or the composition is covalently attached to the sensor.
1113. The device of claim 852 wherein the agent or the composition is non-covalently attached to the sensor.
1114. The device of claim 852 further comprising a coating that absorbs the agent or the composition.
1115. The device of claim 852 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
1116. The device of claim 852 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
1117. The device of claim 852 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
1118. The device of claim 852 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
1119. The device of claim 852 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
1120. The device of claim 852 further comprising a pump that is linked to the sensor.
1121. The device of any one of claims 852-1120 wherein the device monitors cardiac output.
1122. The device of any one of claims 852-1120 wherein the device monitors ejection fraction.
1123. The device of any one of claims 852-1120 wherein the device monitors blood pressure in a heart chamber.
1124. The device of any one of claims 852-1120 wherein the device monitors ventricular wall motions.
1125. The device of any one of claims 852-1120 wherein the device monitors blood flow to a transplanted organ.
1126. The device of any one of claims 852-1120 wherein the device monitors heart rate.
1127. A device, comprising a respiratory sensor and an anti- scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
1128. The device of claim 1127 wherein the agent inhibits cell regeneration.
1129. The device of claim 1127 wherein the agent inhibits angiogenesis.
1130. The device of claim 1127 wherein the agent inhibits fibroblast migration.
1131. The device of claim 1127 wherein the agent inhibits fibroblast proliferation.
1132. The device of claim 1127 wherein the agent inhibits deposition of extracellular matrix.
1133. The device of claim 1127 wherein the agent inhibits tissue remodeling.
1134. The device of claim 1127 wherein the agent is an angiogenesis inhibitor.
1135. The device of claim 1127 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
1136. The device of claim 1127 wherein the agent is a chemokine receptor antagonist.
1137. The device of claim 1127 wherein the agent is a cell cycle inhibitor.
1138. The device of claim 1127 wherein the agent is a taxane.
1139. The device of claim 1127 wherein the agent is an antimicrotubule agent.
1140. The device of claim 1127 wherein the agent is paclitaxel.
1141. The device of claim 1127 wherein the agent is not paclitaxel.
1142. The device of claim 1127 wherein the agent is an analogue or derivative of paclitaxel.
1143. The device of claim 1127 wherein the agent is a vinca alkaloid.
1144. The device of claim 1127 wherein the agent is camptothecin or an analogue or derivative thereof.
1145. The device of claim 1127 wherein the agent is a podophyllotoxin.
1146. The device of claim 1127 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
1147. The device of claim 1127 wherein the agent is an anthracycline.
1148. The device of claim 1127 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
1149. The device of claim 1127 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
1150. The device of claim 1127 wherein the agent is a platinum compound.
1151. The device of claim 1127 wherein the agent is a nitrosourea.
1152. The device of claim 1127 wherein the agent is a nitroimidazole.
1153. The device of claim 1127 wherein the agent is a folic acid antagonist.
1154. The device of claim 1127 wherein the agent is a cytidine analogue.
1155. The device of claim 1127 wherein the agent is a pyrimidine analogue.
1156. The device of claim 1127 wherein the agent is a fluoropyrimidine analogue.
1157. The device of claim 1127 wherein the agent is a purine analogue.
1158. The device of claim 1127 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
1159. The device of claim 1127 wherein the agent is a hydroxyurea.
1160. The device of claim 1127 wherein the agent is a mytomicin or an analogue or derivative thereof.
1161. The device of claim 1127 wherein the agent is an alkyl sulfonate.
1162. The device of claim 1127 wherein the agent is a benzamide or an analogue or derivative thereof.
1163. The device of claim 1127 wherein the agent is a nicotinamide or an analogue or derivative thereof.
1164. The device of claim 1127 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
1165. The device of claim 1127 wherein the agent is a DNA alkylating agent.
1166. The device of claim 1127 wherein the agent is an antimicrotubule agent.
1167. The device of claim 1127 wherein the agent is a topoisomerase inhibitor.
1168. The device of claim 1127 wherein the agent is a DNA cleaving agent.
1169. The device of claim 1127 wherein the agent is an antimetabolite.
1170. The device of claim 1127 wherein the agent inhibits adenosine deaminase.
1171. The device of claim 1127 wherein the agent inhibits purine ring synthesis.
1172. The device of claim 1127 wherein the agent is a nucleotide interconversion inhibitor.
1173. The device of claim 1127 wherein the agent inhibits dihydrofolate reduction.
\ 1174. The device of claim 1127 wherein the agent blocks thymidine monophosphate.
1175. The device of claim 1127 wherein the agent causes DNA damage.
1176. The device of claim 1127 wherein the agent is a DNA intercalation agent.
1177. The device of claim 1127 wherein the agent is a RNA synthesis inhibitor.
1178. The device of claim 1127 wherein the agent is a pyrimidine synthesis inhibitor.
1179. The device of claim 1127 wherein the agent inhibits ribonucleotide synthesis or function.
1180. The device of claim 1127 wherein the agent inhibits thymidine monophosphate synthesis or function.
1181. The device of claim 1127 wherein the agent inhibits DNA synthesis.
1182. The device of claim 1127 wherein the agent causes DNA adduct formation.
1183. The device of claim 1127 wherein the agent inhibits protein synthesis.
1184. The device of claim 1127 wherein the agent inhibits microtubule function.
1185. The device of claim 1127 wherein the agent is a cyclin dependent protein kinase inhibitor.
1186. The device of claim 1127 wherein the agent is an epidermal growth factor kinase inhibitor.
1187. The device of claim 1127 wherein the agent is an elastase inhibitor.
1188. The device of claim 1127 wherein the agent is a factor Xa inhibitor.
1189. The device of claim 1127 wherein the agent is a farnesyltransferase inhibitor.
1190. The device of claim 1127 wherein the agent is a fibrinogen antagonist.
1191. The device of claim 1127 wherein the agent is a guanylate cyclase stimulant.
1192. The device of claim 1127 wherein the agent is a heat shock protein 90 antagonist.
1193. The device of claim 1127 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
1194. The device of claim 1127 wherein the agent is a guanylate cyclase stimulant.
1195. The device of claim 1127 wherein the agent is a HMGCoA reductase inhibitor.
1196. The device of claim 1127 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
1197. The device of claim 1127 wherein the agent is a hydroorotate dehydrogenase inhibitor.
1198. The device of claim 1127 wherein the agent is an IKK2 inhibitor.
1199. The device of claim 1127 wherein the agent is an IL-1 antagonist.
1200. The device of claim 1127 wherein the agent is an ICE antagonist.
1201. The device of claim 1127 wherein the agent is an IRAK antagonist.
1202. The device of claim 1127 wherein the agent is an IL-4 agonist.
1203. The device of claim 1127 wherein the agent is an immunomodulatory agent.
1204. The device of claim 1127 wherein the agent is sirolimus or an analogue or derivative thereof.
1205. The device of claim 1127 wherein the agent is not sirolimus.
1206. The device of claim 1127 wherein the agent is everolimus or an analogue or derivative thereof.
1207. The device of claim 1127 wherein the agent is tacrolimus or an analogue or derivative thereof.
1208. The device of claim 1127 wherein the agent is not tacrolimus.
1209. The device of claim 1127 wherein the agent is biolmus or an analogue or derivative thereof.
1210. The device of claim 1127 wherein the agent is tresperimus or an analogue or derivative thereof.
1211. The device of claim 1127 wherein the agent is auranofin or an analogue or derivative thereof.
1212. The device of claim 1127 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
1213. The device of claim 1127 wherein the agent is gusperimus or an analogue or derivative thereof.
1214. The device of claim 1127 wherein the agent is pimecrolimus or an analogue or derivative thereof.
1215. The device of claim 1127 wherein the agent is ABT-578 or an analogue or derivative thereof.
1216. The device of claim 1127 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
1217. The device of claim 1127 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
1218. The device of claim 1127 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
1219. The device of claim 1127 wherein the agent is a leukotriene inhibitor.
1220. The device of claim 1127 wherein the agent is a MCP-1 antagonist.
1221. The device of claim 1127 wherein the agent is a MMP inhibitor.
1222. The device of claim 1127 wherein the agent is an NF kappa B inhibitor.
1223. The device of claim 1127 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
1224. The device of claim 1127 wherein the agent is an NO antagonist.
1225. The device of claim 1127 wherein the agent is a p38 MAP kinase inhibitor.
1226. The device of claim 1127 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
1227. The device of claim 1127 wherein the agent is a phosphodiesterase inhibitor.
1228. The device of claim 1127 wherein the agent is a TGF beta inhibitor.
1229. The device of claim 1127 wherein the agent is a thromboxane A2 antagonist.
1230. The device of claim 1127 wherein the agent is a TNF alpha antagonist.
1231. The device of claim 1127 wherein the agent is a TACE inhibitor.
1232. The device of claim 1127 wherein the agent is a tyrosine kinase inhibitor.
1233. The device of claim 1127 wherein the agent is a vitronectin inhibitor.
1234. The device of claim 1127 wherein the agent is a fibroblast growth factor inhibitor.
1235. The device of claim 1127 wherein the agent is a protein kinase inhibitor.
1236. The device of claim 1127 wherein the agent is a PDGF receptor kinase inhibitor.
1237. The device of claim 1127 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
1238. The device of claim 1127 wherein the agent is a retinoic acid receptor antagonist.
1239. The device of claim 1127 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
1240. The device of claim 1127 wherein the agent is a fibrinogen antagonist.
1241. The device of claim 1127 wherein the agent is an antimycotic agent.
1242. The device of claim 1127 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
1243. The device of claim 1127 wherein the agent is a bisphosphonate.
1244. The device of claim 1127 wherein the agent is a phospholipase A1 inhibitor.
1245. The device of claim 1127 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
1246. The device of claim 1127 wherein the agent is a macrolide antibiotic.
1247. The device of claim 1127 wherein the agent is a GPIIb/llla receptor antagonist.
1248. The device of claim 1127 wherein the agent is an endothelin receptor antagonist.
1249. The device of claim 1127 wherein the agent is a peroxisome proliferator-activated receptor agonist.
1250. The device of claim 1127 wherein the agent is an estrogen receptor agent.
1251. The device of claim 1127 wherein the agent is a somastostatin analogue.
1252. The device of claim 1127 wherein the agent is a neurokinin 1 antagonist.
1253. The device of claim 1127 wherein the agent is a neurokinin 3 antagonist.
1254. The device of claim 1127 wherein the agent is a VLA-4 antagonist
1255. The device of claim 1127 wherein the agent is an osteoclast inhibitor.
1256. The device of claim 1127 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
1257. The device of claim 1127 wherein the agent is an angiotensin I converting enzyme inhibitor.
1258. The device of claim 1127 wherein the agent is an angiotensin II antagonist.
1259. The device of claim 1127 wherein the agent is an enkephalinase inhibitor.
1260. The device of claim 1127 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
1261. The device of claim 1127 wherein the agent is a protein kinase C inhibitor.
1262. The device of claim 1127 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
1263. The device of claim 1127 wherein the agent is a CXCR3 inhibitor.
1264. The device of claim 1127 wherein the agent is an Itk inhibitor.
1265. The device of claim 1127 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
1266. The device of claim 1127 wherein the agent is a PPAR agonist.
1267. The device of claim 1127 wherein the agent is an immunosuppressant.
1268. The device of claim 1127 wherein the agent is an Erb inhibitor.
1269. The device of claim 1127 wherein the agent is an apoptosis agonist.
1270. The device of claim 1127 wherein the agent is a lipocortin agonist.
1271. The device of claim 1127 wherein the agent is a VCAM-1 antagonist.
1272. The device of claim 1127 wherein the agent is a collagen antagonist.
1273. The device of claim 1127 wherein the agent is an alpha 2 integrin antagonist.
1274. The device of claim 1127 wherein the agent is a TNF alpha inhibitor.
1275. The device of claim 1127 wherein the agent is a nitric oxide inhibitor
1276. The device of claim 1127 wherein the agent is a cathepsin inhibitor.
1277. The device of claim 1127 wherein the agent is not an anti- inflammatory agent.
1278. The device of claim 1127 wherein the agent is not a steroid.
1279. The device of claim 1127 wherein the agent is not a glucocorticosteroid.
1280. The device of claim 1127 wherein the agent is not dexamethasone.
1281. The device of claim 1127 wherein the agent is not an anti- infective agent.
1282. The device of claim 1127 wherein the agent is not an antibiotic.
1283. The device of claim 1127 wherein the agent is not an anti- fugal agent.
1284. The device of claim 1127 wherein the agent is not beclomethasone.
1285. The device of claim 1127 wherein the agent is not dipropionate.
1286. The device of claim 1127, further comprising a coating, wherein the coating comprises the anti-scarring agent and a polymer.
1287. The device of claim 1127, further comprising a coating, wherein the coating comprises the anti-scarring agent.
1288. The device of claim 1127, further comprising a coating, wherein the coating is disposed on a surface of the device.
1289. The device of claim 1127, further comprising a coating, wherein the coating directly contacts the device.
1290. The device of claim 1127, further comprising a coating, wherein the coating indirectly contacts the device.
1291. The device of claim 1127, further comprising a coating, wherein the coating partially covers the device.
1292. The device of claim 1127, further comprising a coating, wherein the coating completely covers the device.
1293. The device of claim 1127, further comprising a coating, wherein the coating is a uniform coating.
1294. The device of claim 1127, further comprising a coating, wherein the coating is a non-uniform coating.
1295. The device of claim 1127, further comprising a coating, wherein the coating is a discontinuous coating.
1296. The device of claim 1127, further comprising a coating, wherein the coating is a patterned coating.
1297. The device of claim 1127, further comprising a coating, wherein the coating has a thickness of 100 μm or less.
1298. The device of claim 1127, further comprising a coating, wherein the coating has a thickness of 10 μm or less.
1299. The device of claim 1127, further comprising a coating, wherein the coating adheres to the surface of the device upon deployment of the device.
1300. The device of claim 1127, further comprising a coating, wherein the coating is stable at room temperature for a period of 1 year.
1301. The device of claim 1127, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1% by weight.
1302. The device of claim 1127, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
1303. The device of claim 1127, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
1304. The device of claim 1127, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
1305. The device of claim 1127, further comprising a coating, wherein the coating further comprises a polymer.
1306. The device of claim 1127, further comprising a first coating having a first composition and the second coating having a second composition.
1307. The device of claim 1127, further comprising a first coating having a first composition and the second coating having a second composition, wherein the first composition and the second composition are different.
1308. The device of claim 1127, further comprising a polymer.
1309. The device of claim 1127, further comprising a polymeric carrier.
1310. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a copolymer.
1311. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a block copolymer.
1312. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a random copolymer.
1313. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a biodegradable polymer.
1314. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-biodegradable polymer.
1315. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophilic polymer.
1316. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophobic polymer.
1317. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophilic domains.
1318. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophobic domains.
1319. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-conductive polymer.
1320. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises an elastomer.
1321. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrogel.
1322. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a silicone polymer.
1323. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrocarbon polymer.
1324. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a styrene-derived polymer.
1325. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a butadiene polymer.
1326. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a macromer.
1327. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises a poly( ethylene glycol) polymer.
1328. The device of claim 1127, further comprising a polymeric carrier, wherein the polymeric carrier comprises an amorphous polymer.
1329. The device of claim 1127, further comprising a lubricious coating.
1330. The device of claim 1127 wherein the anti-scarring agent is located within pores or holes of the device.
1331. The device of claim 1127 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
1332. The device of claim 1127, further comprising a second pharmaceutically active agent
1333. The device of claim 1127, further comprising an anti- inflammatory agent.
1334. The device of claim 1127, further comprising an agent that inhibits infection.
1335. The device of claim 1127, further comprising an agent that inhibits infection, wherein the agent is an anthracycline.
1336. The device of claim 1127, further comprising an agent that inhibits infection, wherein the agent is doxorubicin.
1337. The device of claim 1127, further comprising an agent that inhibits infection, wherein the agent is mitoxantrone.
1338. The device of claim 1127, further comprising an agent that inhibits infection, wherein the agent is a fluoropyrimidine.
1339. The device of claim 1127, further comprising an agent that inhibits infection, wherein the agent is 5-fluorouracil (5-FU).
1340. The device of claim 1127, further comprising an agent that inhibits infection, wherein the agent is a folic acid antagonist.
1341. The device of claim 1127, further comprising an agent that inhibits infection, wherein the agent is methotrexate.
1342. The device of claim 1127, further comprising an agent that inhibits infection, wherein the agent is a podophylotoxin.
1343. The device of claim 1127, further comprising an agent that inhibits infection, wherein the agent is etoposide.
1344. The device of claim 1127, further comprising an agent that inhibits infection, wherein the agent is a camptothecin.
1345. The device of claim 1127, further comprising an agent that inhibits infection, wherein the agent is a hydroxyurea.
1346. The device of claim 1127, further comprising an agent that inhibits infection, wherein the agent is a platinum complex.
1347. The device of claim 1127, further comprising an agent that inhibits infection, wherein the agent is cisplatin.
1348. The device of claim 1127, further comprising an anti- thrombotic agent.
1349. The device of claim 1127, further comprising a visualization agent.
1350. The device of claim 1127, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
1351. The device of claim 1127, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises barium, tantalum, or technetium.
1352. The device of claim 1127, further comprising a visualization agent, wherein the visualization agent is a MRI responsive material.
1353. The device of claim 1127, further comprising a visualization agent, wherein the visualization agent comprises a gadolinium chelate.
1354. The device of claim 1127, further comprising a visualization agent, wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
1355. The device of claim 1127, further comprising a visualization agent, wherein the visualization agent comprises an iron oxide compound.
1356. The device of claim 1127, further comprising a visualization agent, wherein the visualization agent comprises a dye, pigment, or colorant.
1357. The device of claim 1127, further comprising an echogenic material.
1358. The device of claim 1127, further comprising an echogenic material, wherein the echogenic material is in the form of a coating.
1359. The device of claim 1127 wherein the device is sterile.
1360. The device of claim 1127 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
1361. The device of claim 1127 wherein the device is adapted for delivering the anti-scarring agent locally to tissue proximate to the device.
1362. The device of claim 1127 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
1363. The device of claim 1127 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is connective tissue.
1364. The device of claim 1127 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is muscle tissue.
1365. The device of claim 1127 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is nerve tissue.
1366. The device of claim 1127 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is epithelium tissue.
1367. The device of claim 1127 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
1368. The device of claim 1127 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
1369. The device of claim 1127 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
1370. The device of claim 1127 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
1371. The device of claim 1127 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
1372. The device of claim 1127 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
1373. The device of claim 1127 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
1374. The device of claim 1127 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
1375. The device of claim 1127 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
1376. The device of claim 1127 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
1377. The device of claim 1127 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
1378. The device of claim 1127 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
1379. The device of claim 1127 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
1380. The device of claim 1127 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1381. The device of claim 1127 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1382. The device of claim 1127 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1383. The device of claim 1127 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1384. The device of claim 1127 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1385. The device of claim 1127 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1386. The device of claim 1127 wherein the agent or the composition is affixed to the sensor.
1387. The device of claim 1127 wherein the agent or the composition is covalently attached to the sensor.
1388. The device of claim 1127 wherein the agent or the composition is non-covalently attached to the sensor.
1389. The device of claim 1127 further comprising a coating that absorbs the agent or the composition.
1390. The device of claim 1127 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
1391. The device of claim 1127 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
1392. The device of claim 1127 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
1393. The device of claim 1127 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
1394. The device of claim 1127 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
1395. The device of claim 1127 further comprising a pump that is linked to the sensor.
1396. The device of claims 1127-1395 wherein the device monitors pulmonary functions.
1397. A device, comprising an auditory sensor and an anti- scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
1398. The device of claim 1397 wherein the agent inhibits cell regeneration.
1399. The device of claim 1397 wherein the agent inhibits angiogenesis.
1400. The device of claim 1397 wherein the agent inhibits fibroblast migration.
1401. The device of claim 1397 wherein the agent inhibits fibroblast proliferation.
1402. The device of claim 1397 wherein the agent inhibits deposition of extracellular matrix.
1403. The device of claim 1397 wherein the agent inhibits tissue remodeling.
1404. The device of claim 1397 wherein the agent is an angiogenesis inhibitor.
1405. The device of claim 1397 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
1406. The device of claim 1397 wherein the agent is a chemokine receptor antagonist.
1407. The device of claim 1397 wherein the agent is a cell cycle inhibitor.
1408. The device of claim 1397 wherein the agent is a taxane.
1409. The device of claim 1397 wherein the agent is an antimicrotubule agent.
1410. The device of claim 1397 wherein the agent is paclitaxel.
1411. The device of claim 1397 wherein the agent is not paclitaxel.
1412. The device of claim 1397 wherein the agent is an analogue or derivative of paclitaxel.
1413. The device of claim 1397 wherein the agent is a vinca alkaloid.
1414. The device of claim 1397 wherein the agent is camptothecin or an analogue or derivative thereof.
1415. The device of claim 1397 wherein the agent is a podophyllotoxin.
1416. The device of claim 1397 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
1417. The device of claim 1397 wherein the agent is an anthracycline.
1418. The device of claim 1397 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
1419. The device of claim 1397 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
1420. The device of claim 1397 wherein the agent is a platinum compound.
1421. The device of claim 1397 wherein the agent is a nitrosourea.
1422. The device of claim 1397 wherein the agent is a nitroimidazole.
1423. The device of claim 1397 wherein the agent is a folic acid antagonist.
1424. The device of claim 1397 wherein the agent is a cytidine analogue.
1425. The device of claim 1397 wherein the agent is a pyrimidine analogue.
1426. The device of claim 1397 wherein the agent is a fluoropyrimidine analogue.
1427. The device of claim 1397 wherein the agent is a purine analogue.
1428. The device of claim 1397 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
1429. The device of claim 1397 wherein the agent is a hydroxyurea.
1430. The device of claim 1397 wherein the agent is a mytomicin or an analogue or derivative thereof.
1431. The device of claim 1397 wherein the agent is an alkyl sulfonate.
1432. The device of claim 1397 wherein the agent is a benzamide or an analogue or derivative thereof.
1433. The device of claim 1397 wherein the agent is a nicotinamide or an analogue or derivative thereof.
1434. The device of claim 1397 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
1435. The device of claim 1397 wherein the agent is a DNA alkylating agent.
1436. The device of claim 1397 wherein the agent is an antimicrotubule agent.
1437. The device of claim 1397 wherein the agent is a topoisomerase inhibitor.
1438. The device of claim 1397 wherein the agent is a DNA cleaving agent.
1439. The device of claim 1397 wherein the agent is an antimetabolite.
1440. The device of claim 1397 wherein the agent inhibits adenosine deaminase.
1441. The device of claim 1397 wherein the agent inhibits purine ring synthesis.
1442. The device of claim 1397 wherein the agent is a nucleotide interconversion inhibitor.
1443. The device of claim 1397 wherein the agent inhibits dihydrofolate reduction.
1444. The device of claim 1397 wherein the agent blocks thymidine monophosphate.
1445. The device of claim 1397 wherein the agent causes DNA damage.
1446. The device of claim 1397 wherein the agent is a DNA intercalation agent.
1447. The device of claim 1397 wherein the agent is a RNA synthesis inhibitor.
1448. The device of claim 1397 wherein the agent is a pyrimidine synthesis inhibitor.
1449. The device of claim 1397 wherein the agent inhibits ribonucleotide synthesis or function.
1450. The device of claim 1397 wherein the agent inhibits thymidine monophosphate synthesis or function.
1451. The device of claim 1397 wherein the agent inhibits DNA synthesis.
1452. The device of claim 1397 wherein the agent causes DNA adduct formation.
1453. The device of claim 1397 wherein the agent inhibits protein synthesis.
1454. The device of claim 1397 wherein the agent inhibits microtubule function.
1455. The device of claim 1397 wherein the agent is a cyclin dependent protein kinase inhibitor.
1456. The device of claim 1397 wherein the agent is an epidermal growth factor kinase inhibitor.
1457. The device of claim 1397 wherein the agent is an elastase inhibitor.
1458. The device of claim 1397 wherein the agent is a factor Xa inhibitor.
1459. The device of claim 1397 wherein the agent is a farnesyltransferase inhibitor.
1460. The device of claim 1397 wherein the agent is a fibrinogen antagonist.
1461. The device of claim 1397 wherein the agent is a guanylate cyclase stimulant.
1462. The device of claim 1397 wherein the agent is a heat shock protein 90 antagonist.
1463. The device of claim 1397 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
1464. The device of claim 1397 wherein the agent is a guanylate cyclase stimulant.
1465. The device of claim 1397 wherein the agent is a HMGCoA reductase inhibitor.
1466. The device of claim 1397 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
1467. The device of claim 1397 wherein the agent is a hydroorotate dehydrogenase inhibitor.
1468. The device of claim 1397 wherein the agent is an IKK2 inhibitor.
1469. The device of claim 1397 wherein the agent is an IL-1 antagonist.
1470. The device of claim 1397 wherein the agent is an ICE antagonist.
1471. The device of claim 1397 wherein the agent is an IRAK antagonist.
1472. The device of claim 1397 wherein the agent is an IL-4 agonist.
1473. The device of claim 1397 wherein the agent is an immunomodulatory agent.
1474. The device of claim 1397 wherein the agent is sirolimus or an analogue or derivative thereof.
1475. The device of claim 1397 wherein the agent is not sirolimus.
1476. The device of claim 1397 wherein the agent is everolimus or an analogue or derivative thereof.
1477. The device of claim 1397 wherein the agent is tacrolimus or an analogue or derivative thereof.
1478. The device of claim 1397 wherein the agent is not tacrolimus.
1479. The device of claim 1397 wherein the agent is biolmus or an analogue or derivative thereof.
1480. The device of claim 1397 wherein the agent is tresperimus or an analogue or derivative thereof.
1481. The device of claim 1397 wherein the agent is auranofin or an analogue or derivative thereof.
1482. The device of claim 1397 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
1483. The device of claim 1397 wherein the agent is gusperimus or an analogue or derivative thereof.
1484. The device of claim 1397 wherein the agent is pimecrolimus or an analogue or derivative thereof.
1485. The device of claim 1397 wherein the agent is ABT-578 or an analogue or derivative thereof.
1486. The device of claim 1397 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
1487. The device of claim 1397 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
1488. The device of claim 1397 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
1489. The device of claim 1397 wherein the agent is a leukotriene inhibitor.
1490. The device of claim 1397 wherein the agent is a MCP-1 antagonist
1491. The device of claim 1397 wherein the agent is a MMP inhibitor.
1492. The device of claim 1397 wherein the agent is an NF kappa B inhibitor.
1493. The device of claim 1397 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
1494. The device of claim 1397 wherein the agent is an NO antagonist.
1495. The device of claim 1397 wherein the agent is a p38 MAP kinase inhibitor.
1496. The device of claim 1397 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
1497. The device of claim 1397 wherein the agent is a phosphodiesterase inhibitor.
1498. The device of claim 1397 wherein the agent is a TGF beta inhibitor.
1499. The device of claim 1397 wherein the agent is a thromboxane A2 antagonist.
1500. The device of claim 1397 wherein the agent is a TNF alpha antagonist
1501. The device of claim 1397 wherein the agent is a TACE inhibitor.
1502. The device of claim 1397 wherein the agent is a tyrosine kinase inhibitor.
1503. The device of claim 1397 wherein the agent is a vitronectin inhibitor.
1504. The device of claim 1397 wherein the agent is a fibroblast growth factor inhibitor.
1505. The device of claim 1397 wherein the agent is a protein kinase inhibitor.
1506. The device of claim 1397 wherein the agent is a PDGF receptor kinase inhibitor.
1507. The device of claim 1397 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
1508. The device of claim 1397 wherein the agent is a retinoic acid receptor antagonist.
1509. The device of claim 1397 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
1510. The device of claim 1397 wherein the agent is a fibrinogen antagonist.
1511. The device of claim 1397 wherein the agent is an antimycotic agent.
1512. The device of claim 1397 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
1513. The device of claim 1397 wherein the agent is a bisphosphonate.
1514. The device of claim 1397 wherein the agent is a phospholipase A1 inhibitor.
1515. The device of claim 1397 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
1516. The device of claim 1397 wherein the agent is a macrolide antibiotic.
1517. The device of claim 1397 wherein the agent is a GPIIb/llla receptor antagonist.
1518. The device of claim 1397 wherein the agent is an endothelin receptor antagonist
1519. The device of claim 1397 wherein the agent is a peroxisome proliferator-activated receptor agonist.
1520. The device of claim 1397 wherein the agent is an estrogen receptor agent.
1521. The device of claim 1397 wherein the agent is a somastostatin analogue.
1522. The device of claim 1397 wherein the agent is a neurokinin 1 antagonist
1523. The device of claim 1397 wherein the agent is a neurokinin 3 antagonist.
1524. The device of claim 1397 wherein the agent is a VLA-4 antagonist.
1525. The device of claim 1397 wherein the agent is an osteoclast inhibitor.
1526. The device of claim 1397 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
1527. The device of claim 1397 wherein the agent is an angiotensin I converting enzyme inhibitor.
1528. The device of claim 1397 wherein the agent is an angiotensin II antagonist.
1529. The device of claim 1397 wherein the agent is an enkephalinase inhibitor.
1530. The device of claim 1397 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
1531. The device of claim 1397 wherein the agent is a protein kinase C inhibitor.
1532. The device of claim 1397 wherein the agent is a ROCK (rho-associated kinase) inhibitor. I
1533. The device of claim 1397 wherein the agent is a CXCR3 inhibitor.
1534. The device of claim 1397 wherein the agent is an Itk inhibitor.
1535. The device of claim 1397 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
1536. The device of claim 1397 wherein the agent is a PPAR agonist.
1537. The device of claim 1397 wherein the agent is an immunosuppressant.
1538. The device of claim 1397 wherein the agent is an Erb inhibitor.
1539. The device of claim 1397 wherein the agent is an apoptosis agonist.
1540. The device of claim 1397 wherein the agent is a lipocortin agonist.
1541. The device of claim 1397 wherein the agent is a VCAM-1 antagonist.
1542. The device of claim 1397 wherein the agent is a collagen antagonist.
1543. The device of claim 1397 wherein the agent is an alpha 2 integrin antagonist
1544. The device of claim 1397 wherein the agent is a TNF alpha inhibitor.
1545. The device of claim 1397 wherein the agent is a nitric oxide inhibitor
1546. The device of claim 1397 wherein the agent is a cathepsin inhibitor.
1547. The device of claim 1397 wherein the agent is not an anti- inflammatory agent.
1548. The device of claim 1397 wherein the agent is not a steroid.
1549. The device of claim 1397 wherein the agent is not a glucocorticosteroid.
1550. The device of claim 1397 wherein the agent is not dexamethasone.
1551. The device of claim 1397 wherein the agent is not an anti- infective agent.
1552. The device of claim 1397 wherein the agent is not an antibiotic.
1553. The device of claim 1397 wherein the agent is not an anti- fugal agent.
1554. The device of claim 1397 wherein the agent is not beclomethasone.
1555. The device of claim 1397 wherein the agent is not dipropionate.
1556. The device of claim 1397, further comprising a coating, wherein the coating comprises the anti-scarring agent and a polymer.
1557. The device of claim 1397, further comprising a coating, wherein the coating comprises the anti-scarring agent.
1558. The device of claim 1397, further comprising a coating, wherein the coating is disposed on a surface of the device.
1559. The device of claim 1397, further comprising a coating, wherein the coating directly contacts the device.
1560. The device of claim 1397, further comprising a coating, wherein the coating indirectly contacts the device.
1561. The device of claim 1397, further comprising a coating, wherein the coating partially covers the device.
1562. The device of claim 1397, further comprising a coating, wherein the coating completely covers the device.
1563. The device of claim 1397, further comprising a coating, wherein the coating is a uniform coating.
1564. The device of claim 1397, further comprising a coating, wherein the coating is a non-uniform coating.
1565. The device of claim 1397, further comprising a coating, wherein the coating is a discontinuous coating.
1566. The device of claim 1397, further comprising a coating, wherein the coating is a patterned coating.
1567. The device of claim 1397, further comprising a coating, wherein the coating has a thickness of 100 μm or less.
1568. The device of claim 1397, further comprising a coating, wherein the coating has a thickness of 10 μm or less.
1569. The device of claim 1397, further comprising a coating, wherein the coating adheres to the surface of the device upon deployment of the device.
1570. The device of claim 1397, further comprising a coating, wherein the coating is stable at room temperature for a period of 1 year.
1571. The device of claim 1397, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001 % to about 1 % by weight.
1572. The device of claim 1397, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
1573. The device of claim 1397, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
1574. The device of claim 1397, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
1575. The device of claim 1397, further comprising a coating, wherein the coating further comprises a polymer.
1576. The device of claim 1397, further comprising a first coating having a first composition and the second coating having a second composition.
1577. The device of claim 1397, further comprising a first coating having a first composition and the second coating having a second composition, wherein the first composition and the second composition are different.
1578. The device of claim 1397, further comprising a polymer.
1579. The device of claim 1397, further comprising a polymeric carrier.
1580. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a copolymer.
1581. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a block copolymer.
1582. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a random copolymer.
1583. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a biodegradable polymer.
1584. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-biodegradable polymer.
1585. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophilic polymer.
1586. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophobic polymer.
1587. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophilic domains.
1588. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophobic domains.
1589. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-conductive polymer.
1590. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises an elastomer.
1591. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrogel.
1592. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a silicone polymer.
1593. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrocarbon polymer.
1594. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a styrene-derived polymer.
1595. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a butadiene polymer.
1596. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a macromer.
1597. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises a poly( ethylene glycol) polymer.
1598. The device of claim 1397, further comprising a polymeric carrier, wherein the polymeric carrier comprises an amorphous polymer.
1599. The device of claim 1397, further comprising a lubricious coating.
1600. The device of claim 1397 wherein the anti-scarring agent is located within pores or holes of the device.
1601. The device of claim 1397 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
1602. The device of claim 1397, further comprising a second pharmaceutically active agent.
1603. The device of claim 1397, further comprising an anti- inflammatory agent.
1604. The device of claim 1397, further comprising an agent that inhibits infection.
1605. The device of claim 1397, further comprising an agent that inhibits infection, wherein the agent is an anthracycline.
1606. The device of claim 1397, further comprising an agent that inhibits infection, wherein the agent is doxorubicin.
1607. The device of claim 1397, further comprising an agent that inhibits infection, wherein the agent is mitoxantrone.
1608. The device of claim 1397, further comprising an agent that inhibits infection, wherein the agent is a fluoropyrimidine.
1609. The device of claim 1397, further comprising an agent that inhibits infection, wherein the agent is 5-fluorouracil (5-FU).
1610. The device of claim 1397, further comprising an agent that inhibits infection, wherein the agent is a folic acid antagonist.
1611. The device of claim 1397, further comprising an agent that inhibits infection, wherein the agent is methotrexate.
1612. The device of claim 1397, further comprising an agent that inhibits infection, wherein the agent is a podophylotoxin.
1613. The device of claim 1397, further comprising an agent that inhibits infection, wherein the agent is etoposide.
1614. The device of claim 1397, further comprising an agent that inhibits infection, wherein the agent is a camptothecin.
1615. The device of claim 1397, further comprising an agent that inhibits infection, wherein the agent is a hydroxyurea.
1616. The device of claim 1397, further comprising an agent that inhibits infection, wherein the agent is a platinum complex.
1617. The device of claim 1397, further comprising an agent that inhibits infection, wherein the agent is cisplatin.
1618. The device of claim 1397, further comprising an anti- thrombotic agent.
1619. The device of claim 1397, further comprising a visualization agent.
1620. The device of claim 1397, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
1621. The device of claim 1397, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises barium, tantalum, or technetium.
1622. The device of claim 1397, further comprising a visualization agent, wherein the visualization agent is a MRI responsive material.
1623. The device of claim 1397, further comprising a visualization agent, wherein the visualization agent comprises a gadolinium chelate.
1624. The device of claim 1397, further comprising a visualization agent, wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
1625. The device of claim 1397, further comprising a visualization agent, wherein the visualization agent comprises an iron oxide compound.
1626. The device of claim 1397, further comprising a visualization agent, wherein the visualization agent comprises a dye, pigment, or colorant.
1627. The device of claim 1397, further comprising an echogenic material.
1628. The device of claim 1397, further comprising an echogenic material, wherein the echogenic material is in the form of a coating.
1629. The device of claim 1397 wherein the device is sterile.
1630. The device of claim 1397 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
1631. The device of claim 1397 wherein the device is adapted for delivering the anti-scarring agent locally to tissue proximate to the device.
1632. The device of claim 1397 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
1633. The device of claim 1397 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is connective tissue.
1634. The device of claim 1397 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is muscle tissue.
1635. The device of claim 1397 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is nerve tissue.
1636. The device of claim 1397 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is epithelium tissue.
1637. The device of claim 1397 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
1638. The device of claim 1397 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
1639. The device of claim 1397 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
1640. The device of claim 1397 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
1641. The device of claim 1397 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
1642. The device of claim 1397 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
1643. The device of claim 1397 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
1644. The device of claim 1397 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
1645. The device of claim 1397 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
1646. The device of claim 1397 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
1647. The device of claim 1397 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
1648. The device of claim 1397 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
1649. The device of claim 1397 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
1650. The device of claim 1397 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1651. The device of claim 1397 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1652. The device of claim 1397 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1653. The device of claim 1397 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1654. The device of claim 1397 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1655. The device of claim 1397 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1656. The device of claim 1397 wherein the agent or the composition is affixed to the sensor.
1657. The device of claim 1397 wherein the agent or the composition is covalently attached to the sensor.
1658. The device of claim 1397 wherein the agent or the composition is non-covalently attached to the sensor.
1659. The device of claim 1397 further comprising a coating that absorbs the agent or the composition.
1660. The device of claim 1397 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
1661. The device of claim 1397 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
1662. The device of claim 1397 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
1663. The device of claim 1397 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
1664. The device of claim 1397 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
1665. The device of claim 1397 further comprising a pump that is linked to the sensor.
1666. The device of any one of claims 1397-1665 wherein the device is adapted for delivering an electrical signal to an implantable electromechanical transducer that acts on the middle or inner ear.
1667. The device of any one of claims 1397-1665 wherein the device generates an electrical audio signal.
1668. The device of any one of claims 1397-1665 wherein the device is a capacitive sensor that is coupled to a vibrating auditory element.
1669. The device of any one of claims 1397-1665 wherein the device is an electromagnetic sensor.
1670. A device, comprising an electrolyte or metabolite sensor and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
1671. The device of claim 1670 wherein the agent inhibits cell regeneration.
1672. The device of claim 1670 wherein the agent inhibits angiogenesis.
1673. The device of claim 1670 wherein the agent inhibits fibroblast migration.
1674. The device of claim 1670 wherein the agent inhibits fibroblast proliferation.
1675. The device of claim 1670 wherein the agent inhibits deposition of extracellular matrix.
1676. The device of claim 1670 wherein the agent inhibits tissue remodeling.
1677. The device of claim 1670 wherein the agent is an angiogenesis inhibitor.
1678. The device of claim 1670 wherein the agent is a 5- lipoxygenase inhibitor or antagonist
1679. The device of claim 1670 wherein the agent is a chemokine receptor antagonist.
1680. The device of claim 1670 wherein the agent is a cell cycle inhibitor.
1681. The device of claim 1670 wherein the agent is a taxane.
1682. The device of claim 1670 wherein the agent is an antimicrotubule agent.
1683. The device of claim 1670 wherein the agent is paclitaxel.
1684. The device of claim 1670 wherein the agent is not paclitaxel.
1685. The device of claim 1670 wherein the agent is an analogue or derivative of paclitaxel.
1686. The device of claim 1670 wherein the agent is a vinca alkaloid.
1687. The device of claim 1670 wherein the agent is camptothecin or an analogue or derivative thereof.
1688. The device of claim 1670 wherein the agent is a podophyllotoxin.
1689. The device of claim 1670 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
1690. The device of claim 1670 wherein the agent is an anthracycline.
1691. The device of claim 1670 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
1692. The device of claim 1670 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
1693. The device of claim 1670 wherein the agent is a platinum compound.
1694. The device of claim 1670 wherein the agent is a nitrosourea.
1695. The device of claim 1670 wherein the agent is a nitroimidazole.
1696. The device of claim 1670 wherein the agent is a folic acid antagonist.
1697. The device of claim 1670 wherein the agent is a cytidine analogue.
1698. The device of claim 1670 wherein the agent is a pyrimidine analogue.
1699. The device of claim 1670 wherein the agent is a fluoropyrimidine analogue.
1700. The device of claim 1670 wherein the agent is a purine analogue.
1701. The device of claim 1670 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
1702. The device of claim 1670 wherein the agent is a hydroxyurea.
1703. The device of claim 1670 wherein the agent is a mytomicin or an analogue or derivative thereof.
1704. The device of claim 1670 wherein the agent is an alkyl sulfonate.
1705. The device of claim 1670 wherein the agent is a benzamide or an analogue or derivative thereof.
1706. The device of claim 1670 wherein the agent is a nicotinamide or an analogue or derivative thereof.
1707. The device of claim 1670 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
1708. The device of claim 1670 wherein the agent is a DNA alkylating agent.
1709. The device of claim 1670 wherein the agent is an antimicrotubule agent.
1710. The device of claim 1670 wherein the agent is a topoisomerase inhibitor.
1711. The device of claim 1670 wherein the agent is a DNA cleaving agent.
1712. The device of claim 1670 wherein the agent is an antimetabolite.
1713. The device of claim 1670 wherein the agent inhibits adenosine deaminase.
1714. The device of claim 1670 wherein the agent inhibits purine ring synthesis.
1715. The device of claim 1670 wherein the agent is a nucleotide interconversion inhibitor.
1716. The device of claim 1670 wherein the agent inhibits dihydrofolate reduction.
1717. The device of claim 1670 wherein the agent blocks thymidine monophosphate.
1718. The device of claim 1670 wherein the agent causes DNA damage.
1719. The device of claim 1670 wherein the agent is a DNA intercalation agent.
1720. The device of claim 1670 wherein the agent is a RNA synthesis inhibitor.
1721. The device of claim 1670 wherein the agent is a pyrimidine synthesis inhibitor.
1722. The device of claim 1670 wherein the agent inhibits ribonucleotide synthesis or function.
1723. The device of claim 1670 wherein the agent inhibits thymidine monophosphate synthesis or function.
1724. The device of claim 1670 wherein the agent inhibits DNA synthesis.
1725. The device of claim 1670 wherein the agent causes DNA adduct formation.
1726. The device of claim 1670 wherein the agent inhibits protein synthesis.
1727. The device of claim 1670 wherein the agent inhibits microtubule function.
1728. The device of claim 1670 wherein the agent is a cyclin dependent protein kinase inhibitor.
1729. The device of claim 1670 wherein the agent is an epidermal growth factor kinase inhibitor.
1730. The device of claim 1670 wherein the agent is an elastase inhibitor.
1731. The device of claim 1670 wherein the agent is a factor Xa inhibitor.
1732. The device of claim 1670 wherein the agent is a farnesyltransferase inhibitor.
1733. The device of claim 1670 wherein the agent is a fibrinogen antagonist.
1734. The device of claim 1670 wherein the agent is a guanylate cyclase stimulant.
1735. The device of claim 1670 wherein the agent is a heat shock protein 90 antagonist.
1736. The device of claim 1670 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
1737. The device of claim 1670 wherein the agent is a guanylate cyclase stimulant.
1738. The device of claim 1670 wherein the agent is a HMGCoA reductase inhibitor.
1739. The device of claim 1670 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
1740. The device of claim 1670 wherein the agent is a hydroorotate dehydrogenase inhibitor.
1741. The device of claim 1670 wherein the agent is an IKK2 inhibitor.
1742. The device of claim 1670 wherein the agent is an IL-1 antagonist
1743. The device of claim 1670 wherein the agent is an ICE antagonist.
1744. The device of claim 1670 wherein the agent is an IRAK antagonist.
1745. The device of claim 1670 wherein the agent is an IL-4 agonist.
1746. The device of claim 1670 wherein the agent is an immunomodulatory agent.
1747. The device of claim 1670 wherein the agent is sirolimus or an analogue or derivative thereof.
1748. The device of claim 1670 wherein the agent is not sirolimus.
1749. The device of claim 1670 wherein the agent is everolimus or an analogue or derivative thereof.
1750. The device of claim 1670 wherein the agent is tacrolimus or an analogue or derivative thereof.
1751. The device of claim 1670 wherein the agent is not tacrolimus.
1752. The device of claim 1670 wherein the agent is biolmus or an analogue or derivative thereof.
1753. The device of claim 1670 wherein the agent is tresperimus or an analogue or derivative thereof.
1754. The device of claim 1670 wherein the agent is auranofin or an analogue or derivative thereof.
1755. The device of claim 1670 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
1756. The device of claim 1670 wherein the agent is gusperimus or an analogue or derivative thereof.
1757. The device of claim 1670 wherein the agent is pimecrolimus or an analogue or derivative thereof.
1758. The device of claim 1670 wherein the agent is ABT-578 or an analogue or derivative thereof.
1759. The device of claim 1670 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
1760. The device of claim 1670 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
1761. The device of claim 1670 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
1762. The device of claim 1670 wherein the agent is a leukotriene inhibitor.
1763. The device of claim 1670 wherein the agent is a MCP-1 antagonist
1764. The device of claim 1670 wherein the agent is a MMP inhibitor.
1765. The device of claim 1670 wherein the agent is an NF kappa B inhibitor.
1766. The device of claim 1670 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
1767. The device of claim 1670 wherein the agent is an NO antagonist.
1768. The device of claim 1670 wherein the agent is a p38 MAP kinase inhibitor.
1769. The device of claim 1670 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
1770. The device of claim 1670 wherein the agent is a phosphodiesterase inhibitor.
1771. The device of claim 1670 wherein the agent is a TGF beta inhibitor.
1772. The device of claim 1670 wherein the agent is a thromboxane A2 antagonist.
1773. The device of claim 1670 wherein the agent is a TNF alpha antagonist.
1774. The device of claim 1670 wherein the agent is a TACE inhibitor.
1775. The device of claim 1670 wherein the agent is a tyrosine kinase inhibitor.
1776. The device of claim 1670 wherein the agent is a vitronectin inhibitor.
1777. The device of claim 1670 wherein the agent is a fibroblast growth factor inhibitor.
1778. The device of claim 1670 wherein the agent is a protein kinase inhibitor.
1779. The device of claim 1670 wherein the agent is a PDGF receptor kinase inhibitor.
1780. The device of claim 1670 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
1781. The device of claim 1670 wherein the agent is a retinoic acid receptor antagonist.
1782. The device of claim 1670 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
1783. The device of claim 1670 wherein the agent is a fibrinogen antagonist.
1784. The device of claim 1670 wherein the agent is an antimycotic agent.
1785. The device of claim 1670 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
1786. The device of claim 1670 wherein the agent is a bisphosphonate.
1787. The device of claim 1670 wherein the agent is a phospholipase A1 inhibitor.
1788. The device of claim 1670 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
1789. The device of claim 1670 wherein the agent is a macrolide antibiotic.
1790. The device of claim 1670 wherein the agent is a GPIIb/llla receptor antagonist.
1791. The device of claim 1670 wherein the agent is an endothelin receptor antagonist.
1792. The device of claim 1670 wherein the agent is a peroxisome proliferator-activated receptor agonist.
1793. The device of claim 1670 wherein the agent is an estrogen receptor agent.
1794. The device of claim 1670 wherein the agent is a somastostatin analogue.
1795. The device of claim 1670 wherein the agent is a neurokinin 1 antagonist
1796. The device of claim 1670 wherein the agent is a neurokinin 3 antagonist.
1797. The device of claim 1670 wherein the agent is a VLA-4 antagonist.
1798. The device of claim 1670 wherein the agent is an osteoclast inhibitor.
1799. The device of claim 1670 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
1800. The device of claim 1670 wherein the agent is an angiotensin I converting enzyme inhibitor.
1801. The device of claim 1670 wherein the agent is an angiotensin II antagonist.
1802. The device of claim 1670 wherein the agent is an enkephalinase inhibitor.
1803. The device of claim 1670 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
1804. The device of claim 1670 wherein the agent is a protein kinase C inhibitor.
1805. The device of claim 1670 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
1806. The device of claim 1670 wherein the agent is a CXCR3 inhibitor.
1807. The device of claim 1670 wherein the agent is an Itk inhibitor.
1808. The device of claim 1670 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
1809. The device of claim 1670 wherein the agent is a PPAR ' agonist.
1810. The device of claim 1670 wherein the agent is an immunosuppressant.
1811. The device of claim 1670 wherein the agent is an Erb inhibitor.
1812. The device of claim 1670 wherein the agent is an apoptosis agonist.
1813. The device of claim 1670 wherein the agent is a lipocortin agonist.
1814. The device of claim 1670 wherein the agent is a VCAM-1 antagonist.
1815. The device of claim 1670 wherein the agent is a collagen antagonist.
1816. The device of claim 1670 wherein the agent is an alpha 2 integrin antagonist.
1817. The device of claim 1670 wherein the agent is a TNF alpha inhibitor.
1818. The device of claim 1670 wherein the agent is a nitric oxide inhibitor
1819. The device of claim 1670 wherein the agent is a cathepsin inhibitor.
1820. The device of claim 1670 wherein the agent is not an anti- inflammatory agent.
1821. The device of claim 1670 wherein the agent is not a steroid.
1822. The device of claim 1670 wherein the agent is not a glucocorticosteroid.
1823. The device of claim 1670 wherein the agent is not dexamethasone.
1824. The device of claim 1670 wherein the agent is not an anti- infective agent.
1825. The device of claim 1670 wherein the agent is not an antibiotic.
1826. The device of claim 1670 wherein the agent is not an anti- fugal agent.
1827. The device of claim 1670 wherein the agent is not beclomethasone.
1828. The device of claim 1670 wherein the agent is not dipropionate.
1829. The device of claim 1670, further comprising a coating, wherein the coating comprises the anti-scarring agent and a polymer.
1830. The device of claim 1670, further comprising a coating, wherein the coating comprises the anti-scarring agent.
1831. The device of claim 1670, further comprising a coating, wherein the coating is disposed on a surface of the device.
1832. The device of claim 1670, further comprising a coating, wherein the coating directly contacts the device.
1833. The device of claim 1670, further comprising a coating, wherein the coating indirectly contacts the device.
1834. The device of claim 1670, further comprising a coating, wherein the coating partially covers the device.
1835. The device of claim 1670, further comprising a coating, wherein the coating completely covers the device.
1836. The device of claim 1670, further comprising a coating, wherein the coating is a uniform coating.
1837. The device of claim 1670, further comprising a coating, wherein the coating is a non-uniform coating.
1838. The device of claim 1670, further comprising a coating, wherein the coating is a discontinuous coating.
1839. The device of claim 1670, further comprising a coating, wherein the coating is a patterned coating.
1840. The device of claim 1670, further comprising a coating, wherein the coating has a thickness of 100 μm or less.
1841. The device of claim 1670, further comprising a coating, wherein the coating has a thickness of 10 μm or less.
1842. The device of claim 1670, further comprising a coating, wherein the coating adheres to the surface of the device upon deployment of the device.
1843. The device of claim 1670, further comprising a coating, wherein the coating is stable at room temperature for a period of 1 year.
1844. The device of claim 1670, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001 % to about 1% by weight.
1845. The device of claim 1670, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
1846. The device of claim 1670, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
1847. The device of claim 1670, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
1848. The device of claim 1670, further comprising a coating, wherein the coating further comprises a polymer.
1849. The device of claim 1670, further comprising a first coating having a first composition and the second coating having a second composition.
1850. The device of claim 1670, further comprising a first coating having a first composition and the second coating having a second composition, wherein the first composition and the second composition are different.
1851. The device of claim 1670, further comprising a polymer.
1852. The device of claim 1670, further comprising a polymeric carrier.
1853. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a copolymer.
1854. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a block copolymer.
1855. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a random copolymer.
1856. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a biodegradable polymer.
1857. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-biodegradable polymer.
1858. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophilic polymer.
1859. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophobic polymer.
1860. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophilic domains.
1861. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophobic domains.
1862. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-conductive polymer.
1863. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises an elastomer.
1864. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrogel.
1865. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a silicone polymer.
1866. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrocarbon polymer.
1867. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a styrene-derived polymer.
1868. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a butadiene polymer.
1869. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a macromer.
1870. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises a poly( ethylene glycol) polymer.
1871. The device of claim 1670, further comprising a polymeric carrier, wherein the polymeric carrier comprises an amorphous polymer.
1872. The device of claim 1670, further comprising a lubricious coating.
1873. The device of claim 1670 wherein the anti-scarring agent is located within pores or holes of the device.
1874. The device of claim 1670 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
1875. The device of claim 1670, further comprising a second pharmaceutically active agent.
1876. The device of claim 1670, further comprising an anti- inflammatory agent.
1877. The device of claim 1670, further comprising an agent that inhibits infection.
1878. The device of claim 1670, further comprising an agent that inhibits infection, wherein the agent is an anthracycline.
1879. The device of claim 1670, further comprising an agent that inhibits infection, wherein the agent is doxorubicin.
1880. The device of claim 1670, further comprising an agent that inhibits infection, wherein the agent is mitoxantrone.
1881. The device of claim 1670, further comprising an agent that inhibits infection, wherein the agent is a fluoropyrimidine.
1882. The device of claim 1670, further comprising an agent that inhibits infection, wherein the agent is 5-fluorouracil (5-FU).
1883. The device of claim 1670, further comprising an agent that inhibits infection, wherein the agent is a folic acid antagonist.
1884. The device of claim 1670, further comprising an agent that inhibits infection, wherein the agent is methotrexate.
1885. The device of claim 1670, further comprising an agent that inhibits infection, wherein the agent is a podophylotoxin.
1886. The device of claim 1670, further comprising an agent that inhibits infection, wherein the agent is etoposide.
1887. The device of claim 1670, further comprising an agent that inhibits infection, wherein the agent is a camptothecin.
1888. The device of claim 1670, further comprising an agent that inhibits infection, wherein the agent is a hydroxyurea.
1889. The device of claim 1670, further comprising an agent that inhibits infection, wherein the agent is a platinum complex.
1890. The device of claim 1670, further comprising an agent that inhibits infection, wherein the agent is cisplatin.
1891. The device of claim 1670, further comprising an anti- thrombotic agent.
1892. The device of claim 1670, further comprising a visualization agent.
1893. The device of claim 1670, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
1894. The device of claim 1670, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises barium, tantalum, or technetium.
1895. The device of claim 1670, further comprising a visualization agent, wherein the visualization agent is a MRI responsive material.
1896. The device of claim 1670, further comprising a visualization agent, wherein the visualization agent comprises a gadolinium chelate.
1897. The device of claim 1670, further comprising a visualization agent, wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
1898. The device of claim 1670, further comprising a visualization agent, wherein the visualization agent comprises an iron oxide compound.
1899. The device of claim 1670, further comprising a visualization agent, wherein the visualization agent comprises a dye, pigment, or colorant.
1900. The device of claim 1670, further comprising an echogenic material.
1901. The device of claim 1670, further comprising an echogenic material, wherein the echogenic material is in the form of a coating.
1902. The device of claim 1670 wherein the device is sterile.
1903. The device of claim 1670 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
1904. The device of claim 1670 wherein the device is adapted for delivering the anti-scarring agent locally to tissue proximate to the device.
1905. The device of claim 1670 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
1906. The device of claim 1670 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is connective tissue.
1907. The device of claim 1670 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is muscle tissue.
1908. The device of claim 1670 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is nerve tissue.
1909. The device of claim 1670 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is epithelium tissue.
1910. The device of claim 1670 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
1911. The device of claim 1670 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
1912. The device of claim 1670 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
1913. The device of claim 1670 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
1914. The device of claim 1670 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
1915. The device of claim 1670 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
1916. The device of claim 1670 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
1917. The device of claim 1670 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
1918. The device of claim 1670 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
1919. The device of claim 1670 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
1920. The device of claim 1670 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
1921. The device of claim 1670 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
1922. The device of claim 1670 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
1923. The device of claim 1670 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1924. The device of claim 1670 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1925. The device of claim 1670 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1926. The device of claim 1670 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1927. The device of claim 1670 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1928. The device of claim 1670 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
1929. The device of claim 1670 wherein the agent or the composition is affixed to the sensor.
1930. The device of claim 1670 wherein the agent or the composition is covalently attached to the sensor.
1931. The device of claim 1670 wherein the agent or the composition is non-covalently attached to the sensor.
1932. The device of claim 1670 further comprising a coating that absorbs the agent or the composition.
1933. The device of claim 1670 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
1934. The device of claim 1670 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
1935. The device of claim 1670 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
1936. The device of claim 1670 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
1937. The device of claim 1670 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
1938. The device of claim 1670 further comprising a pump that is linked to the sensor.
1939. The device of any one of claims 1670-1938 wherein the device emits a source of radiation directed towards blood to interact with a plurality of detectors that provide an output signal.
1940. The device of any one of claims 1670-1938 wherein the device is a biosensing transponder composed of a dye that has optical properties that change in response to changes in the environment, a photosensor to sense the optical changes, and a transponder for transmitting data to a remote reader.
1941. The device of any one of claims 1670-1938 wherein the device is a monolithic bioelectronic device for detecting at least one analyte within the host
1942. A device, comprising a pump and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
1943. The device of claim 1942 wherein the agent inhibits cell regeneration.
1944. The device of claim 1942 wherein the agent inhibits angiogenesis.
1945. The device of claim 1942 wherein the agent inhibits fibroblast migration.
1946. The device of claim 1942 wherein the agent inhibits fibroblast proliferation.
1947. The device of claim 1942 wherein the agent inhibits deposition of extracellular matrix.
1948. The device of claim 1942 wherein the agent inhibits tissue remodeling.
1949. The device of claim 1942 wherein the agent is an angiogenesis inhibitor.
1950. The device of claim 1942 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
1951. The device of claim 1942 wherein the agent is a chemokine receptor antagonist.
1952. The device of claim 1942 wherein the agent is a cell cycle inhibitor.
1953. The device of claim 1942 wherein the agent is a taxane.
1954. The device of claim 1942 wherein the agent is an antimicrotubule agent.
1955. The device of claim 1942 wherein the agent is paclitaxel.
1956. The device of claim 1942 wherein the agent is not paclitaxel.
1957. The device of claim 1942 wherein the agent is an analogue or derivative of paclitaxel.
1958. The device of claim 1942 wherein the agent is a vinca alkaloid.
1959. The device of claim 1942 wherein the agent is camptothecin or an analogue or derivative thereof.
1960. The device of claim 1942 wherein the agent is a podophyllotoxin.
1961. The device of claim 1942 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
1962. The device of claim 1942 wherein the agent is an anthracycline.
1963. The device of claim 1942 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
1964. The device of claim 1942 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
1965. The device of claim 1942 wherein the agent is a platinum compound.
1966. The device of claim 1942 wherein the agent is a nitrosourea.
1967. The device of claim 1942 wherein the agent is a nitroimidazole.
1968. The device of claim 1942 wherein the agent is a folic acid antagonist.
1969. The device of claim 1942 wherein the agent is a cytidine analogue.
1970. The device of claim 1942 wherein the agent is a pyrimidine analogue.
1971. The device of claim 1942 wherein the agent is a fluoropyrimidine analogue.
1972. The device of claim 1942 wherein the agent is a purine analogue.
1973. The device of claim 1942 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
1974. The device of claim 1942 wherein the agent is a hydroxyurea.
1975. The device of claim 1942 wherein the agent is a mytomicin or an analogue or derivative thereof.
1976. The device of claim 1942 wherein the agent is an alkyl sulfonate.
1977. The device of claim 1942 wherein the agent is a benzamide or an analogue or derivative thereof.
1978. The device of claim 1942 wherein the agent is a nicotinamide or an analogue or derivative thereof.
1979. The device of claim 1942 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
1980. The device of claim 1942 wherein the agent is a DNA alkylating agent.
1981. The device of claim 1942 wherein the agent is an antimicrotubule agent.
1982. The device of claim 1942 wherein the agent is a topoisomerase inhibitor.
1983. The device of claim 1942 wherein the agent is a DNA cleaving agent.
1984. The device of claim 1942 wherein the agent is an antimetabolite.
1985. The device of claim 1942 wherein the agent inhibits adenosine deaminase.
1986. The device of claim 1942 wherein the agent inhibits purine ring synthesis.
1987. The device of claim 1942 wherein the agent is a nucleotide interconversion inhibitor.
1988. The device of claim 1942 wherein the agent inhibits dihydrofolate reduction.
1989. The device of claim 1942 wherein the agent blocks thymidine monophosphate.
1990. The device of claim 1942 wherein the agent causes DNA damage.
1991. The device of claim 1942 wherein the agent is a DNA intercalation agent.
1992. The device of claim 1942 wherein the agent is a RNA synthesis inhibitor.
1993. The device of claim 1942 wherein the agent is a pyrimidine synthesis inhibitor.
1994. The device of claim 1942 wherein the agent inhibits ribonucleotide synthesis or function.
1995. The device of claim 1942 wherein the agent inhibits thymidine monophosphate synthesis or function.
1996. The device of claim 1942 wherein the agent inhibits DNA synthesis.
1997. The device of claim 1942 wherein the agent causes DNA adduct formation.
1998. The device of claim 1942 wherein the agent inhibits protein synthesis.
1999. The device of claim 1942 wherein the agent inhibits microtubule function.
2000. The device of claim 1942 wherein the agent is a cyclin dependent protein kinase inhibitor.
2001. The device of claim 1942 wherein the agent is an epidermal growth factor kinase inhibitor.
2002. The device of claim 1942 wherein the agent is an elastase inhibitor.
2003. The device of claim 1942 wherein the agent is a factor Xa inhibitor.
2004. The device of claim 1942 wherein the agent is a farnesyltransferase inhibitor.
2005. The device of claim 1942 wherein the agent is a fibrinogen antagonist.
2006. The device of claim 1942 wherein the agent is a guanylate cyclase stimulant.
2007. The device of claim 1942 wherein the agent is a heat shock protein 90 antagonist.
2008. The device of claim 1942 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
2009. The device of claim 1942 wherein the agent is a guanylate cyclase stimulant.
2010. The device of claim 1942 wherein the agent is a HMGCoA reductase inhibitor.
2011. The device of claim 1942 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
2012. The device of claim 1942 wherein the agent is a hydroorotate dehydrogenase inhibitor.
2013. The device of claim 1942 wherein the agent is an IKK2 inhibitor.
2014. The device of claim 1942 wherein the agent is an IL-1 antagonist.
2015. The device of claim 1942 wherein the agent is an ICE antagonist.
2016. The device of claim 1942 wherein the agent is an IRAK antagonist.
2017. The device of claim 1942 wherein the agent is an IL-4 agonist.
2018. The device of claim 1942 wherein the agent is an immunomodulatory agent.
2019. The device of claim 1942 wherein the agent is sirolimus or an analogue or derivative thereof.
2020. The device of claim 1942 wherein the agent is not sirolimus.
2021. The device of claim 1942 wherein the agent is everolimus or an analogue or derivative thereof.
2022. The device of claim 1942 wherein the agent is tacrolimus or an analogue or derivative thereof.
2023. The device of claim 1942 wherein the agent is not tacrolimus.
2024. The device of claim 1942 wherein the agent is biolmus or an analogue or derivative thereof.
2025. The device of claim 1942 wherein the agent is tresperimus or an analogue or derivative thereof.
2026. The device of claim 1942 wherein the agent is auranofin or an analogue or derivative thereof.
2027. The device of claim 1942 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
2028. The device of claim 1942 wherein the agent is gusperimus or an analogue or derivative thereof.
2029. The device of claim 1942 wherein the agent is pimecrolimus or an analogue or derivative thereof.
2030. The device of claim 1942 wherein the agent is ABT-578 or an analogue or derivative thereof.
2031. The device of claim 1942 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
2032. The device of claim 1942 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
2033. The device of claim 1942 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
2034. The device of claim 1942 wherein the agent is a leukotriene inhibitor.
2035. The device of claim 1942 wherein the agent is a MCP-1 antagonist.
2036. The device of claim 1942 wherein the agent is a MMP inhibitor.
2037. The device of claim 1942 wherein the agent is an NF kappa B inhibitor.
2038. The device of claim 1942 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
2039. The device of claim 1942 wherein the agent is an NO antagonist.
2040. The device of claim 1942 wherein the agent is a p38 MAP kinase inhibitor.
2041. The device of claim 1942 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
2042. The device of claim 1942 wherein the agent is a phosphodiesterase inhibitor.
2043. The device of claim 1942 wherein the agent is a TGF beta inhibitor.
2044. The device of claim 1942 wherein the agent is a thromboxane A2 antagonist.
2045. The device of claim 1942 wherein the agent is a TNF alpha antagonist.
2046. The device of claim 1942 wherein the agent is a TACE inhibitor.
2047. The device of claim 1942 wherein the agent is a tyrosine kinase inhibitor.
2048. The device of claim 1942 wherein the agent is a vitronectin inhibitor.
2049. The device of claim 1942 wherein the agent is a fibroblast growth factor inhibitor.
2050. The device of claim 1942 wherein the agent is a protein kinase inhibitor.
2051. The device of claim 1942 wherein the agent is a PDGF receptor kinase inhibitor.
2052. The device of claim 1942 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
2053. The device of claim 1942 wherein the agent is a retinoic acid receptor antagonist.
2054. The device of claim 1942 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
2055. The device of claim 1942 wherein the agent is a fibrinogen antagonist.
2056. The device of claim 1942 wherein the agent is an antimycotic agent.
2057. The device of claim 1942 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
2058. The device of claim 1942 wherein the agent is a bisphosphonate.
2059. The device of claim 1942 wherein the agent is a phospholipase A1 inhibitor.
2060. The device of claim 1942 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
2061. The device of claim 1942 wherein the agent is a macrolide antibiotic.
2062. The device of claim 1942 wherein the agent is a GPIIb/llla receptor antagonist.
2063. The device of claim 1942 wherein the agent is an endothelin receptor antagonist.
2064. The device of claim 1942 wherein the agent is a peroxisome proliferator-activated receptor agonist.
2065. The device of claim 1942 wherein the agent is an estrogen receptor agent.
2066. The device of claim 1942 wherein the agent is a somastostatin analogue.
2067. The device of claim 1942 wherein the agent is a neurokinin 1 antagonist.
2068. The device of claim 1942 wherein the agent is a neurokinin 3 antagonist.
2069. The device of claim 1942 wherein the agent is a VLA-4 antagonist.
2070. The device of claim 1942 wherein the agent is an osteoclast inhibitor.
2071. The device of claim 1942 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
2072. The device of claim 1942 wherein the agent is an angiotensin I converting enzyme inhibitor.
2073. The device of claim 1942 wherein the agent is an angiotensin II antagonist.
2074. The device of claim 1942 wherein the agent is an enkephalinase inhibitor.
2075. The device of claim 1942 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
2076. The device of claim 1942 wherein the agent is a protein kinase C inhibitor.
2077. The device of claim 1942 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
2078. The device of claim 1942 wherein the agent is a CXCR3 inhibitor.
2079. The device of claim 1942 wherein the agent is an Itk inhibitor.
2080. The device of claim 1942 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
2081. The device of claim 1942 wherein the agent is a PPAR agonist.
2082. The device of claim 1942 wherein the agent is an immunosuppressant
2083. The device of claim 1942 wherein the agent is an Erb inhibitor.
2084. The device of claim 1942 wherein the agent is an apoptosis agonist.
2085. The device of claim 1942 wherein the agent is a lipocortin agonist. o
2086. The device of claim 1942 wherein the agent is a VCAM-1 antagonist.
2087. The device of claim 1942 wherein the agent is a collagen antagonist
2088. The device of claim 1942 wherein the agent is an alpha 2 integrin antagonist.
2089. The device of claim 1942 wherein the agent is a TNF alpha inhibitor.
2090. The device of claim 1942 wherein the agent is a nitric oxide inhibitor
2091. The device of claim 1942 wherein the agent is a cathepsin inhibitor.
2092. The device of claim 1942 wherein the agent is not an anti- inflammatory agent.
2093. The device of claim 1942 wherein the agent is not a steroid.
2094. The device of claim 1942 wherein the agent is not a glucocorticosteroid.
2095. The device of claim 1942 wherein the agent is not dexamethasone.
2096. The device of claim 1942 wherein the agent is not an anti- infective agent.
2097. The device of claim 1942 wherein the agent is not an antibiotic.
2098. The device of claim 1942 wherein the agent is not an anti- fugal agent.
2099. The device of claim 1942 wherein the agent is not beclomethasone.
2100. The device of claim 1942 wherein the agent is not dipropionate.
2101. The device of claim 1942, further comprising a coating, wherein the coating comprises the anti-scarring agent and a polymer.
2102. The device of claim 1942, further comprising a coating, wherein the coating comprises the anti-scarring agent.
2103. The device of claim 1942, further comprising a coating, wherein the coating is disposed on a surface of the device.
2104. The device of claim 1942, further comprising a coating, wherein the coating directly contacts the device.
2105. The device of claim 1942, further comprising a coating, wherein the coating indirectly contacts the device.
2106. The device of claim 1942, further comprising a coating, wherein the coating partially covers the device.
2107. The device of claim 1942, further comprising a coating, wherein the coating completely covers the device.
2108. The device of claim 1942, further comprising a coating, wherein the coating is a uniform coating.
2109. The device of claim 1942, further comprising a coating, wherein the coating is a non-uniform coating.
2110. The device of claim 1942, further comprising a coating, wherein the coating is a discontinuous coating.
2111. The device of claim 1942, further comprising a coating, wherein the coating is a patterned coating.
2112. The device of claim 1942, further comprising a coating, wherein the coating has a thickness of 100 μm or less.
2113. The device of claim 1942, further comprising a coating, wherein the coating has a thickness of 10 μm or less.
2114. The device of claim 1942, further comprising a coating, wherein the coating adheres to the surface of the device upon deployment of the device.
2115. The device of claim 1942, further comprising a coating, wherein the coating is stable at room temperature for a period of 1 year.
2116. The device of claim 1942, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1% by weight.
2117. The device of claim 1942, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
2118. The device of claim 1942, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
2119. The device of claim 1942, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
2120. The device of claim 1942, further comprising a coating, wherein the coating further comprises a polymer.
2121. The device of claim 1942, further comprising a first coating having a first composition and the second coating having a second composition.
2122. The device of claim 1942, further comprising a first coating having a first composition and the second coating having a second composition, wherein the first composition and the second composition are different.
2123. The device of claim 1942, further comprising a polymer.
2124. The device of claim 1942, further comprising a polymeric carrier.
2125. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a copolymer.
2126. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a block copolymer.
2127. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a random copolymer.
2128. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a biodegradable polymer.
2129. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-biodegradable polymer.
2130. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophilic polymer.
2131. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophobic polymer.
2132. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophilic domains.
2133. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophobic domains.
2134. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-conductive polymer.
2135. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises an elastomer.
2136. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrogel.
2137. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a silicone polymer.
2138. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrocarbon polymer.
2139. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a styrene-derived polymer.
2140. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a butadiene polymer.
2141. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a macromer.
2142. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
2143. The device of claim 1942, further comprising a polymeric carrier, wherein the polymeric carrier comprises an amorphous polymer.
2144. The device of claim 1942, further comprising a lubricious coating.
2145. The device of claim 1942 wherein the anti-scarring agent is located within pores or holes of the device.
2146. The device of claim 1942 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
2147. The device of claim 1942, further comprising a second pharmaceutically active agent
2148. The device of claim 1942, further comprising an anti- inflammatory agent
2149. The device of claim 1942, further comprising an agent that inhibits infection.
2150. The device of claim 1942, further comprising an agent that inhibits infection, wherein the agent is an anthracycline.
2151. The device of claim 1942, further comprising an agent that inhibits infection, wherein the agent is doxorubicin.
2152. The device of claim 1942, further comprising an agent that inhibits infection, wherein the agent is mitoxantrone.
2153. The device of claim 1942, further comprising an agent that inhibits infection, wherein the agent is a fluoropyrimidine.
2154. The device of claim 1942, further comprising an agent that inhibits infection, wherein the agent is 5-fluorouracil (5-FU).
2155. The device of claim 1942, further comprising an agent that inhibits infection, wherein the agent is a folic acid antagonist.
2156. The device of claim 1942, further comprising an agent that inhibits infection, wherein the agent is methotrexate.
2157. The device of claim 1942, further comprising an agent that inhibits infection, wherein the agent is a podophylotoxin.
2158. The device of claim 1942, further comprising an agent that inhibits infection, wherein the agent is etoposide.
2159. The device of claim 1942, further comprising an agent that inhibits infection, wherein the agent is a camptothecin.
2160. The device of claim 1942, further comprising an agent that inhibits infection, wherein the agent is a hydroxyurea.
2161. The device of claim 1942, further comprising an agent that inhibits infection, wherein the agent is a platinum complex.
2162. The device of claim 1942, further comprising an agent that inhibits infection, wherein the agent is cisplatin.
2163. The device of claim 1942, further comprising an anti- thrombotic agent.
2164. The device of claim 1942, further comprising a visualization agent.
2165. The device of claim 1942, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
2166. The device of claim 1942, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises barium, tantalum, or technetium.
2167. The device of claim 1942, further comprising a visualization agent, wherein the visualization agent is a MRI responsive material.
2168. The device of claim 1942, further comprising a visualization agent, wherein the visualization agent comprises a gadolinium chelate.
2169. The device of claim 1942, further comprising a visualization agent, wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
2170. The device of claim 1942, further comprising a visualization agent, wherein the visualization agent comprises an iron oxide compound.
2171. The device of claim 1942, further comprising a visualization agent, wherein the visualization agent comprises a dye, pigment, or colorant.
2172. The device of claim 1942, further comprising an echogenic material.
2173. The device of claim 1942, further comprising an echogenic material, wherein the echogenic material is in the form of a coating.
2174. The device of claim 1942 wherein the device is sterile.
2175. The device of claim 1942 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
2176. The device of claim 1942 wherein the device is adapted for delivering the anti-scarring agent locally to tissue proximate to the device.
2177. The device of claim 1942 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
2178. The device of claim 1942 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is connective tissue.
2179. The device of claim 1942 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is muscle tissue.
2180. The device of claim 1942 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is nerve tissue.
2181. The device of claim 1942 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is epithelium tissue.
2182. The device of claim 1942 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year. 2005/051871
2183. The device of claim 1942 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
2184. The device of claim 1942 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
2185. The device of claim 1942 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
2186. The device of claim 1942 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
2187. The device of claim 1942 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
2188. The device of claim 1942 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
2189. The device of claim 1942 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days. (
2190. The device of claim 1942 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent
2191. The device of claim 1942 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
2192. The device of claim 1942 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
2193. The device of claim 1942 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent
2194. The device of claim 1942 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
2195. The device of claim 1942 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2196. The device of claim 1942 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2197. The device of claim 1942 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2198. The device of claim 1942 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2199. The device of claim 1942 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2200. The device of claim 1942 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2201. The device of claim 1942 wherein the agent or the composition is affixed to the pump.
2202. The device of claim 1942 wherein the agent or the composition is covalently attached to the pump.
2203. The device of claim 1942 wherein the agent or the composition is non-covalently attached to the pump.
2204. The device of claim 1942 further comprising a coating that absorbs the agent or the composition.
2205. The device of claim 1942 wherein the pump is interweaved with a thread composed of, or coated with, the agent or the composition.
2206. The device of claim 1942 wherein a portion of the pump is covered with a sleeve that contains the agent or the composition.
2207. The device of claim 1942 wherein the pump is completely covered with a sleeve that contains the agent or the composition.
2208. The device of claim 1942 wherein a portion of the pump is covered with a mesh that contains the agent or the composition.
2209. The device of claim 1942 wherein the pump is completely covered with a mesh that contains the agent or the composition.
2210. The device of claim 1942 further comprising a sensor that is linked to the pump.
2211. The device of any one of claims 1942-2210 wherein the device is adapted for delivering insulin.
2212. The device of any one of claims 1942-2210 wherein the device is adapted for delivering a narcotic.
2213. The device of any one of claims 1942-2210 wherein the device is adapted for delivering a chemotherapeutic agent.
2214. The device of any one of claims 1942-2210 wherein the device is adapted for delivering an anti-arrhythmic drug.
2215. The device of any one of claims 1942-2210 wherein the device is adapted for delivering an anti-spasmotic drug.
2216. The device of any one of claims 1942-2210 wherein the device is adapted for delivering an anti-spastic agent.
2217. The device of any one of claims 1942-2210 wherein the device is adapted for delivering an antibiotic.
2218. The device of any one of claims 1942-2210 wherein the device is adapted for delivering a drug only when changes in the host are detected.
2219. The device of any one of claims 1942-2210 wherein the device is adapted for delivering a drug as a continuous slow release.
2220. The device of any one of claims 1942-2210 wherein the device is adapted for delivering a drug at prescribed dosages in a pulsatile manner.
2221. The device of any one of claims 1942-2210 wherein the device is a programmable drug delivery pump.
2222. The device of any one of claims 1942-2210 wherein the device is adapted for intraocularly delivering a drug.
2223. The device of any one of claims 1942-2210 wherein the device is adapted for intrathecally delivering a drug.
2224. The device of any one of claims 1942-2210 wherein the device is adapted for intraperitoneally delivering a drug.
2225. The device of any one of claims 1942-2210 wherein the device is adapted for intra-arterially delivering a drug.
2226. The device of any one of claims 1942-2210 wherein the device is adapted for intracardiac delivery of a drug.
2227. The device of any one of claims 1942-2210 wherein the device is an implantable osmotic pump.
2228. The device of any one of claims 1942-2210 wherein the device is an ocular drug delivery pump.
2229. The device of any one of claims 1942-2210 wherein the device is metering system.
2230. The device of any one of claims 1942-2210 wherein the device is a peristaltic (roller) pump.
2231. The device of any one of claims 1942-2210 wherein the device is an electronically driven pump.
2232. The device of any one of claims 1942-2210 wherein the device is an elastomeric pump.
2233. The device of any one of claims 1942-2210 wherein the device is a spring contraction pump.
2234. The device of any one of claims 1942-2210 wherein the device is a gas-driven pump.
2235. The device of any one of claims 1942-2210 wherein the device is a hydraulic pump.
2236. The device of any one of claims 1942-2210 wherein the device is a piston-dependent pump.
2237. The device of any one of claims 1942-2210 wherein the device is a non-piston-dependent pump.
2238. The device of any one of claims 1942-2210 wherein the device is a dispensing chamber.
2239. The device of any one of claims 1942-2210 wherein the device is an infusion pump.
2240. The device of any one of claims 1942-2210 wherein the device is a passive pump.
2241. A device, comprising an implantable insulin pump and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
2242. The device of claim 2241 wherein the agent inhibits cell regeneration.
2243. The device of claim 2241 wherein the agent inhibits angiogenesis.
2244. The device of claim 2241 wherein the agent inhibits fibroblast migration.
2245. The device of claim 2241 wherein the agent inhibits fibroblast proliferation.
2246. The device of claim 2241 wherein the agent inhibits deposition of extracellular matrix.
2247. The device of claim 2241 wherein the agent inhibits tissue remodeling.
2248. The device of claim 2241 wherein the agent is an angiogenesis inhibitor. i
2249. The device of claim 2241 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
2250. The device of claim 2241 wherein the agent is a chemokine receptor antagonist.
2251. The device of claim 2241 wherein the agent is a cell cycle inhibitor.
2252. The device of claim 2241 wherein the agent is a taxane.
2253. The device of claim 2241 wherein the agent is an antimicrotubule agent.
2254. The device of claim 2241 wherein the agent is paclitaxel.
2255. The device of claim 2241 wherein the agent is not paclitaxel.
2256. The device of claim 2241 wherein the agent is an analogue or derivative of paclitaxel.
2257. The device of claim 2241 wherein the agent is a vinca alkaloid.
2258. The device of claim 2241 wherein the agent is camptothecin or an analogue or derivative thereof.
2259. The device of claim 2241 wherein the agent is a podophyllotoxin.
2260. The device of claim 2241 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
2261. The device of claim 2241 wherein the agent is an anthracycline.
2262. The device of claim 2241 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
2263. The device of claim 2241 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
2264. The device of claim 2241 wherein the agent is a platinum compound.
2265. The device of claim 2241 wherein the agent is a nitrosourea.
2266. The device of claim 2241 wherein the agent is a nitroimidazole.
2267. The device of claim 2241 wherein the agent is a folic acid antagonist.
2268. The device of claim 2241 wherein the agent is a cytidine analogue.
2269. The device of claim 2241 wherein the agent is a pyrimidine analogue.
2270. The device of claim 2241 wherein the agent is a fluoropyrimidine analogue.
2271. The device of claim 2241 wherein the agent is a purine analogue.
2272. The device of claim 2241 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
2273. The device of claim 2241 wherein the agent is a hydroxyurea.
2274. The device of claim 2241 wherein the agent is a mytomicin or an analogue or derivative thereof.
2275. The device of claim 2241 wherein the agent is an alkyl sulfonate.
2276. The device of claim 2241 wherein the agent is a benzamide or an analogue or derivative thereof.
2277. The device of claim 2241 wherein the agent is a nicotinamide or an analogue or derivative thereof.
2278. The device of claim 2241 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
2279. The device of claim 2241 wherein the agent is a DNA alkylating agent.
2280. The device of claim 2241 wherein the agent is an antimicrotubule agent.
2281. The device of claim 2241 wherein the agent is a topoisomerase inhibitor.
2282. The device of claim 2241 wherein the agent is a DNA cleaving agent.
2283. The device of claim 2241 wherein the agent is an antimetabolite.
2284. The device of claim 2241 wherein the agent inhibits adenosine deaminase.
2285. The device of claim 2241 wherein the agent inhibits purine ring synthesis.
2286. The device of claim 2241 wherein the agent is a nucleotide interconversion inhibitor.
2287. The device of claim 2241 wherein the agent inhibits dihydrofolate reduction.
2288. The device of claim 2241 wherein the agent blocks thymidine monophosphate.
2289. The device of claim 2241 wherein the agent causes DNA damage.
2290. The device of claim 2241 wherein the agent is a DNA intercalation agent.
2291. The device of claim 2241 wherein the agent is a RNA synthesis inhibitor.
2292. The device of claim 2241 wherein the agent is a pyrimidine synthesis inhibitor.
2293. The device of claim 2241 wherein the agent inhibits ribonucleotide synthesis or function.
2294. The device of claim 2241 wherein the agent inhibits thymidine monophosphate synthesis or function.
2295. The device of claim 2241 wherein the agent inhibits DNA synthesis.
2296. The device of claim 2241 wherein the agent causes DNA adduct formation.
2297. The device of claim 2241 wherein the agent inhibits protein synthesis.
2298. The device of claim 2241 wherein the agent inhibits microtubule function.
2299. The device of claim 2241 wherein the agent is a cyclin dependent protein kinase inhibitor.
2300. The device of claim 2241 wherein the agent is an epidermal growth factor kinase inhibitor.
2301. The device of claim 2241 wherein the agent is an elastase inhibitor.
2302. The device of claim 2241 wherein the agent is a factor Xa inhibitor.
2303. The device of claim 2241 wherein the agent is a farnesyltransferase inhibitor.
2304. The device of claim 2241 wherein the agent is a fibrinogen antagonist.
2305. The device of claim 2241 wherein the agent is a guanylate cyclase stimulant.
2306. The device of claim 2241 wherein the agent is a heat shock protein 90 antagonist.
2307. The device of claim 2241 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
2308. The device of claim 2241 wherein the agent is a guanylate cyclase stimulant.
2309. The device of claim 2241 wherein the agent is a HMGCoA reductase inhibitor.
2310. The device of claim 2241 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
2311. The device of claim 2241 wherein the agent is a hydroorotate dehydrogenase inhibitor.
2312. The device of claim 2241 wherein the agent is an IKK2 inhibitor.
2313. The device of claim 2241 wherein the agent is an IL-1 antagonist.
2314. The device of claim 2241 wherein the agent is an ICE antagonist.
2315. The device of claim 2241 wherein the agent is an IRAK antagonist.
2316. The device of claim 2241 wherein the agent is an IL-4 agonist.
2317. The device of claim 2241 wherein the agent is an immunomodulatory agent.
2318. The device of claim 2241 wherein the agent is sirolimus or an analogue or derivative thereof.
2319. The device of claim 2241 wherein the agent is not sirolimus.
2320. The device of claim 2241 wherein the agent is everolimus or an analogue or derivative thereof.
2321. The device of claim 2241 wherein the agent is tacrolimus or an analogue or derivative thereof.
2322. The device of claim 2241 wherein the agent is not tacrolimus.
2323. The device of claim 2241 wherein the agent is biolmus or an analogue or derivative thereof.
2324. The device of claim 2241 wherein the agent is tresperimus or an analogue or derivative thereof.
2325. The device of claim 2241 wherein the agent is auranofin or an analogue or derivative thereof.
2326. The device of claim 2241 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
2327. The device of claim 2241 wherein the agent is gusperimus or an analogue or derivative thereof.
2328. The device of claim 2241 wherein the agent is pimecrolimus or an analogue or derivative thereof.
2329. The device of claim 2241 wherein the agent is ABT-578 or an analogue or derivative thereof.
2330. The device of claim 2241 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
2331. The device of claim 2241 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
2332. The device of claim 2241 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
2333. The device of claim 2241 wherein the agent is a leukotriene inhibitor.
2334. The device of claim 2241 wherein the agent is a MCP-1 antagonist.
2335. The device of claim 2241 wherein the agent is a MMP inhibitor. 2005/051871
2336. The device of claim 2241 wherein the agent is an NF kappa B inhibitor.
2337. The device of claim 2241 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
2338. The device of claim 2241 wherein the agent is an NO antagonist.
2339. The device of claim 2241 wherein the agent is a p38 MAP kinase inhibitor.
2340. The device of claim 2241 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
2341. The device of claim 2241 wherein the agent is a phosphodiesterase inhibitor.
2342. The device of claim 2241 wherein the agent is a TGF beta inhibitor.
2343. The device of claim 2241 wherein the agent is a thromboxane A2 antagonist.
2344. The device of claim 2241 wherein the agent is a TNF alpha antagonist.
2345. The device of claim 2241 wherein the agent is a TACE inhibitor.
2346. The device of claim 2241 wherein the agent is a tyrosine kinase inhibitor.
2347. The device of claim 2241 wherein the agent is a vitronectin inhibitor.
2348. The device of claim 2241 wherein the agent is a fibroblast growth factor inhibitor.
2349. The device of claim 2241 wherein the agent is a protein kinase inhibitor.
2350. The device of claim 2241 wherein the agent is a PDGF receptor kinase inhibitor.
2351. The device of claim 2241 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
2352. The device of claim 2241 wherein the agent is a retinoic acid receptor antagonist.
2353. The device of claim 2241 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
2354. The device of claim 2241 wherein the agent is a fibrinogen antagonist.
2355. The device of claim 2241 wherein the agent is an antimycotic agent.
2356. The device of claim 2241 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
2357. The device of claim 2241 wherein the agent is a bisphosphonate.
2358. The device of claim 2241 wherein the agent is a phospholipase A1 inhibitor.
2359. The device of claim 2241 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
2360. The device of claim 2241 wherein the agent is a macrolide antibiotic.
2361. The device of claim 2241 wherein the agent is a GPIIb/llla receptor antagonist.
2362. The device of claim 2241 wherein the agent is an endothelin receptor antagonist.
2363. The device of claim 2241 wherein the agent is a peroxisome proliferator-activated receptor agonist.
2364. The device of claim 2241 wherein the agent is an estrogen receptor agent.
2365. The device of claim 2241 wherein the agent is a somastostatin analogue.
2366. The device of claim 2241 wherein the agent is a neurokinin 1 antagonist.
2367. The device of claim 2241 wherein the agent is a neurokinin 3 antagonist.
2368. The device of claim 2241 wherein the agent is a VLA-4 antagonist. 2005/051871
2369. The device of claim 2241 wherein the agent is an osteoclast inhibitor.
2370. The device of claim 2241 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
2371. The device of claim 2241 wherein the agent is an angiotensin I converting enzyme inhibitor.
2372. The device of claim 2241 wherein the agent is an angiotensin II antagonist.
2373. The device of claim 2241 wherein the agent is an enkephalinase inhibitor.
2374. The device of claim 2241 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
2375. The device of claim 2241 wherein the agent is a protein kinase C inhibitor.
2376. The device of claim 2241 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
2377. The device of claim 2241 wherein the agent is a CXCR3 inhibitor.
2378. The device of claim 2241 wherein the agent is an Itk inhibitor.
2379. The device of claim 2241 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
2380. The device of claim 2241 wherein the agent is a PPAR agonist.
2381. The device of claim 2241 wherein the agent is an immunosuppressant
2382. The device of claim 2241 wherein the agent is an Erb inhibitor.
2383. The device of claim 2241 wherein the agent is an apoptosis agonist.
2384. The device of claim 2241 wherein the agent is a lipocortin agonist.
2385. The device of claim 2241 wherein the agent is a VCAM-1 antagonist.
2386. The device of claim 2241 wherein the agent is a collagen antagonist.
2387. The device of claim 2241 wherein the agent is an alpha 2 integrin antagonist.
2388. The device of claim 2241 wherein the agent is a TNF alpha inhibitor.
2389. The device of claim 2241 wherein the agent is a nitric oxide inhibitor
2390. The device of claim 2241 wherein the agent is a cathepsin inhibitor.
2391. The device of claim 2241 wherein the agent is not an anti- inflammatory agent.
2392. The device of claim 2241 wherein the agent is not a steroid.
2393. The device of claim 2241 wherein the agent is not a glucocorticosteroid.
2394. The device of claim 2241 wherein the agent is not dexamethasone.
2395. The device of claim 2241 wherein the agent is not an anti- infective agent.
2396. The device of claim 2241 wherein the agent is not an antibiotic.
2397. The device of claim 2241 wherein the agent is not an anti- fugal agent.
2398. The device of claim 2241 wherein the agent is not an anti- infective agent.
2399. The device of claim 2241 wherein the agent is not an antibiotic.
2400. The device of claim 2241 wherein the agent is not an anti- fungal agent.
2401. The device of claim 2241 wherein the agent is not beclomethasone.
2402. The device of claim 2241 wherein the agent is not dipropionate.
2403. The device of claim 2241 , further comprising a coating, wherein the coating comprises the anti-scarring agent and a polymer.
2404. The device of claim 2241 , further comprising a coating, wherein the coating indirectly contacts the device.
2405. The device of claim 2241 , further comprising a coating, wherein the coating partially covers the device.
2406. The device of claim 2241 , further comprising a coating, wherein the coating completely covers the device.
2407. The device of claim 2241 , further comprising a coating, wherein the coating is a uniform coating.
2408. The device of claim 2241 , further comprising a coating, wherein the coating is a non-uniform coating.
2409. The device of claim 2241 , further comprising a coating, wherein the coating is a discontinuous coating.
2410. The device of claim 2241 , further comprising a coating, wherein the coating is a patterned coating.
2411. The device of claim 2241 , further comprising a coating, wherein the coating has a thickness of 100 μm or less.
2412. The device of claim 2241 , further comprising a coating, wherein the coating has a thickness of 10 μm or less. 2005/051871
2413. The device of claim 2241 , further comprising a coating, wherein the coating adheres to the surface of the device upon deployment of the device.
2414. The device of claim 2241 , further comprising a coating, wherein the coating is stable at room temperature for a period of 1 year.
2415. The device of claim 2241 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001 % to about 1 % by weight.
2416. The device of claim 2241 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight
2417. The device of claim 2241 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
2418. The device of claim 2241 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
2419. The device of claim 2241 , further comprising a coating, wherein the coating further comprises a polymer.
2420. The device of claim 2241 , further comprising a first coating having a first composition and the second coating having a second composition.
2421. The device of claim 2241 , further comprising a first coating having a first composition and the second coating having a second composition, wherein the first composition and the second composition are different.
2422. The device of claim 2241 , further comprising a polymer.
2423. The device of claim 2241 , further comprising a polymeric carrier.
2424. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a copolymer.
2425. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a block copolymer.
2426. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a random copolymer.
2427. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a biodegradable polymer.
2428. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-biodegradable polymer.
2429. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophilic polymer.
2430. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophobic polymer.
2431. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophilic domains.
2432. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophobic domains.
2433. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-conductive polymer.
2434. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises an elastomer.
2435. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrogel.
2436. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a silicone polymer.
2437. The device of claim 2241, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrocarbon polymer.
2438. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a styrene-derived polymer.
2439. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a butadiene polymer.
2440. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a macromer.
2441. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
2442. The device of claim 2241 , further comprising a polymeric carrier, wherein the polymeric carrier comprises an amorphous polymer.
2443. The device of claim 2241 , further comprising a lubricious coating.
2444. The device of claim 2241 wherein the anti-scarring agent is located within pores or holes of the device.
2445. The device of claim 2241 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
2446. The device of claim 2241 , further comprising a second pharmaceutically active agent.
2447. The device of claim 2241 , further comprising an anti- inflammatory agent.
2448. The device of claim 2241 , further comprising an agent that inhibits infection.
2449. The device of claim 2241 , further comprising an agent that inhibits infection, wherein the agent is an anthracycline.
2450. The device of claim 2241 , further comprising an agent that inhibits infection, wherein the agent is doxorubicin.
2451. The device of claim 2241 , further comprising an agent that inhibits infection, wherein the agent is mitoxantrone.
2452. The device of claim 2241 , further comprising an agent that inhibits infection, wherein the agent is a fluoropyrimidine.
2453. The device of claim 2241 , further comprising an agent that inhibits infection, wherein the agent is 5-fluorouracil (5-FU).
2454. The device of claim 2241 , further comprising an agent that inhibits infection, wherein the agent is a folic acid antagonist.
2455. The device of claim 2241 , further comprising an agent that inhibits infection, wherein the agent is methotrexate.
2456. The device of claim 2241 , further comprising an agent that inhibits infection, wherein the agent is a podophylotoxin.
2457. The device of claim 2241 , further comprising an agent that inhibits infection, wherein the agent is etoposide.
2458. The device of claim 2241 , further comprising an agent that inhibits infection, wherein the agent is a camptothecin.
2459. The device of claim 2241 , further comprising an agent that inhibits infection, wherein the agent is a hydroxyurea.
2460. The device of claim 2241 , further comprising an agent that inhibits infection, wherein the agent is a platinum complex.
2461. The device of claim 2241 , further comprising an agent that inhibits infection, wherein the agent is cisplatin.
2462. The device of claim 2241 , further comprising an anti- thrombotic agent.
2463. The device of claim 2241 , further comprising a visualization agent
2464. The device of claim 2241 , further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
2465. The device of claim 2241 , further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises barium, tantalum, or technetium.
2466. The device of claim 2241 , further comprising a visualization agent, wherein the visualization agent is a MRI responsive material.
2467. The device of claim 2241 , further comprising a visualization agent, wherein the visualization agent comprises a gadolinium chelate.
2468. The device of claim 2241 , further comprising a visualization agent, wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
2469. The device of claim 2241 , further comprising a visualization agent, wherein the visualization agent comprises an iron oxide compound.
2470. The device of claim 2241 , further comprising a visualization agent, wherein the visualization agent comprises a dye, pigment, or colorant.
2471. The device of claim 2241 , further comprising an echogenic material.
2472. The device of claim 2241 , further comprising an echogenic material, wherein the echogenic material is in the form of a coating.
2473. The device of claim 2241 wherein the device is sterile.
2474. The device of claim 2241 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
2475. The device of claim 2241 wherein the device is adapted for delivering the anti-scarring agent locally to tissue proximate to the device.
2476. The device of claim 2241 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
2477. The device of claim 2241 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is connective tissue.
2478. The device of claim 2241 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is muscle tissue.
2479. The device of claim 2241 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is nerve tissue.
2480. The device of claim 2241 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is epithelium tissue.
2481. The device of claim 2241 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year. 2005/051871
2482. The device of claim 2241 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
2483. The device of claim 2241 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
2484. The device of claim 2241 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
2485. The device of claim 2241 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
2486. The device of claim 2241 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
2487. The device of claim 2241 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
2488. The device of claim 2241 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
2489. The device of claim 2241 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
2490. The device of claim 2241 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
2491. The device of claim 2241 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
2492. The device of claim 2241 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
2493. The device of claim 2241 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
2494. The device of claim 2241 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2495. The device of claim 2241 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2496. The device of claim 2241 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2497. The device of claim 2241 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2498. The device of claim 2241 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2499. The device of claim 2241 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2500. The device of claim 2241 wherein the agent or the composition is affixed to the pump.
2501. The device of claim 2241 wherein the agent or the composition is covalently attached to the pump.
2502. The device of claim 2241 wherein the agent or the composition is non-covalently attached to the pump.
2503. The device of claim 2241 further comprising a coating that absorbs the agent or the composition.
2504. The device of claim 2241 wherein the pump is interweaved with a thread composed of, or coated with, the agent or the composition.
2505. The device of claim 2241 wherein a portion of the pump is covered with a sleeve that contains the agent or the composition.
2506. The device of claim 2241 wherein the pump is completely covered with a sleeve that contains the agent or the composition.
2507. The device of claim 2241 wherein a portion of the pump is covered with a mesh that contains the agent or the composition.
2508. The device of claim 2241 wherein the pump is completely covered with a mesh that contains the agent or the composition.
2509. The device of claim 2241 further comprising a sensor that is linked to the pump.
2510. The device of any one of claims 2241-2509 further comprising a single channel catheter with a sensor implanted in a vessel that transmits blood chemistry to the implantable insulin pump to dispense mediation through the catheter.
2511. A device, comprising an intrathecal drug delivery pump and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
2512. The device of claim 2511 wherein the agent inhibits cell regeneration.
2513. The device of claim 2511 wherein the agent inhibits angiogenesis.
2514. The device of claim 2511 wherein the agent inhibits fibroblast migration.
2515. The device of claim 2511 wherein the agent inhibits fibroblast proliferation.
2516. The device of claim 2511 wherein the agent inhibits deposition of extracellular matrix.
2517. The device of claim 2511 wherein the agent inhibits tissue remodeling.
2518. The device of claim 2511 wherein the agent is an angiogenesis inhibitor.
2519. The device of claim 2511 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
2520. The device of claim 2511 wherein the agent is a chemokine receptor antagonist.
2521. The device of claim 2511 wherein the agent is a cell cycle inhibitor.
2522. The device of claim 2511 wherein the agent is a taxane.
2523. The device of claim 2511 wherein the agent is an antimicrotubule agent.
2524. The device of claim 2511 wherein the agent is paclitaxel.
2525. The device of claim 2511 wherein the agent is not paclitaxel.
2526. The device of claim 2511 wherein the agent is an analogue or derivative of paclitaxel.
2527. The device of claim 2511 wherein the agent is a vinca alkaloid.
2528. The device of claim 2511 wherein the agent is camptothecin or an analogue or derivative thereof.
2529. The device of claim 2511 wherein the agent is a podophyllotoxin.
2530. The device of claim 2511 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
2531. The device of claim 2511 wherein the agent is an anthracycline.
2532. The device of claim 2511 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
2533. The device of claim 2511 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
2534. The device of claim 2511 wherein the agent is a platinum compound.
2535. The device of claim 2511 wherein the agent is a nitrosourea.
2536. The device of claim 2511 wherein the agent is a nitroimidazole.
2537. The device of claim 2511 wherein the agent is a folic acid antagonist.
2538. The device of claim 2511 wherein the agent is a cytidine analogue.
2539. The device of claim 2511 wherein the agent is a pyrimidine analogue.
2540. The device of claim 2511 wherein the agent is a fluoropyrimidine analogue.
2541. The device of claim 2511 wherein the agent is a purine analogue.
2542. The device of claim 2511 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
2543. The device of claim 2511 wherein the agent is a hydroxyurea.
2544. The device of claim 2511 wherein the agent is a mytomicin or an analogue or derivative thereof.
2545. The device of claim 2511 wherein the agent is an alkyl sulfonate.
2546. The device of claim 2511 wherein the agent is a benzamide or an analogue or derivative thereof.
2547. The device of claim 2511 wherein the agent is a nicotinamide or an analogue or derivative thereof.
2548. The device of claim 2511 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
2549. The device of claim 2511 wherein the agent is a DNA alkylating agent.
2550. The device of claim 2511 wherein the agent is an antimicrotubule agent.
2551. The device of claim 2511 wherein the agent is a topoisomerase inhibitor.
2552. The device of claim 2511 wherein the agent is a DNA cleaving agent.
2553. The device of claim 2511 wherein the agent is an antimetabolite.
2554. The device of claim 2511 wherein the agent inhibits adenosine deaminase.
2555. The device of claim 2511 wherein the agent inhibits purine ring synthesis.
2556. The device of claim 2511 wherein the agent is a nucleotide interconversion inhibitor.
2557. The device of claim 2511 wherein the agent inhibits dihydrofolate reduction.
2558. The device of claim 2511 wherein the agent blocks thymidine monophosphate.
2559. The device of claim 2511 wherein the agent causes DNA damage.
2560. The device of claim 2511 wherein the agent is a DNA intercalation agent.
2561. The device of claim 2511 wherein the agent is a RNA synthesis inhibitor.
2562. The device of claim 2511 wherein the agent is a pyrimidine synthesis inhibitor.
2563. The device of claim 2511 wherein the agent inhibits ribonucleotide synthesis or function.
2564. The device of claim 2511 wherein the agent inhibits thymidine monophosphate synthesis or function.
2565. The device of claim 2511 wherein the agent inhibits DNA synthesis.
2566. The device of claim 2511 wherein the agent causes DNA adduct formation.
2567. The device of claim 2511 wherein the agent inhibits protein synthesis.
2568. The device of claim 2511 wherein the agent inhibits microtubule function.
2569. The device of claim 2511 wherein the agent is a cyclin dependent protein kinase inhibitor.
2570. The device of claim 2511 wherein the agent is an epidermal growth factor kinase inhibitor.
2571. The device of claim 2511 wherein the agent is an elastase inhibitor.
2572. The device of claim 2511 wherein the agent is a factor Xa inhibitor.
2573. The device of claim 2511 wherein the agent is a farnesyltransferase inhibitor.
2574. The device of claim 2511 wherein the agent is a fibrinogen antagonist.
2575. The device of claim 2511 wherein the agent is a guanylate cyclase stimulant.
2576. The device of claim 2511 wherein the agent is a heat shock protein 90 antagonist.
2577. The device of claim 2511 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
2578. The device of claim 2511 wherein the agent is a guanylate cyclase stimulant.
2579. The device of claim 2511 wherein the agent is a HMGCoA reductase inhibitor.
2580. The device of claim 2511 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
2581. The device of claim 2511 wherein the agent is a hydroorotate dehydrogenase inhibitor.
2582. The device of claim 2511 wherein the agent is an IKK2 inhibitor.
2583. The device of claim 2511 wherein the agent is an IL-1 antagonist.
2584. The device of claim 2511 wherein the agent is an ICE antagonist.
2585. The device of claim 2511 wherein the agent is an IRAK antagonist.
2586. The device of claim 2511 wherein the agent is an IL-4 agonist.
2587. The device of claim 2511 wherein the agent is an immunomodulatory agent.
2588. The device of claim 2511 wherein the agent is sirolimus or an analogue or derivative thereof.
2589. The device of claim 2511 wherein the agent is not sirolimus.
2590. The device of claim 2511 wherein the agent is everolimus or an analogue or derivative thereof.
2591. The device of claim 2511 wherein the agent is tacrolimus or an analogue or derivative thereof.
2592. The device of claim 2511 wherein the agent is not tacrolimus.
2593. The device of claim 2511 wherein the agent is biolmus or an analogue or derivative thereof.
2594. The device of claim 2511 wherein the agent is tresperimus or an analogue or derivative thereof.
2595. The device of claim 2511 wherein the agent is auranofin or an analogue or derivative thereof.
2596. The device of claim 2511 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
2597. The device of claim 2511 wherein the agent is gusperimus or an analogue or derivative thereof.
2598. The device of claim 2511 wherein the agent is pimecrolimus or an analogue or derivative thereof.
2599. The device of claim 2511 wherein the agent is ABT-578 or an analogue or derivative thereof.
2600. The device of claim 2511 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
2601. The device of claim 2511 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
2602. The device of claim 2511 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
2603. The device of claim 2511 wherein the agent is a leukotriene inhibitor.
2604. The device of claim 2511 wherein the agent is a MCP-1 antagonist.
2605. The device of claim 2511 wherein the agent is a MMP inhibitor.
2606. The device of claim 2511 wherein the agent is an NF kappa B inhibitor.
2607. The device of claim 2511 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
2608. The device of claim 2511 wherein the agent is an NO antagonist.
2609. The device of claim 2511 wherein the agent is a p38 MAP kinase inhibitor.
2610. The device of claim 2511 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
2611. The device of claim 2511 wherein the agent is a phosphodiesterase inhibitor.
2612. The device of claim 2511 wherein the agent is a TGF beta inhibitor.
2613. The device of claim 2511 wherein the agent is a thromboxane A2 antagonist.
2614. The device of claim 2511 wherein the agent is a TNF alpha antagonist.
2615. The device of claim 2511 wherein the agent is a TACE inhibitor.
2616. The device of claim 2511 wherein the agent is a tyrosine kinase inhibitor.
2617. The device of claim 2511 wherein the agent is a vitronectin inhibitor.
2618. The device of claim 2511 wherein the agent is a fibroblast growth factor inhibitor.
2619. The device of claim 2511 wherein the agent is a protein kinase inhibitor.
2620. The device of claim 2511 wherein the agent is a PDGF receptor kinase inhibitor.
2621. The device of claim 2511 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
2622. The device of claim 2511 wherein the agent is a retinoic acid receptor antagonist.
2623. The device of claim 2511 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
2624. The device of claim 2511 wherein the agent is a fibrinogen antagonist.
2625. The device of claim 2511 wherein the agent is an antimycotic agent.
2626. The device of claim 2511 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
2627. The device of claim 2511 wherein the agent is a bisphosphonate.
2628. The device of claim 2511 wherein the agent is a phospholipase A1 inhibitor.
2629. The device of claim 2511 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
2630. The device of claim 2511 wherein the agent is a macrolide antibiotic.
2631. The device of claim 2511 wherein the agent is a GPIIb/llla receptor antagonist.
2632. The device of claim 2511 wherein the agent is an endothelin receptor antagonist.
2633. The device of claim 2511 wherein the agent is a peroxisome proliferator-activated receptor agonist.
2634. The device of claim 2511 wherein the agent is an estrogen receptor agent.
2635. The device of claim 2511 wherein the agent is a somastostatin analogue.
2636. The device of claim 2511 wherein the agent is a neurokinin 1 antagonist.
2637. The device of claim 2511 wherein the agent is a neurokinin 3 antagonist.
2638. The device of claim 2511 wherein the agent is a VLA-4 antagonist.
2639. The device of claim 2511 wherein the agent is an osteoclast inhibitor.
2640. The device of claim 2511 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
2641. The device of claim 2511 wherein the agent is an angiotensin I converting enzyme inhibitor.
2642. The device of claim 2511 wherein the agent is an angiotensin II antagonist.
2643. The device of claim 2511 wherein the agent is an enkephalinase inhibitor.
2644. The device of claim 2511 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
2645. The device of claim 2511 wherein the agent is a protein kinase C inhibitor.
2646. The device of claim 2511 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
2647. The device of claim 2511 wherein the agent is a CXCR3 inhibitor.
2648. The device of claim 2511 wherein the agent is an Itk inhibitor.
2649. The device of claim 2511 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
2650. The device of claim 2511 wherein the agent is a PPAR agonist.
2651. The device of claim 2511 wherein the agent is an immunosuppressant.
2652. The device of claim 2511 wherein the agent is an Erb inhibitor.
2653. The device of claim 2511 wherein the agent is an apoptosis agonist.
2654. The device of claim 2511 wherein the agent is a lipocortin agonist.
2655. The device of claim 2511 wherein the agent is a VCAM-1 antagonist.
2656. The device of claim 2511 wherein the agent is a collagen antagonist.
2657. The device of claim 2511 wherein the agent is an alpha 2 integrin antagonist.
2658. The device of claim 2511 wherein the agent is a TNF alpha inhibitor.
2659. The device of claim 2511 wherein the agent is a nitric oxide inhibitor
2660. The device of claim 2511 wherein the agent is a cathepsin inhibitor.
2661. The device of claim 2511 wherein the agent is not an anti- inflammatory agent.
2662. The device of claim 2511 wherein the agent is not a steroid.
2663. The device of claim 2511 wherein the agent is not a glucocorticosteroid.
2664. The device of claim 2511 wherein the agent is not dexamethasone.
2665. The device of claim 2511 wherein the agent is not an anti- infective agent.
2666. The device of claim 2511 wherein the agent is not an antibiotic.
2667. The device of claim 2511 wherein the agent is not an anti- fugal agent.
2668. The device of claim 2511 wherein the agent is not beclomethasone.
2669. The device of claim 2511 wherein the agent is not dipropionate.
2670. The device of claim 2511 , further comprising a coating, wherein the coating comprises the anti-scarring agent and a polymer.
2671. The device of claim 2511 , further comprising a coating, wherein the coating comprises the anti-scarring agent.
2672. The device of claim 2511 , further comprising a coating, wherein the coating is disposed on a surface of the device.
2673. The device of claim 2511 , further comprising a coating, wherein the coating directly contacts the device.
2674. The device of claim 2511 , further comprising a coating, wherein the coating indirectly contacts the device.
2675. The device of claim 2511 , further comprising a coating, wherein the coating partially covers the device.
2676. The device of claim 2511 , further comprising a coating, wherein the coating completely covers the device.
2677. The device of claim 2511 , further comprising a coating, wherein the coating is a uniform coating.
2678. The device of claim 2511 , further comprising a coating, wherein the coating is a non-uniform coating.
2679. The device of claim 2511 , further comprising a coating, wherein the coating is a discontinuous coating.
2680. The device of claim 2511 , further comprising a coating, wherein the coating is a patterned coating.
2681. The device of claim 2511 , further comprising a coating, wherein the coating has a thickness of 100 μm or less.
2682. The device of claim 2511 , further comprising a coating, wherein the coating has a thickness of 10 μm or less.
2683. The device of claim 2511 , further comprising a coating, wherein the coating adheres to the surface of the device upon deployment of the device.
2684. The device of claim 2511 , further comprising a coating, wherein the coating is stable at room temperature for a period of 1 year.
2685. The device of claim 2511 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1 % by weight.
2686. The device of claim 2511 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
2687. The device of claim 2511 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
2688. The device of claim 2511 , further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
2689. The device of claim 2511 , further comprising a coating, wherein the coating further comprises a polymer.
2690. The device of claim 2511 , further comprising a first coating having a first composition and the second coating having a second composition.
2691. The device of claim 2511 , further comprising a first coating having a first composition and the second coating having a second composition, wherein the first composition and the second composition are different.
2692. The device of claim 2511 , further comprising a polymer.
2693. The device of claim 2511 , further comprising a polymeric carrier.
2694. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a copolymer.
2695. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a block copolymer. i
2696. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a random copolymer.
2697. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a biodegradable polymer.
2698. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-biodegradable polymer.
2699. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophilic polymer.
2700. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophobic polymer.
2701. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophilic domains.
2702. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophobic domains.
2703. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-conductive polymer.
2704. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises an elastomer.
2705. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrogel.
2706. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a silicone polymer.
2707. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrocarbon polymer.
2708. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a styrene-derived polymer.
2709. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a butadiene polymer.
2710. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a macromer.
2711. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises a poly( ethylene glycol) polymer.
2712. The device of claim 2511 , further comprising a polymeric carrier, wherein the polymeric carrier comprises an amorphous polymer.
2713. The device of claim 2511 , further comprising a lubricious coating.
2714. The device of claim 2511 wherein the anti-scarring agent is located within pores or holes of the device.
2715. The device of claim 2511 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
2716. The device of claim 2511 , further comprising a second pharmaceutically active agent.
2717. The device of claim 2511 , further comprising an anti- inflammatory agent.
2718. The device of claim 2511 , further comprising an agent that inhibits infection.
2719. The device of claim 2511 , further comprising an agent that inhibits infection, wherein the agent is an anthracycline.
2720. The device of claim 2511 , further comprising an agent that inhibits infection, wherein the agent is doxorubicin.
2721. The device of claim 2511 , further comprising an agent that inhibits infection, wherein the agent is mitoxantrone.
2722. The device of claim 2511 , further comprising an agent that inhibits infection, wherein the agent is a fluoropyrimidine.
2723. The device of claim 2511 , further comprising an agent that inhibits infection, wherein the agent is 5-fluorouracil (5-FU).
2724. The device of claim 2511 , further comprising an agent that inhibits infection, wherein the agent is a folic acid antagonist.
2725. The device of claim 2511 , further comprising an agent that inhibits infection, wherein the agent is methotrexate.
2726. The device of claim 2511 , further comprising an agent that inhibits infection, wherein the agent is a podophylotoxin.
2727. The device of claim 2511 , further comprising an agent that inhibits infection, wherein the agent is etoposide.
2728. The device of claim 2511 , further comprising an agent that inhibits infection, wherein the agent is a camptothecin.
2729. The device of claim 2511 , further comprising an agent that inhibits infection, wherein the agent is a hydroxyurea.
2730. The device of claim 2511 , further comprising an agent that inhibits infection, wherein the agent is a platinum complex.
2731. The device of claim 2511 , further comprising an agent that inhibits infection, wherein the agent is cisplatin.
2732. The device of claim 2511 , further comprising an anti- thrombotic agent.
2733. The device of claim 2511 , further comprising a visualization agent.
2734. The device of claim 2511 , further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
2735. The device of claim 2511 , further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises barium, tantalum, or technetium.
2736. The device of claim 2511 , further comprising a visualization agent, wherein the visualization agent is a MRI responsive material.
2737. The device of claim 2511 , further comprising a visualization agent, wherein the visualization agent comprises a gadolinium chelate.
2738. The device of claim 2511 , further comprising a visualization agent, wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
2739. The device of claim 2511 , further comprising a visualization agent, wherein the visualization agent comprises an iron oxide compound.
2740. The device of claim 2511 , further comprising a visualization agent, wherein the visualization agent comprises a dye, pigment, or colorant.
2741. The device of claim 2511 , further comprising an echogenic material.
2742. The device of claim 2511 , further comprising an echogenic material, wherein the echogenic material is in the form of a coating.
2743. The device of claim 2511 wherein the device is sterile.
2744. The device of claim 2511 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
2745. The device of claim 2511 wherein the device is adapted for delivering the anti-scarring agent locally to tissue proximate to the device.
2746. The device of claim 2511 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
2747. The device of claim 2511 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is connective tissue.
2748. The device of claim 2511 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is muscle tissue.
2749. The device of claim 2511 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is nerve tissue.
2750. The device of claim 2511 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is epithelium tissue.
2751. The device of claim 2511 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
2752. The device of claim 2511 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
2753. The device of claim 2511 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
2754. The device of claim 2511 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
2755. The device of claim 2511 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
2756. The device of claim 2511 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
2757. The device of claim 2511 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
2758. The device of claim 2511 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
2759. The device of claim 2511 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent
2760. The device of claim 2511 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
2761. The device of claim 2511 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
2762. The device of claim 2511 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
2763. The device of claim 2511 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
2764. The device of claim 2511 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2765. The device of claim 2511 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2766. The device of claim 2511 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2767. The device of claim 2511 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2768. The device of claim 2511 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2769. The device of claim 2511 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
2770. The device of claim 2511 wherein the agent or the composition is affixed to the pump.
2771. The device of claim 2511 wherein the agent or the composition is covalently attached to the pump.
2772. The device of claim 2511 wherein the agent or the composition is non-covalently attached to the pump.
2773. The device of claim 2511 further comprising a coating that absorbs the agent or the composition.
2774. The device of claim 2511 wherein the pump is interweaved with a thread composed of, or coated with, the agent or the composition.
2775. The device of claim 2511 wherein a portion of the pump is covered with a sleeve that contains the agent or the composition.
2776. The device of claim 2511 wherein the pump is completely covered with a sleeve that contains the agent or the composition.
2777. The device of claim 2511 wherein a portion of the pump is covered with a mesh that contains the agent or the composition.
2778. The device of claim 2511 wherein the pump is completely covered with a mesh that contains the agent or the composition.
2779. The device of claim 2511 further comprising a sensor that is linked to the pump.
2780. The device of any one of claims 2511-2779 wherein the device is adapted for delivering pain medication directly into the cerebrospinal fluid of the intrathecal space surrounding the spinal cord.
2781. The device of any one of claims 2511-2779 wherein the device is adapted for delivering a drug to the brain.
2782. The device of any one of claims 2511-2779 wherein the device is adapted for intrathecal delivering baclofen.
2783. The device of any one of claims 2511-2779 wherein the device further comprises an intraspinal catheter.
2784. The device of any one of claims 2511 -2779 further comprising a second intrathecal drug delivery pump.
2785. The device of any one of claims 2511-2779 wherein the device further comprises a catheter and an electrode.
2786. A device, comprising an implantable drug delivery pump for chemotherapy and an anti-scarring agent or a composition comprising an anti- scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
2787. The device of claim 2786 wherein the agent inhibits cell regeneration.
2788. The device of claim 2786 wherein the agent inhibits angiogenesis.
2789. The device of claim 2786 wherein the agent inhibits fibroblast migration.
2790. The device of claim 2786 wherein the agent inhibits fibroblast proliferation.
2791. The device of claim 2786 wherein the agent inhibits deposition of extracellular matrix.
2792. The device of claim 2786 wherein the agent inhibits tissue remodeling.
2793. The device of claim 2786 wherein the agent is an angiogenesis inhibitor.
2794. The device of claim 2786 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
2795. The device of claim 2786 wherein the agent is a chemokine receptor antagonist.
2796. The device of claim 2786 wherein the agent is a cell cycle inhibitor.
2797. The device of claim 2786 wherein the agent is a taxane.
2798. The device of claim 2786 wherein the agent is an antimicrotubule agent.
2799. The device of claim 2786 wherein the agent is paclitaxel.
2800. The device of claim 2786 wherein the agent is not paclitaxel.
2801. The device of claim 2786 wherein the agent is an analogue or derivative of paclitaxel.
2802. The device of claim 2786 wherein the agent is a vinca alkaloid.
2803. The device of claim 2786 wherein the agent is camptothecin or an analogue or derivative thereof.
2804. The device of claim 2786 wherein the agent is a podophyllotoxin.
2805. The device of claim 2786 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
2806. The device of claim 2786 wherein the agent is an anthracycline.
2807. The device of claim 2786 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
2808. The device of claim 2786 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
2809. The device of claim 2786 wherein the agent is a platinum compound.
2810. The device of claim 2786 wherein the agent is a nitrosourea.
2811. The device of claim 2786 wherein the agent is a nitroimidazole.
2812. The device of claim 2786 wherein the agent is a folic acid antagonist.
2813. The device of claim 2786 wherein the agent is a cytidine analogue.
2814. The device of claim 2786 wherein the agent is a pyrimidine analogue.
2815. The device of claim 2786 wherein the agent is a fluoropyrimidine analogue.
2816. The device of claim 2786 wherein the agent is a purine analogue.
2817. The device of claim 2786 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
2818. The device of claim 2786 wherein the agent is a hydroxyurea.
2819. The device of claim 2786 wherein the agent is a mytomicin or an analogue or derivative thereof.
2820. The device of claim 2786 wherein the agent is an alkyl sulfonate.
2821. The device of claim 2786 wherein the agent is a benzamide or an analogue or derivative thereof.
2822. The device of claim 2786 wherein the agent is a nicotinamide or an analogue or derivative thereof.
2823. The device of claim 2786 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
2824. The device of claim 2786 wherein the agent is a DNA alkylating agent.
2825. The device of claim 2786 wherein the agent is an antimicrotubule agent
2826. The device of claim 2786 wherein the agent is a topoisomerase inhibitor.
2827. The device of claim 2786 wherein the agent is a DNA cleaving agent.
2828. The device of claim 2786 wherein the agent is an antimetabolite.
2829. The device of claim 2786 wherein the agent inhibits adenosine deaminase.
2830. The device of claim 2786 wherein the agent inhibits purine ring synthesis.
2831. The device of claim 2786 wherein the agent is a nucleotide interconversion inhibitor.
2832. The device of claim 2786 wherein the agent inhibits dihydrofolate reduction.
2833. The device of claim 2786 wherein the agent blocks thymidine monophosphate.
2834. The device of claim 2786 wherein the agent causes DNA damage.
2835. The device of claim 2786 wherein the agent is a DNA intercalation agent.
2836. The device of claim 2786 wherein the agent is a RNA synthesis inhibitor.
2837. The device of claim 2786 wherein the agent is a pyrimidine synthesis inhibitor.
2838. The device of claim 2786 wherein the agent inhibits ribonucleotide synthesis or function.
2839. The device of claim 2786 wherein the agent inhibits thymidine monophosphate synthesis or function.
2840. The device of claim 2786 wherein the agent inhibits DNA synthesis.
2841. The device of claim 2786 wherein the agent causes DNA adduct formation.
2842. The device of claim 2786 wherein the agent inhibits protein synthesis.
2843. The device of claim 2786 wherein the agent inhibits microtubule function.
2844. The device of claim 2786 wherein the agent is a cyclin dependent protein kinase inhibitor.
2845. The device of claim 2786 wherein the agent is an epidermal growth factor kinase inhibitor.
2846. The device of claim 2786 wherein the agent is an elastase inhibitor.
2847. The device of claim 2786 wherein the agent is a factor Xa inhibitor.
2848. The device of claim 2786 wherein the agent is a farnesyltransferase inhibitor.
2849. The device of claim 2786 wherein the agent is a fibrinogen antagonist.
2850. The device of claim 2786 wherein the agent is a guanylate cyclase stimulant.
2851. The device of claim 2786 wherein the agent is a heat shock protein 90 antagonist.
2852. The device of claim 2786 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
2853. The device of claim 2786 wherein the agent is a guanylate cyclase stimulant
2854. The device of claim 2786 wherein the agent is a HMGCoA reductase inhibitor.
2855. The device of claim 2786 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
2856. The device of claim 2786 wherein the agent is a hydroorotate dehydrogenase inhibitor.
2857. The device of claim 2786 wherein the agent is an IKK2 inhibitor.
2858. The device of claim 2786 wherein the agent is an IL-1 antagonist.
2859. The device of claim 2786 wherein the agent is an ICE antagonist.
2860. The device of claim 2786 wherein the agent is an IRAK antagonist.
2861. The device of claim 2786 wherein the agent is an IL-4 agonist.
2862. The device of claim 2786 wherein the agent is an immunomodulatory agent.
2863. The device of claim 2786 wherein the agent is sirolimus or an analogue or derivative thereof.
2864. The device of claim 2786 wherein the agent is not sirolimus.
2865. The device of claim 2786 wherein the agent is everolimus or an analogue or derivative thereof.
2866. The device of claim 2786 wherein the agent is tacrolimus or an analogue or derivative thereof.
2867. The device of claim 2786 wherein the agent is not tacrolimus.
2868. The device of claim 2786 wherein the agent is biolmus or an analogue or derivative thereof.
2869. The device of claim 2786 wherein the agent is tresperimus or an analogue or derivative thereof.
2870. The device of claim 2786 wherein the agent is auranofin or an analogue or derivative thereof.
2871. The device of claim 2786 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
2872. The device of claim 2786 wherein the agent is gusperimus or an analogue or derivative thereof.
2873. The device of claim 2786 wherein the agent is pimecrolimus or an analogue or derivative thereof.
2874. The device of claim 2786 wherein the agent is ABT-578 or an analogue or derivative thereof.
2875. The device of claim 2786 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
2876. The device of claim 2786 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
2877. The device of claim 2786 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
2878. The device of claim 2786 wherein the agent is a leukotriene inhibitor.
2879. The device of claim 2786 wherein the agent is a MCP-1 antagonist.
2880. The device of claim 2786 wherein the agent is a MMP inhibitor.
2881. The device of claim 2786 wherein the agent is an NF kappa B inhibitor.
2882. The device of claim 2786 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
2883. The device of claim 2786 wherein the agent is an NO antagonist.
2884. The device of claim 2786 wherein the agent is a p38 MAP kinase inhibitor.
2885. The device of claim 2786 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
2886. The device of claim 2786 wherein the agent is a phosphodiesterase inhibitor.
2887. The device of claim 2786 wherein the agent is a TGF beta inhibitor.
2888. The device of claim 2786 wherein the agent is a thromboxane A2 antagonist.
2889. The device of claim 2786 wherein the agent is a TNF alpha antagonist.
2890. The device of claim 2786 wherein the agent is a TACE inhibitor.
2891. The device of claim 2786 wherein the agent is a tyrosine kinase inhibitor.
2892. The device of claim 2786 wherein the agent is a vitronectin inhibitor.
2893. The device of claim 2786 wherein the agent is a fibroblast growth factor inhibitor.
2894. The device of claim 2786 wherein the agent is a protein kinase inhibitor.
2895. The device of claim 2786 wherein the agent is a PDGF receptor kinase inhibitor.
2896. The device of claim 2786 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
2897. The device of claim 2786 wherein the agent is a retinoic acid receptor antagonist.
2898. The device of claim 2786 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
2899. The device of claim 2786 wherein the agent is a fibrinogen antagonist.
2900. The device of claim 2786 wherein the agent is an antimycotic agent.
2901. The device of claim 2786 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
2902. The device of claim 2786 wherein the agent is a bisphosphonate.
2903. The device of claim 2786 wherein the agent is a phospholipase A1 inhibitor.
2904. The device of claim 2786 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
2905. The device of claim 2786 wherein the agent is a macrolide antibiotic.
2906. The device of claim 2786 wherein the agent is a GPIIb/llla receptor antagonist.
2907. The device of claim 2786 wherein the agent is an endothelin receptor antagonist.
2908. The device of claim 2786 wherein the agent is a peroxisome proliferator-activated receptor agonist.
2909. The device of claim 2786 wherein the agent is an estrogen receptor agent.
2910. The device of claim 2786 wherein the agent is a somastostatin analogue.
2911. The device of claim 2786 wherein the agent is a neurokinin 1 antagonist.
2912. The device of claim 2786 wherein the agent is a neurokinin 3 antagonist.
2913. The device of claim 2786 wherein the agent is a VLA-4 antagonist.
2914. The device of claim 2786 wherein the agent is an osteoclast inhibitor.
2915. The device of claim 2786 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
2916. The device of claim 2786 wherein the agent is an angiotensin I converting enzyme inhibitor.
2917. The device of claim 2786 wherein the agent is an angiotensin II antagonist.
2918. The device of claim 2786 wherein the agent is an enkephalinase inhibitor.
2919. The device of claim 2786 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
2920. The device of claim 2786 wherein the agent is a protein kinase C inhibitor.
2921. The device of claim 2786 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
2922. The device of claim 2786 wherein the agent is a CXCR3 inhibitor.
2923. The device of claim 2786 wherein the agent is an Itk inhibitor.
2924. The device of claim 2786 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
2925. The device of claim 2786 wherein the agent is a PPAR agonist.
2926. The device of claim 2786 wherein the agent is an immunosuppressant
2927. The device of claim 2786 wherein the agent is an Erb inhibitor.
2928. The device of claim 2786 wherein the agent is an apoptosis agonist.
2929. The device of claim 2786 wherein the agent is a lipocortin agonist.
2930. The device of claim 2786 wherein the agent is a VCAM-1 antagonist
2931. The device of claim 2786 wherein the agent is a collagen antagonist.
2932. The device of claim 2786 wherein the agent is an alpha 2 integrin antagonist.
2933. The device of claim 2786 wherein the agent is a TNF alpha inhibitor.
2934. The device of claim 2786 wherein the agent is a nitric oxide inhibitor
2935. The device of claim 2786 wherein the agent is a cathepsin inhibitor.
2936. The device of claim 2786 wherein the agent is not an anti- inflammatory agent.
2937. The device of claim 2786 wherein the agent is not a steroid.
2938. The device of claim 2786 wherein the agent is not a glucocorticosteroid.
2939. The device of claim 2786 wherein the agent is not dexamethasone.
2940. The device of claim 2786 wherein the agent is not an anti- infective agent.
2941. The device of claim 2786 wherein the agent is not an antibiotic.
2942. The device of claim 2786 wherein the agent is not an anti- fugal agent.
2943. The device of claim 2786 wherein the agent is not beclomethasone.
2944. The device of claim 2786 wherein the agent is not dipropionate.
2945. The device of claim 2786, further comprising a coating, wherein the coating comprises the anti-scarring agent and a polymer.
2946. The device of claim 2786, further comprising a coating, wherein the coating comprises the anti-scarring agent.
2947. The device of claim 2786, further comprising a coating, wherein the coating is disposed on a surface of the device.
2948. The device of claim 2786, further comprising a coating, wherein the coating directly contacts the device.
2949. The device of claim 2786, further comprising a coating, wherein the coating indirectly contacts the device.
2950. The device of claim 2786, further comprising a coating, wherein the coating partially covers the device.
2951. The device of claim 2786, further comprising a coating, wherein the coating completely covers the device.
2952. The device of claim 2786, further comprising a coating, wherein the coating is a uniform coating.
2953. The device of claim 2786, further comprising a coating, wherein the coating is a non-uniform coating.
2954. The device of claim 2786, further comprising a coating, wherein the coating is a discontinuous coating.
2955. The device of claim 2786, further comprising a coating, wherein the coating is a patterned coating.
2956. The device of claim 2786, further comprising a coating, wherein the coating has a thickness of 100 μm or less.
2957. The device of claim 2786, further comprising a coating, wherein the coating has a thickness of 10 μm or less.
2958. The device of claim 2786, further comprising a coating, wherein the coating adheres to the surface of the device upon deployment of the device.
2959. The device of claim 2786, further comprising a coating, wherein the coating is stable at room temperature for a period of 1 year.
2960. The device of claim 2786, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1% by weight.
2961. The device of claim 2786, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
2962. The device of claim 2786, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight
2963. The device of claim 2786, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
2964. The device of claim 2786, further comprising a coating, wherein the coating further comprises a polymer.
2965. The device of claim 2786, further comprising a first coating having a first composition and the second coating having a second composition.
2966. The device of claim 2786, further comprising a first coating having a first composition and the second coating having a second composition, wherein the first composition and the second composition are different.
2967. The device of claim 2786, further comprising a polymer.
2968. The device of claim 2786, further comprising a polymeric carrier.
2969. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a copolymer.
2970. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a block copolymer.
2971. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a random copolymer.
2972. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a biodegradable polymer.
2973. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-biodegradable polymer.
2974. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophilic polymer.
2975. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophobic polymer.
2976. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophilic domains.
2977. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophobic domains.
2978. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-conductive polymer.
2979. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises an elastomer.
2980. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrogel.
2981. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a silicone polymer.
2982. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrocarbon polymer.
2983. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a styrene-derived polymer.
2984. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a butadiene polymer.
2985. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a macromer.
2986. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
2987. The device of claim 2786, further comprising a polymeric carrier, wherein the polymeric carrier comprises an amorphous polymer.
2988. The device of claim 2786, further comprising a lubricious coating.
2989. The device of claim 2786 wherein the anti-scarring agent is located within pores or holes of the device.
2990. The device of claim 2786 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
2991. The device of claim 2786, further comprising a second pharmaceutically active agent
2992. The device of claim 2786, further comprising an anti- inflammatory agent.
2993. The device of claim 2786, further comprising an agent that inhibits infection.
2994. The device of claim 2786, further comprising an agent that inhibits infection, wherein the agent is an anthracycline.
2995. The device of claim 2786, further comprising an agent that inhibits infection, wherein the agent is doxorubicin.
2996. The device of claim 2786, further comprising an agent that inhibits infection, wherein the agent is mitoxantrone.
2997. The device of claim 2786, further comprising an agent that inhibits infection, wherein the agent is a fluoropyrimidine.
2998. The device of claim 2786, further comprising an agent that inhibits infection, wherein the agent is 5-fluorouracil (5-FU).
2999. The device of claim 2786, further comprising an agent that inhibits infection, wherein the agent is a folic acid antagonist.
3000. The device of claim 2786, further comprising an agent that inhibits infection, wherein the agent is methotrexate.
3001. The device of claim 2786, further comprising an agent that inhibits infection, wherein the agent is a podophylotoxin.
3002. The device of claim 2786, further comprising an agent that inhibits infection, wherein the agent is etoposide.
3003. The device of claim 2786, further comprising an agent that inhibits infection, wherein the agent is a camptothecin.
3004. The device of claim 2786, further comprising an agent that inhibits infection, wherein the agent is a hydroxyurea.
3005. The device of claim 2786, further comprising an agent that inhibits infection, wherein the agent is a platinum complex.
3006. The device of claim 2786, further comprising an agent that inhibits infection, wherein the agent is cisplatin.
3007. The device of claim 2786, further comprising an anti- thrombotic agent.
3008. The device of claim 2786, further comprising a visualization agent.
3009. The device of claim 2786, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
3010. The device of claim 2786, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises barium, tantalum, or technetium.
3011. The device of claim 2786, further comprising a visualization agent, wherein the visualization agent is a MRI responsive material.
3012. The device of claim 2786, further comprising a visualization agent, wherein the visualization agent comprises a gadolinium chelate.
3013. The device of claim 2786, further comprising a visualization agent, wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
3014. The device of claim 2786, further comprising a visualization agent, wherein the visualization agent comprises an iron oxide compound.
3015. The device of claim 2786, further comprising a visualization agent, wherein the visualization agent comprises a dye, pigment, or colorant.
3016. The device of claim 2786, further comprising an echogenic material.
3017. The device of claim 2786, further comprising an echogenic material, wherein the echogenic material is in the form of a coating.
3018. The device of claim 2786 wherein the device is sterile.
3019. The device of claim 2786 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
3020. The device of claim 2786 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
3021. The device of claim 2786 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
3022. The device of claim 2786 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is connective tissue.
3023. The device of claim 2786 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is muscle tissue.
3024. The device of claim 2786 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is nerve tissue.
3025. The device of claim 2786 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is epithelium tissue.
3026. The device of claim 2786 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
3027. The device of claim 2786 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
3028. The device of claim 2786 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
3029. The device of claim 2786 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
3030. The device of claim 2786 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
3031. The device of claim 2786 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
3032. The device of claim 2786 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
3033. The device of claim 2786 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
3034. The device of claim 2786 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
3035. The device of claim 2786 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
3036. The device of claim 2786 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
3037. The device of claim 2786 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
3038. The device of claim 2786 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
3039. The device of claim 2786 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3040. The device of claim 2786 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3041. The device of claim 2786 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3042. The device of claim 2786 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3043. The device of claim 2786 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3044. The device of claim 2786 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3045. The device of claim 2786 wherein the agent or the composition is affixed to the pump.
3046. The device of claim 2786 wherein the agent or the composition is covalently attached to the pump.
3047. The device of claim 2786 wherein the agent or the composition is non-covalently attached to the pump.
3048. The device of claim 2786 further comprising a coating that absorbs the agent or the composition.
3049. The device of claim 2786 wherein the pump is interweaved with a thread composed of, or coated with, the agent or the composition.
3050. The device of claim 2786 wherein a portion of the pump is covered with a sleeve that contains the agent or the composition.
3051. The device of claim 2786 wherein the pump is completely covered with a sleeve that contains the agent or the composition.
3052. The device of claim 2786 wherein a portion of the pump is covered with a mesh that contains the agent or the composition.
3053. The device of claim 2786 wherein the pump is completely covered with a mesh that contains the agent or the composition.
3054. The device of claim 2786 further comprising a sensor that is linked to the pump.
3055. The device of any one of claims 2786-3054 wherein the device is adapted for delivering 2'-deoxy 5-fluorouridine.
3056. The device of any one of claims 2786-3054 wherein the host has a solid tumor, and the device is adapted for infusing a chemotherapeutic agent to the solid tumor.
3057. The device of any one of claims 2786-3054 wherein the host has a tumor, and the device is adapted for infusing a chemotherapeutic agent to the blood vessels that supply the tumor.
3058. The device of any one of claims 2786-3054 wherein the host has a hepatic tumor, and the device is adapted for delivering a chemotherapeutic agent to the artery that provides blood supply to the liver of the host.
3059. A device, comprising a drug delivery pump for treating heart disease and an anti-scarring agent or a composition comprising an anti- scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
3060. The device of claim 3059 wherein the agent inhibits cell regeneration.
3061. The device of claim 3059 wherein the agent inhibits angiogenesis.
3062. The device of claim 3059 wherein the agent inhibits fibroblast migration.
3063. The device of claim 3059 wherein the agent inhibits fibroblast proliferation.
3064. The device of claim 3059 wherein the agent inhibits deposition of extracellular matrix.
3065. The device of claim 3059 wherein the agent inhibits tissue remodeling.
3066. The device of claim 3059 wherein the agent is an angiogenesis inhibitor.
3067. The device of claim 3059 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
3068. The device of claim 3059 wherein the agent is a chemokine receptor antagonist.
3069. The device of claim 3059 wherein the agent is a cell cycle inhibitor.
3070. The device of claim 3059 wherein the agent is a taxane.
3071. The device of claim 3059 wherein the agent is an antimicrotubule agent.
3072. The device of claim 3059 wherein the agent is paclitaxel.
3073. The device of claim 3059 wherein the agent is not paclitaxel.
3074. The device of claim 3059 wherein the agent is an analogue or derivative of paclitaxel.
3075. The device of claim 3059 wherein the agent is a vinca alkaloid.
3076. The device of claim 3059 wherein the agent is camptothecin or an analogue or derivative thereof.
3077. The device of claim 3059 wherein the agent is a podophyllotoxin.
3078. The device of claim 3059 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
3079. The device of claim 3059 wherein the agent is an anthracycline.
3080. The device of claim 3059 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
3081. The device of claim 3059 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
3082. The device of claim 3059 wherein the agent is a platinum compound.
3083. The device of claim 3059 wherein the agent is a nitrosourea.
3084. The device of claim 3059 wherein the agent is a nitroimidazole.
3085. The device of claim 3059 wherein the agent is a folic acid antagonist.
3086. The device of claim 3059 wherein the agent is a cytidine analogue.
3087. The device of claim 3059 wherein the agent is a pyrimidine analogue.
3088. The device of claim 3059 wherein the agent is a fluoropyrimidine analogue.
3089. The device of claim 3059 wherein the agent is a purine analogue.
3090. The device of claim 3059 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
3091. The device of claim 3059 wherein the agent is a hydroxyurea.
3092. The device of claim 3059 wherein the agent is a mytomicin or an analogue or derivative thereof.
3093. The device of claim 3059 wherein the agent is an alkyl sulfonate.
3094. The device of claim 3059 wherein the agent is a benzamide or an analogue or derivative thereof.
3095. The device of claim 3059 wherein the agent is a nicotinamide or an analogue or derivative thereof.
3096. The device of claim 3059 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
3097. The device of claim 3059 wherein the agent is a DNA alkylating agent.
3098. The device of claim 3059 wherein the agent is an antimicrotubule agent.
3099. The device of claim 3059 wherein the agent is a topoisomerase inhibitor.
3100. The device of claim 3059 wherein the agent is a DNA cleaving agent.
3101. The device of claim 3059 wherein the agent is an antimetabolite.
3102. The device of claim 3059 wherein the agent inhibits adenosine deaminase.
3103. The device of claim 3059 wherein the agent inhibits purine ring synthesis.
3104. The device of claim 3059 wherein the agent is a nucleotide interconversion inhibitor.
3105. The device of claim 3059 wherein the agent inhibits dihydrofolate reduction.
3106. The device of claim 3059 wherein the agent blocks thymidine monophosphate.
3107. The device of claim 3059 wherein the agent causes DNA damage.
3108. The device of claim 3059 wherein the agent is a DNA intercalation agent.
3109. The device of claim 3059 wherein the agent is a RNA synthesis inhibitor.
3110. The device of claim 3059 wherein the agent is a pyrimidine synthesis inhibitor.
3111. The device of claim 3059 wherein the agent inhibits ribonucleotide synthesis or function.
3112. The device of claim 3059 wherein the agent inhibits thymidine monophosphate synthesis or function.
3113. The device of claim 3059 wherein the agent inhibits DNA synthesis.
3114. The device of claim 3059 wherein the agent causes DNA adduct formation.
3115. The device of claim 3059 wherein the agent inhibits protein synthesis.
3116. The device of claim 3059 wherein the agent inhibits microtubule function.
3117. The device of claim 3059 wherein the agent is a cyclin dependent protein kinase inhibitor.
3118. The device of claim 3059 wherein the agent is an epidermal growth factor kinase inhibitor.
3119. The device of claim 3059 wherein the agent is an elastase inhibitor.
3120. The device of claim 3059 wherein the agent is a factor Xa inhibitor.
3121. The device of claim 3059 wherein the agent is a farnesyltransferase inhibitor.
3122. The device of claim 3059 wherein the agent is a fibrinogen antagonist.
3123. The device of claim 3059 wherein the agent is a guanylate cyclase stimulant.
3124. The device of claim 3059 wherein the agent is a heat shock protein 90 antagonist.
3125. The device of claim 3059 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
3126. The device of claim 3059 wherein the agent is a guanylate cyclase stimulant.
3127. The device of claim 3059 wherein the agent is a HMGCoA reductase inhibitor.
3128. The device of claim 3059 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
3129. The device of claim 3059 wherein the agent is a hydroorotate dehydrogenase inhibitor.
3130. The device of claim 3059 wherein the agent is an IKK2 inhibitor.
3131. The device of claim 3059 wherein the agent is an IL-1 antagonist.
3132. The device of claim 3059 wherein the agent is an ICE antagonist.
3133. The device of claim 3059 wherein the agent is an IRAK antagonist.
3134. The device of claim 3059 wherein the agent is an IL-4 agonist.
3135. The device of claim 3059 wherein the agent is an immunomodulatory agent.
3136. The device of claim 3059 wherein the agent is sirolimus or an analogue or derivative thereof.
3137. The device of claim 3059 wherein the agent is not sirolimus.
3138. The device of claim 3059 wherein the agent is everolimus or an analogue or derivative thereof.
3139. The device of claim 3059 wherein the agent is tacrolimus or an analogue or derivative thereof.
3140. The device of claim 3059 wherein the agent is not tacrolimus.
3141. The device of claim 3059 wherein the agent is biolmus or an analogue or derivative thereof.
3142. The device of claim 3059 wherein the agent is tresperimus or an analogue or derivative thereof.
3143. The device of claim 3059 wherein the agent is auranofin or an analogue or derivative thereof.
3144. The device of claim 3059 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
3145. The device of claim 3059 wherein the agent is gusperimus or an analogue or derivative thereof.
3146. The device of claim 3059 wherein the agent is pimecrolimus or an analogue or derivative thereof.
3147. The device of claim 3059 wherein the agent is ABT-578 or an analogue or derivative thereof.
3148. The device of claim 3059 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
3149. The device of claim 3059 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
3150. The device of claim 3059 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
3151. The device of claim 3059 wherein the agent is a leukotriene inhibitor.
3152. The device of claim 3059 wherein the agent is a MCP-1 antagonist.
3153. The device of claim 3059 wherein the agent is a MMP inhibitor.
3154. The device of claim 3059 wherein the agent is an NF kappa B inhibitor.
3155. The device of claim 3059 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
3156. The device of claim 3059 wherein the agent is an NO antagonist.
3157. The device of claim 3059 wherein the agent is a p38 MAP kinase inhibitor.
3158. The device of claim 3059 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
3159. The device of claim 3059 wherein the agent is a phosphodiesterase inhibitor.
3160. The device of claim 3059 wherein the agent is a TGF beta inhibitor.
3161. The device of claim 3059 wherein the agent is a thromboxane A2 antagonist.
3162. The device of claim 3059 wherein the agent is a TNF alpha antagonist.
3163. The device of claim 3059 wherein the agent is a TACE inhibitor.
3164. The device of claim 3059 wherein the agent is a tyrosine kinase inhibitor.
3165. The device of claim 3059 wherein the agent is a vitronectin inhibitor.
3166. The device of claim 3059 wherein the agent is a fibroblast growth factor inhibitor.
3167. The device of claim 3059 wherein the agent is a protein kinase inhibitor.
3168. The device of claim 3059 wherein the agent is a PDGF receptor kinase inhibitor.
3169. The device of claim 3059 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
3170. The device of claim 3059 wherein the agent is a retinoic acid receptor antagonist.
3171. The device of claim 3059 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
3172. The device of claim 3059 wherein the agent is a fibrinogen antagonist.
3173. The device of claim 3059 wherein the agent is an antimycotic agent.
3174. The device of claim 3059 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
3175. The device of claim 3059 wherein the agent is a bisphosphonate.
3176. The device of claim 3059 wherein the agent is a phospholipase A1 inhibitor.
3177. The device of claim 3059 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
3178. The device of claim 3059 wherein the agent is a macrolide antibiotic.
3179. The device of claim 3059 wherein the agent is a GPIIb/llla receptor antagonist.
3180. The device of claim 3059 wherein the agent is an endothelin receptor antagonist.
3181. The device of claim 3059 wherein the agent is a peroxisome proliferator-activated receptor agonist.
3182. The device of claim 3059 wherein the agent is an estrogen receptor agent.
3183. The device of claim 3059 wherein the agent is a somastostatin analogue.
3184. The device of claim 3059 wherein the agent is a neurokinin 1 antagonist.
3185. The device of claim 3059 wherein the agent is a neurokinin 3 antagonist.
3186. The device of claim 3059 wherein the agent is a VLA-4 antagonist.
3187. The device of claim 3059 wherein the agent is an osteoclast inhibitor.
3188. The device of claim 3059 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
3189. The device of claim 3059 wherein the agent is an angiotensin I converting enzyme inhibitor.
3190. The device of claim 3059 wherein the agent is an angiotensin II antagonist.
3191. The device of claim 3059 wherein the agent is an enkephalinase inhibitor.
3192. The device of claim 3059 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
3193. The device of claim 3059 wherein the agent is a protein kinase C inhibitor.
3194. The device of claim 3059 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
3195. The device of claim 3059 wherein the agent is a CXCR3 inhibitor.
3196. The device of claim 3059 wherein the agent is an Itk inhibitor.
3197. The device of claim 3059 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
3198. The device of claim 3059 wherein the agent is a PPAR agonist.
3199. The device of claim 3059 wherein the agent is an immunosuppressant.
3200. The device of claim 3059 wherein the agent is an Erb inhibitor.
3201. The device of claim 3059 wherein the agent is an apoptosis agonist.
3202. The device of claim 3059 wherein the agent is a lipocortin agonist.
3203. The device of claim 3059 wherein the agent is a VCAM-1 antagonist.
3204. The device of claim 3059 wherein the agent is a collagen antagonist.
3205. The device of claim 3059 wherein the agent is an alpha 2 integrin antagonist.
3206. The device of claim 3059 wherein the agent is a TNF alpha inhibitor.
3207. The device of claim 3059 wherein the agent is a nitric oxide inhibitor
3208. The device of claim 3059 wherein the agent is a cathepsin inhibitor.
3209. The device of claim 3059 wherein the agent is not an anti- inflammatory agent.
3210. The device of claim 3059 wherein the agent is not a steroid.
3211. The device of claim 3059 wherein the agent is not a glucocorticosteroid.
3212. The device of claim 3059 wherein the agent is not dexamethasone.
3213. The device of claim 3059 wherein the agent is not an anti- infective agent.
3214. The device of claim 3059 wherein the agent is not an antibiotic.
3215. The device of claim 3059 wherein the agent is not an anti- fugal agent.
3216. The device of claim 3059 wherein the agent is not beclomethasone.
3217. The device of claim 3059 wherein the agent is not dipropionate.
3218. The device of claim 3059, further comprising a coating, wherein the coating comprises the anti-scarring agent and a polymer.
3219. The device of claim 3059, further comprising a coating, wherein the coating comprises the anti-scarring agent.
3220. The device of claim 3059, further comprising a coating, wherein the coating is disposed on a surface of the device.
3221. The device of claim 3059, further comprising a coating, wherein the coating directly contacts the device.
3222. The device of claim 3059, further comprising a coating, wherein the coating indirectly contacts the device.
3223. The device of claim 3059, further comprising a coating, wherein the coating partially covers the device.
3224. The device of claim 3059, further comprising a coating, wherein the coating completely covers the device.
3225. The device of claim 3059, further comprising a coating, wherein the coating is a uniform coating.
3226. The device of claim 3059, further comprising a coating, wherein the coating is a non-uniform coating.
3227. The device of claim 3059, further comprising a coating, wherein the coating is a discontinuous coating.
3228. The device of claim 3059, further comprising a coating, wherein the coating is a patterned coating.
3229. The device of claim 3059, further comprising a coating, wherein the coating has a thickness of 100 μm or less.
3230. The device of claim 3059, further comprising a coating, wherein the coating has a thickness of 10 μm or less.
3231. The device of claim 3059, further comprising a coating, wherein the coating adheres to the surface of the device upon deployment of the device.
3232. The device of claim 3059, further comprising a coating, wherein the coating is stable at room temperature for a period of 1 year.
3233. The device of claim 3059, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1% by weight.
3234. The device of claim 3059, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
3235. The device of claim 3059, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
3236. The device of claim 3059, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
3237. The device of claim 3059, further comprising a coating, wherein the coating further comprises a polymer.
3238. The device of claim 3059, further comprising a first coating having a first composition and the second coating having a second composition.
3239. The device of claim 3059, further comprising a first coating having a first composition and the second coating having a second composition, wherein the first composition and the second composition are different.
3240. The device of claim 3059, further comprising a polymer.
3241. The device of claim 3059, further comprising a polymeric carrier.
3242. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a copolymer.
3243. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a block copolymer.
3244. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a random copolymer.
3245. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a biodegradable polymer.
3246. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-biodegradable polymer.
3247. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophilic polymer.
3248. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophobic polymer.
3249. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophilic domains.
3250. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophobic domains.
3251. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-conductive polymer.
3252. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises an elastomer.
3253. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrogel.
3254. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a silicone polymer.
3255. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrocarbon polymer.
3256. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a styrene-derived polymer.
3257. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a butadiene polymer.
3258. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a macromer.
3259. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
3260. The device of claim 3059, further comprising a polymeric carrier, wherein the polymeric carrier comprises an amorphous polymer.
3261. The device of claim 3059, further comprising a lubricious coating.
3262. The device of claim 3059 wherein the anti-scarring agent is located within pores or holes of the device.
3263. The device of claim 3059 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
3264. The device of claim 3059, further comprising a second pharmaceutically active agent.
3265. The device of claim 3059, further comprising an anti- inflammatory agent.
3266. The device of claim 3059, further comprising an agent that inhibits infection.
3267. The device of claim 3059, further comprising an agent that inhibits infection, wherein the agent is an anthracycline.
3268. The device of claim 3059, further comprising an agent that inhibits infection, wherein the agent is doxorubicin.
3269. The device of claim 3059, further comprising an agent that inhibits infection, wherein the agent is mitoxantrone.
3270. The device of claim 3059, further comprising an agent that inhibits infection, wherein the agent is a fluoropyrimidine.
3271. The device of claim 3059, further comprising an agent that inhibits infection, wherein the agent is 5-fluorouracil (5-FU).
3272. The device of claim 3059, further comprising an agent that inhibits infection, wherein the agent is a folic acid antagonist.
3273. The device of claim 3059, further comprising an agent that inhibits infection, wherein the agent is methotrexate.
3274. The device of claim 3059, further comprising an agent that inhibits infection, wherein the agent is a podophylotoxin.
3275. The device of claim 3059, further comprising an agent that inhibits infection, wherein the agent is etoposide.
3276. The device of claim 3059, further comprising an agent that inhibits infection, wherein the agent is a camptothecin.
3277. The device of claim 3059, further comprising an agent that inhibits infection, wherein the agent is a hydroxyurea.
3278. The device of claim 3059, further comprising an agent that inhibits infection, wherein the agent is a platinum complex.
3279. The device of claim 3059, further comprising an agent that inhibits infection, wherein the agent is cisplatin.
3280. The device of claim 3059, further comprising an anti- thrombotic agent.
3281. The device of claim 3059, further comprising a visualization agent.
3282. The device of claim 3059, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
3283. The device of claim 3059, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises barium, tantalum, or technetium.
3284. The device of claim 3059, further comprising a visualization agent, wherein the visualization agent is a MRI responsive material.
3285. The device of claim 3059, further comprising a visualization agent, wherein the visualization agent comprises a gadolinium chelate.
3286. The device of claim 3059, further comprising a visualization agent, wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
3287. The device of claim 3059, further comprising a visualization agent, wherein the visualization agent comprises an iron oxide compound.
3288. The device of claim 3059, further comprising a visualization agent, wherein the visualization agent comprises a dye, pigment, or colorant.
3289. The device of claim 3059, further comprising an echogenic material.
3290. The device of claim 3059, further comprising an echogenic material, wherein the echogenic material is in the form of a coating.
3291. The device of claim 3059 wherein the device is sterile.
3292. The device of claim 3059 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
3293. The device of claim 3059 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
3294. The device of claim 3059 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
3295. The device of claim 3059 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is connective tissue.
3296. The device of claim 3059 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is muscle tissue.
3297. The device of claim 3059 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is nerve tissue.
3298. The device of claim 3059 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is epithelium tissue.
3299. The device of claim 3059 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
3300. The device of claim 3059 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
3301. The device of claim 3059 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
3302. The device of claim 3059 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
3303. The device of claim 3059 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
3304. The device of claim 3059 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
3305. The device of claim 3059 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
3306. The device of claim 3059 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
3307. The device of claim 3059 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
3308. The device of claim 3059 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
3309. The device of claim 3059 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent
3310. The device of claim 3059 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
3311. The device of claim 3059 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent
3312. The device of claim 3059 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3313. The device of claim 3059 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3314. The device of claim 3059 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3315. The device of claim 3059 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3316. The device of claim 3059 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3317. The device of claim 3059 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3318. The device of claim 3059 wherein the agent or the composition is affixed to the pump.
3319. The device of claim 3059 wherein the agent or the composition is covalently attached to the pump.
3320. The device of claim 3059 wherein the agent or the composition is non-covalently attached to the pump.
3321. The device of claim 3059 further comprising a coating that absorbs the agent or the composition.
3322. The device of claim 3059 wherein the pump is interweaved with a thread composed of, or coated with, the agent or the composition.
3323. The device of claim 3059 wherein a portion of the pump is covered with a sleeve that contains the agent or the composition.
3324. The device of claim 3059 wherein the pump is completely covered with a sleeve that contains the agent or the composition.
3325. The device of claim 3059 wherein a portion of the pump is covered with a mesh that contains the agent or the composition.
3326. The device of claim 3059 wherein the pump is completely covered with a mesh that contains the agent or the composition.
3327. The device of claim 3059 further comprising a sensor that is linked to the pump.
3328. The device of any one of claims 3059-3327 wherein the device is an implantable cardiac electrode that delivers stimulation energy and dispenses drug adjacent to the stimulation site.
3329. A device, comprising a drug delivery implant (i.e., a pump) and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
3330. The device of claim 3329 wherein the agent inhibits cell regeneration.
3331. The device of claim 3329 wherein the agent inhibits angiogenesis.
3332. The device of claim 3329 wherein the agent inhibits fibroblast migration.
3333. The device of claim 3329 wherein the agent inhibits fibroblast proliferation.
3334. The device of claim 3329 wherein the agent inhibits deposition of extracellular matrix.
3335. The device of claim 3329 wherein the agent inhibits tissue remodeling.
3336. The device of claim 3329 wherein the agent is an angiogenesis inhibitor.
3337. The device of claim 3329 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
3338. The device of claim 3329 wherein the agent is a chemokine receptor antagonist.
3339. The device of claim 3329 wherein the agent is a cell cycle inhibitor.
3340. The device of claim 3329 wherein the agent is a taxane.
3341. The device of claim 3329 wherein the agent is an antimicrotubule agent.
3342. The device of claim 3329 wherein the agent is paclitaxel.
3343. The device of claim 3329 wherein the agent is not paclitaxel.
3344. The device of claim 3329 wherein the agent is an analogue or derivative of paclitaxel.
3345. The device of claim 3329 wherein the agent is a vinca alkaloid.
3346. The device of claim 3329 wherein the agent is camptothecin or an analogue or derivative thereof.
3347. The device of claim 3329 wherein the agent is a podophyllotoxin.
3348. The device of claim 3329 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
3349. The device of claim 3329 wherein the agent is an anthracycline.
3350. The device of claim 3329 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
3351. The device of claim 3329 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
3352. The device of claim 3329 wherein the agent is a platinum compound.
3353. The device of claim 3329 wherein the agent is a nitrosourea.
3354. The device of claim 3329 wherein the agent is a nitroimidazole.
3355. The device of claim 3329 wherein the agent is a folic acid antagonist.
3356. The device of claim 3329 wherein the agent is a cytidine analogue.
3357. The device of claim 3329 wherein the agent is a pyrimidine analogue.
3358. The device of claim 3329 wherein the agent is a fluoropyrimidine analogue.
3359. The device of claim 3329 wherein the agent is a purine analogue.
3360. The device of claim 3329 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
3361. The device of claim 3329 wherein the agent is a hydroxyurea.
3362. The device of claim 3329 wherein the agent is a mytomicin or an analogue or derivative thereof.
3363. The device of claim 3329 wherein the agent is an alkyl sulfonate.
3364. The device of claim 3329 wherein the agent is a benzamide or an analogue or derivative thereof.
3365. The device of claim 3329 wherein the agent is a nicotinamide or an analogue or derivative thereof.
3366. The device of claim 3329 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
3367. The device of claim 3329 wherein the agent is a DNA alkylating agent.
3368. The device of claim 3329 wherein the agent is an antimicrotubule agent.
3369. The device of claim 3329 wherein the agent is a topoisomerase inhibitor.
3370. The device of claim 3329 wherein the agent is a DNA cleaving agent.
3371. The device of claim 3329 wherein the agent is an antimetabolite.
3372. The device of claim 3329 wherein the agent inhibits adenosine deaminase.
3373. The device of claim 3329 wherein the agent inhibits purine ring synthesis.
3374. The device of claim 3329 wherein the agent is a nucleotide interconversion inhibitor.
3375. The device of claim 3329 wherein the agent inhibits dihydrofolate reduction.
3376. The device of claim 3329 wherein the agent blocks thymidine monophosphate.
3377. The device of claim 3329 wherein the agent causes DNA damage.
3378. The device of claim 3329 wherein the agent is a DNA intercalation agent.
3379. The device of claim 3329 wherein the agent is a RNA synthesis inhibitor.
3380. The device of claim 3329 wherein the agent is a pyrimidine synthesis inhibitor.
3381. The device of claim 3329 wherein the agent inhibits ribonucleotide synthesis or function.
3382. The device of claim 3329 wherein the agent inhibits thymidine monophosphate synthesis or function.
3383. The device of claim 3329 wherein the agent inhibits DNA synthesis.
3384. The device of claim 3329 wherein the agent causes DNA adduct formation.
3385. The device of claim 3329 wherein the agent inhibits protein synthesis.
3386. The device of claim 3329 wherein the agent inhibits microtubule function.
3387. The device of claim 3329 wherein the agent is a cyclin dependent protein kinase inhibitor.
3388. The device of claim 3329 wherein the agent is an epidermal growth factor kinase inhibitor.
3389. The device of claim 3329 wherein the agent is an elastase inhibitor.
3390. The device of claim 3329 wherein the agent is a factor Xa inhibitor.
3391. The device of claim 3329 wherein the agent is a farnesyltransferase inhibitor.
3392. The device of claim 3329 wherein the agent is a fibrinogen antagonist.
3393. The device of claim 3329 wherein the agent is a guanylate cyclase stimulant.
3394. The device of claim 3329 wherein the agent is a heat shock protein 90 antagonist.
3395. The device of claim 3329 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
3396. The device of claim 3329 wherein the agent is a guanylate cyclase stimulant.
3397. The device of claim 3329 wherein the agent is a HMGCoA reductase inhibitor.
3398. The device of claim 3329 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
3399. The device of claim 3329 wherein the agent is a hydroorotate dehydrogenase inhibitor.
3400. The device of claim 3329 wherein the agent is an IKK2 inhibitor.
3401. The device of claim 3329 wherein the agent is an IL-1 antagonist.
3402. The device of claim 3329 wherein the agent is an ICE antagonist.
3403. The device of claim 3329 wherein the agent is an IRAK antagonist.
3404. The device of claim 3329 wherein the agent is an IL-4 agonist.
3405. The device of claim 3329 wherein the agent is an immunomodulatory agent.
3406. The device of claim 3329 wherein the agent is sirolimus or an analogue or derivative thereof.
3407. The device of claim 3329 wherein the agent is not sirolimus.
3408. The device of claim 3329 wherein the agent is everolimus or an analogue or derivative thereof.
3409. The device of claim 3329 wherein the agent is tacrolimus or an analogue or derivative thereof.
3410. The device of claim 3329 wherein the agent is not tacrolimus.
3411. The device of claim 3329 wherein the agent is biolmus or an analogue or derivative thereof. 2005/051871
3412. The device of claim 3329 wherein the agent is tresperimus or an analogue or derivative thereof.
3413. The device of claim 3329 wherein the agent is auranofin or an analogue or derivative thereof.
3414. The device of claim 3329 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
3415. The device of claim 3329 wherein the agent is gusperimus or an analogue or derivative thereof.
3416. The device of claim 3329 wherein the agent is pimecrolimus or an analogue or derivative thereof.
3417. The device of claim 3329 wherein the agent is ABT-578 or an analogue or derivative thereof.
3418. The device of claim 3329 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
3419. The device of claim 3329 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
3420. The device of claim 3329 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
3421. The device of claim 3329 wherein the agent is a leukotriene inhibitor.
3422. The device of claim 3329 wherein the agent is a MCP-1 antagonist
3423. The device of claim 3329 wherein the agent is a MMP inhibitor.
3424. The device of claim 3329 wherein the agent is an NF kappa B inhibitor.
3425. The device of claim 3329 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
3426. The device of claim 3329 wherein the agent is an NO antagonist.
3427. The device of claim 3329 wherein the agent is a p38 MAP kinase inhibitor.
3428. The device of claim 3329 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
3429. The device of claim 3329 wherein the agent is a phosphodiesterase inhibitor.
3430. The device of claim 3329 wherein the agent is a TGF beta inhibitor.
3431. The device of claim 3329 wherein the agent is a thromboxane A2 antagonist.
3432. The device of claim 3329 wherein the agent is a TNF alpha antagonist.
3433. The device of claim 3329 wherein the agent is a TACE inhibitor.
3434. The device of claim 3329 wherein the agent is a tyrosine kinase inhibitor.
3435. The device of claim 3329 wherein the agent is a vitronectin inhibitor.
3436. The device of claim 3329 wherein the agent is a fibroblast growth factor inhibitor.
3437. The device of claim 3329 wherein the agent is a protein kinase inhibitor.
3438. The device of claim 3329 wherein the agent is a PDGF receptor kinase inhibitor.
3439. The device of claim 3329 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
3440. The device of claim 3329 wherein the agent is a retinoic acid receptor antagonist.
3441. The device of claim 3329 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
3442. The device of claim 3329 wherein the agent is a fibrinogen antagonist.
3443. The device of claim 3329 wherein the agent is an antimycotic agent.
3444. The device of claim 3329 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
3445. The device of claim 3329 wherein the agent is a bisphosphonate.
3446. The device of claim 3329 wherein the agent is a phospholipase A1 inhibitor.
3447. The device of claim 3329 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
3448. The device of claim 3329 wherein the agent is a macrolide antibiotic.
3449. The device of claim 3329 wherein the agent is a GPIIb/llla receptor antagonist.
3450. The device of claim 3329 wherein the agent is an endothelin receptor antagonist.
3451. The device of claim 3329 wherein the agent is a peroxisome proliferator-activated receptor agonist.
3452. The device of claim 3329 wherein the agent is an estrogen receptor agent.
3453. The device of claim 3329 wherein the agent is a somastostatin analogue.
3454. The device of claim 3329 wherein the agent is a neurokinin 1 antagonist.
3455. The device of claim 3329 wherein the agent is a neurokinin 3 antagonist.
3456. The device of claim 3329 wherein the agent is a VLA-4 antagonist.
3457. The device of claim 3329 wherein the agent is an osteoclast inhibitor.
3458. The device of claim 3329 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
3459. The device of claim 3329 wherein the agent is an angiotensin I converting enzyme inhibitor.
3460. The device of claim 3329 wherein the agent is an angiotensin II antagonist.
3461. The device of claim 3329 wherein the agent is an enkephalinase inhibitor.
3462. The device of claim 3329 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
3463. The device of claim 3329 wherein the agent is a protein kinase C inhibitor.
3464. The device of claim 3329 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
3465. The device of claim 3329 wherein the agent is a CXCR3 inhibitor.
3466. The device of claim 3329 wherein the agent is an Itk inhibitor.
3467. The device of claim 3329 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
3468. The device of claim 3329 wherein the agent is a PPAR agonist.
3469. The device of claim 3329 wherein the agent is an immunosuppressant
3470. The device of claim 3329 wherein the agent is an Erb inhibitor.
3471. The device of claim 3329 wherein the agent is an apoptosis agonist.
3472. The device of claim 3329 wherein the agent is a lipocortin agonist.
3473. The device of claim 3329 wherein the agent is a VCAM-1 antagonist.
3474. The device of claim 3329 wherein the agent is a collagen antagonist.
3475. The device of claim 3329 wherein the agent is an alpha 2 integrin antagonist.
3476. The device of claim 3329 wherein the agent is a TNF alpha inhibitor.
3477. The device of claim 3329 wherein the agent is a nitric oxide inhibitor
3478. The device of claim 3329 wherein the agent is a cathepsin inhibitor.
3479. The device of claim 3329 wherein the agent is not an anti- inflammatory agent.
3480. The device of claim 3329 wherein the agent is not a steroid.
3481. The device of claim 3329 wherein the agent is not a glucocorticosteroid.
3482. The device of claim 3329 wherein the agent is not dexamethasone.
3483. The device of claim 3329 wherein the agent is not an anti- infective agent.
3484. The device of claim 3329 wherein the agent is not an antibiotic.
3485. The device of claim 3329 wherein the agent is not an anti- fugal agent.
3486. The device of claim 3329 wherein the agent is not beclomethasone.
3487. The device of claim 3329 wherein the agent is not dipropionate.
3488. The device of claim 3329, further comprising a coating, wherein the coating comprises the anti-scarring agent and a polymer.
3489. The device of claim 3329, further comprising a coating, wherein the coating comprises the anti-scarring agent.
3490. The device of claim 3329, further comprising a coating, wherein the coating is disposed on a surface of the device.
3491. The device of claim 3329, further comprising a coating, wherein the coating directly contacts the device.
3492. The device of claim 3329, further comprising a coating, wherein the coating indirectly contacts the device.
3493. The device of claim 3329, further comprising a coating, wherein the coating partially covers the device.
3494. The device of claim 3329, further comprising a coating, wherein the coating completely covers the device.
3495. The device of claim 3329, further comprising a coating, wherein the coating is a uniform coating.
3496. The device of claim 3329, further comprising a coating, wherein the coating is a non-uniform coating.
3497. The device of claim 3329, further comprising a coating, wherein the coating is a discontinuous coating.
3498. The device of claim 3329, further comprising a coating, wherein the coating is a patterned coating.
3499. The device of claim 3329, further comprising a coating, wherein the coating has a thickness of 100 μm or less.
3500. The device of claim 3329, further comprising a coating, wherein the coating has a thickness of 10 μm or less.
3501. The device of claim 3329, further comprising a coating, wherein the coating adheres to the surface of the device upon deployment of the device.
3502. The device of claim 3329, further comprising a coating, wherein the coating is stable at room temperature for a period of 1 year.
3503. The device of claim 3329, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001 % to about 1% by weight.
3504. The device of claim 3329, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
3505. The device of claim 3329, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight
3506. The device of claim 3329, further comprising a coating, wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
3507. The device of claim 3329, further comprising a coating, wherein the coating further comprises a polymer. 2005/051871
3508. The device of claim 3329, further comprising a first coating having a first composition and the second coating having a second composition.
3509. The device of claim 3329, further comprising a first coating having a first composition and the second coating having a second composition, wherein the first composition and the second composition are different.
3510. The device of claim 3329, further comprising a polymer.
3511. The device of claim 3329, further comprising a polymeric carrier.
3512. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a copolymer.
3513. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a block copolymer.
3514. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a random copolymer.
3515. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a biodegradable polymer.
3516. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-biodegradable polymer.
3517. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophilic polymer. 2005/051871
3518. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrophobic polymer.
3519. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophilic domains.
3520. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a polymer having hydrophobic domains.
3521. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a non-conductive polymer.
3522. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises an elastomer.
3523. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrogel.
3524. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a silicone polymer.
3525. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a hydrocarbon polymer.
3526. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a styrene-derived polymer.
3527. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a butadiene polymer.
3528. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a macromer.
3529. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
3530. The device of claim 3329, further comprising a polymeric carrier, wherein the polymeric carrier comprises an amorphous polymer.
3531. The device of claim 3329, further comprising a lubricious coating.
3532. The device of claim 3329 wherein the anti-scarring agent is located within pores or holes of the device.
3533. The device of claim 3329 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
3534. The device of claim 3329, further comprising a second pharmaceutically active agent.
3535. The device of claim 3329, further comprising an anti- inflammatory agent.
3536. The device of claim 3329, further comprising an agent that inhibits infection.
3537. The device of claim 3329, further comprising an agent that inhibits infection, wherein the agent is an anthracycline.
3538. The device of claim 3329, further comprising an agent that inhibits infection, wherein the agent is doxorubicin.
3539. The device of claim 3329, further comprising an agent that inhibits infection, wherein the agent is mitoxantrone.
3540. The device of claim 3329, further comprising an agent that inhibits infection, wherein the agent is a fluoropyrimidine.
3541. The device of claim 3329, further comprising an agent that inhibits infection, wherein the agent is 5-fluorouracil (5-FU).
3542. The device of claim 3329, further comprising an agent that inhibits infection, wherein the agent is a folic acid antagonist.
3543. The device of claim 3329, further comprising an agent that inhibits infection, wherein the agent is methotrexate.
3544. The device of claim 3329, further comprising an agent that inhibits infection, wherein the agent is a podophylotoxin.
3545. The device of claim 3329, further comprising an agent that inhibits infection, wherein the agent is etoposide.
3546. The device of claim 3329, further comprising an agent that inhibits infection, wherein the agent is a camptothecin.
3547. The device of claim 3329, further comprising an agent that inhibits infection, wherein the agent is a hydroxyurea.
3548. The device of claim 3329, further comprising an agent that inhibits infection, wherein the agent is a platinum complex.
3549. The device of claim 3329, further comprising an agent that inhibits infection, wherein the agent is cisplatin.
3550. The device of claim 3329, further comprising an anti- thrombotic agent.
3551. The device of claim 3329, further comprising a visualization agent.
3552. The device of claim 3329, further comprising a visualization agent, wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
3553. The device of claim 3329, further comprising a visualization agent, 'Wherein the visualization agent is a radiopaque material, wherein the radiopaque material comprises barium, tantalum, ortechnetium.
3554. The device of claim 3329, further comprising a visualization agent, wherein the visualization agent is a MRI responsive material.
3555. The device of claim 3329, further comprising a visualization agent, wherein the visualization agent comprises a gadolinium chelate.
3556. The device of claim 3329, further comprising a visualization agent, wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
3557. The device of claim 3329, further comprising a visualization agent, wherein the visualization agent comprises an iron oxide compound.
3558. The device of claim 3329, further comprising a visualization agent, wherein the visualization agent comprises a dye, pigment, or colorant.
3559. The device of claim 3329, further comprising an echogenic material.
3560. The device of claim 3329, further comprising an echogenic material, wherein the echogenic material is in the form of a coating.
3561. The device of claim 3329 wherein the device is sterile.
3562. The device of claim 3329 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
3563. The device of claim 3329 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
3564. The device of claim 3329 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
3565. The device of claim 3329 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is connective tissue.
3566. The device of claim 3329 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is muscle tissue.
3567. The device of claim 3329 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is nerve tissue.
3568. The device of claim 3329 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, wherein the tissue is epithelium tissue.
3569. The device of claim 3329 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
3570. The device of claim 3329 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
3571. The device of claim 3329 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
3572. The device of claim 3329 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
3573. The device of claim 3329 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
3574. The device of claim 3329 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
3575. The device of claim 3329 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
3576. The device of claim 3329 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti- scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
3577. The device of claim 3329 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
3578. The device of claim 3329 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
3579. The device of claim 3329 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
3580. The device of claim 3329 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
3581. The device of claim 3329 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
3582. The device of claim 3329 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3583. The device of claim 3329 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3584. The device of claim 3329 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3585. The device of claim 3329 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3586. The device of claim 3329 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3587. The device of claim 3329 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
3588. The device of claim 3329 wherein the agent or the composition is affixed to the pump.
3589. The device of claim 3329 wherein the agent or the composition is covalently attached to the pump.
3590. The device of claim 3329 wherein the agent or the composition is non-covalently attached to the pump.
3591. The device of claim 3329 further comprising a coating that absorbs the agent or the composition.
3592. The device of claim 3329 wherein the pump is interweaved with a thread composed of, or coated with, the agent or the composition.
3593. The device of claim 3329 wherein a portion of the pump is covered with a sleeve that contains the agent or the composition.
3594. The device of claim 3329 wherein the pump is completely covered with a sleeve that contains the agent or the composition.
3595. The device of claim 3329 wherein a portion of the pump is covered with a mesh that contains the agent or the composition.
3596. The device of claim 3329 wherein the pump is completely covered with a mesh that contains the agent or the composition.
3597. The device of claim 3329 further comprising a sensor that is linked to the pump.
3598. A method for inhibiting scarring comprising placing a sensor and an anti-scarring agent or a composition comprising an anti-scarring agent into an animal host, wherein the agent inhibits scarring.
3599. The method of claim 3598 wherein the agent inhibits cell regeneration.
3600. The method of claim 3598 wherein the agent inhibits angiogenesis.
3601. The method of claim 3598 wherein the agent inhibits fibroblast migration.
3602. The method of claim 3598 wherein the agent inhibits fibroblast proliferation.
3603. The method of claim 3598 wherein the agent inhibits deposition of extracellular matrix.
3604. The method of claim 3598 wherein the agent inhibits tissue remodeling.
3605. The method of claim 3598 wherein the agent is an angiogenesis inhibitor.
3606. The method of claim 3598 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
3607. The method of claim 3598 wherein the agent is a chemokine receptor antagonist.
3608. The method of claim 3598 wherein the agent is a cell cycle inhibitor.
3609. The method of claim 3598 wherein the agent is a taxane.
3610. The method of claim 3598 wherein the agent is an antimicrotubule agent.
3611. The method of claim 3598 wherein the agent is paclitaxel.
3612. The method of claim 3598 wherein the agent is not paclitaxel.
3613. The method of claim 3598 wherein the agent is an analogue or derivative of paclitaxel.
3614. The method of claim 3598 wherein the agent is a vinca alkaloid.
3615. The method of claim 3598 wherein the agent is camptothecin or an analogue or derivative thereof.
3616. The method of claim 3598 wherein the agent is a podophyllotoxin.
3617. The method of claim 3598 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
3618. The method of claim 3598 wherein the agent is an anthracycline.
3619. The method of claim 3598 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
3620. The method of claim 3598 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
3621. The method of claim 3598 wherein the agent is a platinum compound.
3622. The method of claim 3598 wherein the agent is a nitrosourea.
3623. The method of claim 3598 wherein the agent is a nitroimidazole.
3624. The method of claim 3598 wherein the agent is a folic acid antagonist. 2005/051871
3625. The method of claim 3598 wherein the agent is a cytidine analogue.
3626. The method of claim 3598 wherein the agent is a pyrimidine analogue.
3627. The method of claim 3598 wherein the agent is a fluoropyrimidine analogue.
3628. The method of claim 3598 wherein the agent is a purine analogue.
3629. The method of claim 3598 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
3630. The method of claim 3598 wherein the agent is a hydroxyurea.
3631. The method of claim 3598 wherein the agent is a mytomicin or an analogue or derivative thereof.
3632. The method of claim 3598 wherein the agent is an alkyl sulfonate.
3633. The method of claim 3598 wherein the agent is a benzamide or an analogue or derivative thereof.
3634. The method of claim 3598 wherein the agent is a nicotinamide or an analogue or derivative thereof.
3635. The method of claim 3598 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
3636. The method of claim 3598 wherein the agent is a DNA alkylating agent.
3637. The method of claim 3598 wherein the agent is an antimicrotubule agent.
3638. The method of claim 3598 wherein the agent is a topoisomerase inhibitor.
3639. The method of claim 3598 wherein the agent is a DNA cleaving agent.
3640. The method of claim 3598 wherein the agent is an antimetabolite.
3641. The method of claim 3598 wherein the agent inhibits adenosine deaminase.
3642. The method of claim 3598 wherein the agent inhibits purine ring synthesis.
3643. The method of claim 3598 wherein the agent is a nucleotide interconversion inhibitor.
3644. The method of claim 3598 wherein the agent inhibits dihydrofolate reduction.
3645. The method of claim 3598 wherein the agent blocks thymidine monophosphate.
3646. The method of claim 3598 wherein the agent causes DNA damage.
3647. The method of claim 3598 wherein the agent is a DNA intercalation agent.
3648. The method of claim 3598 wherein the agent is a RNA synthesis inhibitor.
3649. The method of claim 3598 wherein the agent is a pyrimidine synthesis inhibitor.
3650. The method of claim 3598 wherein the agent inhibits ribonucleotide synthesis or function.
3651. The method of claim 3598 wherein the agent inhibits thymidine monophosphate synthesis or function.
3652. The method of claim 3598 wherein the agent inhibits DNA synthesis.
3653. The method of claim 3598 wherein the agent causes DNA adduct formation.
3654. The method of claim 3598 wherein the agent inhibits protein synthesis.
3655. The method of claim 3598 wherein the agent inhibits microtubule function.
3656. The method of claim 3598 wherein the agent is a cyclin dependent protein kinase inhibitor.
3657. The method of claim 3598 wherein the agent is an epidermal growth factor kinase inhibitor.
3658. The method of claim 3598 wherein the agent is an elastase inhibitor.
3659. The method of claim 3598 wherein the agent is a factor Xa inhibitor.
3660. The method of claim 3598 wherein the agent is a farnesyltransferase inhibitor.
3661. The method of claim 3598 wherein the agent is a fibrinogen antagonist.
3662. The method of claim 3598 wherein the agent is a guanylate cyclase stimulant.
3663. The method of claim 3598 wherein the agent is a heat shock protein 90 antagonist
3664. The method of claim 3598 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
3665. The method of claim 3598 wherein the agent is a guanylate cyclase stimulant.
3666. The method of claim 3598 wherein the agent is a HMGCoA reductase inhibitor.
3667. The method of claim 3598 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
3668. The method of claim 3598 wherein the agent is a hydroorotate dehydrogenase inhibitor.
3669. The method of claim 3598 wherein the agent is an IKK2 inhibitor.
3670. The method of claim 3598 wherein the agent is an IL-1 antagonist.
3671. The method of claim 3598 wherein the agent is an ICE antagonist.
3672. The method of claim 3598 wherein the agent is an IRAK antagonist.
3673. The method of claim 3598 wherein the agent is an IL-4 agonist.
3674. The method of claim 3598 wherein the agent is an immunomodulatory agent.
3675. The method of claim 3598 wherein the agent is sirolimus or an analogue or derivative thereof.
3676. The method of claim 3598 wherein the agent is not sirolimus.
3677. The method of claim 3598 wherein the agent is everolimus or an analogue or derivative thereof.
3678. The method of claim 3598 wherein the agent is tacrolimus or an analogue or derivative thereof.
3679. The method of claim 3598 wherein the agent is not tacrolimus.
3680. The method of claim 3598 wherein the agent is biolmus or an analogue or derivative thereof.
3681. The method of claim 3598 wherein the agent is tresperimus or an analogue or derivative thereof.
3682. The method of claim 3598 wherein the agent is auranofin or an analogue or derivative thereof.
3683. The method of claim 3598 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
3684. The method of claim 3598 wherein the agent is gusperimus or an analogue or derivative thereof.
3685. The method of claim 3598 wherein the agent is pimecrolimus or an analogue or derivative thereof.
3686. The method of claim 3598 wherein the agent is ABT-578 or an analogue or derivative thereof.
3687. The method of claim 3598 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
3688. The method of claim 3598 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof. 2005/051871
3689. The method of claim 3598 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
3690. The method of claim 3598 wherein the agent is a leukotriene inhibitor.
3691. The method of claim 3598 wherein the agent is a MCP-1 antagonist.
3692. The method of claim 3598 wherein the agent is a MMP inhibitor.
3693. The method of claim 3598 wherein the agent is an NF kappa B inhibitor.
3694. The method of claim 3598 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
3695. The method of claim 3598 wherein the agent is an NO antagonist.
3696. The method of claim 3598 wherein the agent is a p38 MAP kinase inhibitor.
3697. The method of claim 3598 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
3698. The method of claim 3598 wherein the agent is a phosphodiesterase inhibitor.
3699. The method of claim 3598 wherein the agent is a TGF beta inhibitor.
3700. The method of claim 3598 wherein the agent is a thromboxane A2 antagonist.
3701. The method of claim 3598 wherein the agent is a TNF alpha antagonist.
3702. The method of claim 3598 wherein the agent is a TACE inhibitor.
3703. The method of claim 3598 wherein the agent is a tyrosine kinase inhibitor.
3704. The method of claim 3598 wherein the agent is a vitronectin inhibitor.
3705. The method of claim 3598 wherein the agent is a fibroblast growth factor inhibitor.
3706. The method of claim 3598 wherein the agent is a protein kinase inhibitor.
3707. The method of claim 3598 wherein the agent is a PDGF receptor kinase inhibitor.
3708. The method of claim 3598 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
3709. The method of claim 3598 wherein the agent is a retinoic acid receptor antagonist.
3710. The method of claim 3598 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
3711. The method of claim 3598 wherein the agent is a fibrinogen antagonist.
3712. The method of claim 3598 wherein the agent is an antimycotic agent.
3713. The method of claim 3598 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
3714. The method of claim 3598 wherein the agent is a bisphosphonate.
3715. The method of claim 3598 wherein the agent is a phospholipase A1 inhibitor.
3716. The method of claim 3598 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
3717. The method of claim 3598 wherein the agent is a macrolide antibiotic.
3718. The method of claim 3598 wherein the agent is a GPIIb/llla receptor antagonist.
3719. The method of claim 3598 wherein the agent is an endothelin receptor antagonist.
3720. The method of claim 3598 wherein the agent is a peroxisome proliferator-activated receptor agonist.
3721. The method of claim 3598 wherein the agent is an estrogen receptor agent.
3722. The method of claim 3598 wherein the agent is a somastostatin analogue.
3723. The method of claim 3598 wherein the agent is a neurokinin 1 antagonist.
3724. The method of claim 3598 wherein the agent is a neurokinin 3 antagonist.
3725. The method of claim 3598 wherein the agent is a VLA-4 antagonist.
3726. The method of claim 3598 wherein the agent is an osteoclast inhibitor.
3727. The method of claim 3598 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
3728. The method of claim 3598 wherein the agent is an angiotensin I converting enzyme inhibitor.
3729. The method of claim 3598 wherein the agent is an angiotensin II antagonist.
3730. The method of claim 3598 wherein the agent is an enkephalinase inhibitor.
3731. The method of claim 3598 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
3732. The method of claim 3598 wherein the agent is a protein kinase C inhibitor.
3733. The method of claim 3598 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
3734. The method of claim 3598 wherein the agent is a CXCR3 inhibitor.
3735. The method of claim 3598 wherein the agent is an Itk inhibitor.
3736. The method of claim 3598 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
3737. The method of claim 3598 wherein the agent is a PPAR agonist.
3738. The method of claim 3598 wherein the agent is an immunosuppressant.
3739. The method of claim 3598 wherein the agent is an Erb inhibitor.
3740. The method of claim 3598 wherein the agent is an apoptosis agonist.
3741. The method of claim 3598 wherein the agent is a lipocortin agonist.
3742. The method of claim 3598 wherein the agent is a VCAM-1 antagonist. 2005/051871
3743. The method of claim 3598 wherein the agent is a collagen antagonist
3744. The method of claim 3598 wherein the agent is an alpha 2 integrin antagonist.
3745. The method of claim 3598 wherein the agent is a TNF alpha inhibitor.
3746. The method of claim 3598 wherein the agent is a nitric oxide inhibitor
3747. The method of claim 3598 wherein the agent is a cathepsin inhibitor.
3748. The method of claim 3598 wherein the agent is not an anti- inflammatory agent
3749. The method of claim 3598 wherein the agent is not a steroid.
3750. The method of claim 3598 wherein the agent is not a glucocorticosteroid.
3751. The method of claim 3598 wherein the agent is not dexamethasone.
3752. The method of claim 3598 wherein the agent is not beclomethasone.
3753. The method of claim 3598 wherein the agent is not dipropionate.
3754. The method of claim 3598 wherein the agent is not an anti- infective agent.
3755. The method of claim 3598 wherein the agent is not an antibiotic.
3756. The method of claim 3598 wherein the agent is not an anti- fungal agent.
3757. The method of claim 3598, wherein the composition comprises a polymer.
3758. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a copolymer.
3759. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a block copolymer.
3760. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a random copolymer.
3761. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a biodegradable polymer.
3762. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-biodegradable polymer. '
3763. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophilic polymer. 2005/051871
3764. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophobic polymer.
3765. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophilic domains.
3766. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophobic domains.
3767. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-conductive polymer.
3768. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, an elastomer.
3769. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrogel.
3770. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a silicone polymer.
3771. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrocarbon polymer.
3772. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a styrene-derived polymer.
3773. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a butadiene-derived polymer.
3774. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a macromer.
3775. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, a poly(ethy!ene glycol) polymer.
3776. The method of claim 3598, wherein the composition comprises a polymer, and the polymer is, or comprises, an amorphous polymer.
3777. The method of claim 3598, wherein the composition further comprises a second pharmaceutically active agent.
3778. The method of claim 3598, wherein the composition further comprises an anti-inflammatory agent.
3779. The method of claim 3598, wherein the composition further comprises an agent that inhibits infection.
3780. The method of claim 3598, wherein the composition further comprises an anthracycline.
3781. The method of claim 3598, wherein the composition further comprises doxorubicin.
3782. The method of claim 3598 wherein the composition further comprises mitoxantrone.
3783. The method of claim 3598 wherein the composition further comprises a fluoropyrimidine.
3784. The method of claim 3598, wherein the composition further comprises 5-fluorouracil (5-FU).
3785. The method of claim 3598, wherein the composition further comprises a folic acid antagonist.
3786. The method of claim 3598, wherein the composition further comprises methotrexate.
3787. The method of claim 3598, wherein the composition further comprises a podophylotoxin.
3788. The method of claim 3598, wherein the composition further comprises etoposide.
3789. The method of claim 3598, wherein the composition further comprises camptothecin.
3790. The method of claim 3598, wherein the composition further comprises a hydroxyurea.
3791. The method of claim 3598, wherein the composition further comprises a platinum complex.
3792. The method of claim 3598, wherein the composition further comprises cisplatin.
3793. The method of claim 3598 wherein the composition further comprises an anti-thrombotic agent.
3794. The method of claim 3598, wherein the composition further comprises a visualization agent.
3795. The method of claim 3598, wherein the composition further comprises a visualization agent, and the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
3796. The method of claim 3598, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, barium, tantalum, or technetium.
3797. The method of claim 3598, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, an MRI responsive material.
3798. The method of claim 3598, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a gadolinium chelate.
3799. The method of claim 3598, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron, magnesium, manganese, copper, or chromium.
3800. The method of claim 3598, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron oxide compound.
3801. The method of claim 3598, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a dye, pigment, or colorant.
3802. The method of claim 3598 wherein the agent is released in effective concentrations from the composition comprising the agent by diffusion over a period ranging from the time of administration to about 90 days.
3803. The method of claim 3598 wherein the agent is released in effective concentrations from the composition comprising the agent by erosion of the composition over a period ranging from the time of administration to about 90 days.
3804. The method of claim 3598 wherein the composition further comprises an inflammatory cytokine.
3805. The method of claim 3598 wherein the composition further comprises an agent that stimulates cell proliferation.
3806. The method of claim 3598 wherein the composition further comprises a polymeric carrier.
3807. The method of claim 3598 wherein the composition is in the form of a gel, paste, or spray.
3808. The method of claim 3598 wherein the sensor is partially constructed with the agent or the composition.
3809. The method of claim 3598 wherein the sensor is impregnated with the agent or the composition.
3810. The method of claim 3598, wherein the agent or the composition forms a coating, and the coating directly contacts the sensor.
3811. The method of claim 3598, wherein the agent or the composition forms a coating, and the coating indirectly contacts the sensor. 2005/051871
3812. The method of claim 3598 wherein the agent or the composition forms a coating, and the coating partially covers the sensor.
3813. The method of claim 3598, wherein the agent or the composition forms a coating, and the coating completely covers the sensor.
3814. The method of claim 3598 wherein the agent or the composition is located within pores or holes of the sensor.
3815. The method of claim 3598 wherein the agent or the composition is located within a channel, lumen, or divet of the sensor.
3816. The method of claim 3598 wherein the sensor further comprises an echogenic material.
3817. The method of claim 3598 wherein the sensor further comprises an echogenic material, wherein the echogenic material is in the form of a coating.
3818. The method of claim 3598 wherein the sensor is sterile.
3819. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor.
3820. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is connective tissue.
3821. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is muscle tissue. 2005/051871
3822. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is nerve tissue.
3823. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is epithelium tissue.
3824. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from the time of deployment of the sensor to about 1 year.
3825. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 month to 6 months.
3826. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 - 90 days.
3827. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a constant rate.
3828. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at an increasing rate.
3829. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a decreasing rate.
3830. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the sensor comprises about 0.01 μg to about 10 μg of the agent.
3831. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 μg to about 10 mg of the agent.
3832. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 mg to about 250 mg of the agent
3833. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the sensor comprises about 250 mg to about 1000 mg of the agent.
3834. The method of claim 3598 wherein the agent is delivered from the sensor, wherein the sensor comprises about 1000 mg to about 2500 mg of the agent.
3835. The method of claim 3598 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises less than 0.01 μg of the agent per mm2 of sensor surface to which the agent is applied.
3836. The method of claim 3598 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 0.01 μg to 2005/051871
about 1 μg of the agent per mm2 of sensor surface to which the agent is applied.
3837. The method of claim 3598 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1 μg to about 10 μg of the agent per mm2 of sensor surface to which the agent is applied.
3838. The method of claim 3598 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 10 μg to about 250 μg of the agent per mm2 of sensor surface to which the agent is applied.
3839. The method of claim 3598 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 250 μg to about 1000 μg of the agent per mm2 of sensor surface to which the agent is applied.
3840. The method of claim 3598 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1000 μg to about 2500 μg of the agent per mm2 of sensor surface to which the agent is i applied.
3841. The method of claim 3598, wherein the sensor further comprises a coating, and the coating is a uniform coating.
3842. The method of claim 3598, wherein the sensor further comprises a coating, and the coating is a non-uniform coating.
3843. The method of claim 3598, wherein the sensor further comprises a coating, and the coating is a discontinuous coating.
3844. The method of claim 3598, wherein the sensor further comprises a coating, and the coating is a patterned coating.
3845. The method of claim 3598, wherein the sensor further comprises a coating, and the coating has a thickness of 100 μm or less.
3846. The method of claim 3598, wherein the sensor further comprises a coating, and the coating has a thickness of 10 μm or less.
3847. The method of claim 3598, wherein the sensor further comprises a coating, and the coating adheres to the surface of the sensor upon deployment of the sensor.
3848. The method of claim 3598, wherein the sensor further comprises a coating, and the coating is stable at room temperature for a period of at least 1 year.
3849. The method of claim 3598, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 0.0001 % to about 1 % by weight.
3850. The method of claim 3598, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
3851. The method of claim 3598, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
3852. The method of claim 3598, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 25% to about 70% by weight. 2005/051871
3853. The method of claim 3598, wherein the sensor further comprises a coating, and the coating comprises a polymer.
3854. The method of claim 3598, wherein the sensor comprises a first coating having a first composition and a second coating having a second composition.
3855. The method of claim 3598, wherein the sensor comprises a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
3856. The method of claim 3598 wherein the agent or the composition is affixed to the sensor.
3857. The method of claim 3598 wherein the agent or the composition is covalently attached to the sensor.
3858. The method of claim 3598 wherein the agent or the composition is non-covalently attached to the sensor.
3859. The method of claim 3598 wherein the sensor comprises a coating that absorbs the agent or the composition.
3860. The method of claim 3598 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
3861. The method of claim 3598 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
3862. The method of claim 3598 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
3863. The method of claim 3598 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
3864. The method of claim 3598 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
3865. The method of claim 3598 wherein the sensor is linked to a pump.
3866. The method of claim 3598 wherein the agent or the composition is applied to the sensor surface prior to to the placing of the sensor into the host.
3867. The method of claim 3598 wherein the agent or the composition is applied to the sensor surface during the placing of the sensor into the host.
3868. The method of claim 3598 wherein the agent or the composition is applied to the sensor surface immediately after the placing of the sensor into the host.
3869. The method of claim 3598 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor prior to to the placing of the sensor into the host.
3870. The method of claim 3598 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor during the placing of the sensor into the host.
3871. The method of claim 3598 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor immediately after the placing of the sensor into the host
3872. The method of claim 3598 wherein the agent or the composition is topically applied into the anatomical space where the sensor is placed.
3873. The method of claim 3598 wherein the agent or the composition is percutaneously injected into the tissue in the host surrounding the sensor.
3874. The method of any one of claims 3598-3873 wherein the sensor is a blood or tissue glucose monitor.
3875. The method of any one of claims 3598-3873 wherein the sensor is an electrolyte sensor.
3876. The method of any one of claims 3598-3873 wherein the sensor is a blood constituent sensor.
3877. The method of any one of claims 3598-3873 wherein the sensor is a temperature sensor.
3878. The method of any one of claims 3598-3873 wherein the sensor is a pH sensor.
3879. The method of any one of claims 3598-3873 wherein the sensor is an optical sensor.
3880. The method of any one of claims 3598-3873 wherein the sensor is an amperometric sensor. 2005/051871
3881. The method of any one of claims 3598-3873 wherein the sensor is a pressure sensor.
3882. The method of any one of claims 3598-3873 wherein the sensor is a biosensor.
3883. The method of any one of claims 3598-3873 wherein the sensor is a sensing transponder.
3884. The method of any one of claims 3598-3873 wherein the sensor is a strain sensor.
3885. The method of any one of claims 3598-3873 wherein the sensor is a magnetoresistive sensor.
3886. The method of any one of claims 3598-3873 wherein the sensor is a cardiac sensor.
3887. The method of any one of claims 3598-3873 wherein the sensor is a respiratory sensor.
3888. The method of any one of claims 3598-3873 wherein the sensor is an auditory sensor.
3889. The method of any one of claims 3598-3873 wherein the sensor is a metabolite sensor.
3890. The method of any one of claims 3598-3873 wherein the sensor detects mechanical changes.
3891. The method of any one of claims 3598-3873 wherein the sensor detects physical changes.
3892. The method of any one of claims 3598-3873 wherein the sensor detects electrochemical changes.
3893. The method of any one of claims 3598-3873 wherein the sensor detects magnetic changes.
3894. The method of any one of claims 3598-3873 wherein the sensor detects acceleration changes.
3895. The method of any one of claims 3598-3873 wherein the sensor detects ionizing radiation changes.
3896. The method of any one of claims 3598-3873 wherein the sensor detects acoustic wave changes.
3897. The method of any one of claims 3598-3873 wherein the sensor detects chemical changes.
3898. The method of any one of claims 3598-3873 wherein the sensor detects drug concentration changes.
3899. The method of any one of claims 3598-3873 wherein the sensor detects hormone changes.
3900. The method of any one of claims 3598-3873 wherein the sensor detects barometric changes.
3901. A method for inhibiting scarring comprising placing a blood or tissue glucose monitor (i.e., a sensor) and an anti-scarring agent or a composition comprising an anti-scarring agent into an animal host, wherein the agent inhibits scarring.
3902. The method of claim 3901 wherein the agent inhibits cell regeneration.
3903. The method of claim 3901 wherein the agent inhibits angiogenesis.
3904. The method of claim 3901 wherein the agent inhibits fibroblast migration.
3905. The method of claim 3901 wherein the agent inhibits fibroblast proliferation.
3906. The method of claim 3901 wherein the agent inhibits deposition of extracellular matrix.
3907. The method of claim 3901 wherein the agent inhibits tissue remodeling.
3908. The method of claim 3901 wherein the agent is an angiogenesis inhibitor.
3909. The method of claim 3901 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
3910. The method of claim 3901 wherein the agent is a chemokine receptor antagonist.
3911. The method of claim 3901 wherein the agent is a cell cycle inhibitor.
3912. The method of claim 3901 wherein the agent is a taxane.
3913. The method of claim 3901 wherein the agent is an antimicrotubule agent.
3914. The method of claim 3901 wherein the agent is paclitaxel.
3915. The method of claim 3901 wherein the agent is not paclitaxel.
3916. The method of claim 3901 wherein the agent is an analogue or derivative of paclitaxel.
3917. The method of claim 3901 wherein the agent is a vinca alkaloid.
3918. The method of claim 3901 wherein the agent is camptothecin or an analogue or derivative thereof.
3919. The method of claim 3901 wherein the agent is a podophyllotoxin.
3920. The method of claim 3901 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
3921. The method of claim 3901 wherein the agent is an anthracycline.
3922. The method of claim 3901 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
3923. The method of claim 3901 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
3924. The method of claim 3901 wherein the agent is a platinum compound.
3925. The method of claim 3901 wherein the agent is a nitrosourea.
3926. The method of claim 3901 wherein the agent is a nitroimidazole.
3927. The method of claim 3901 wherein the agent is a folic acid antagonist.
3928. The method of claim 3901 wherein the agent is a cytidine analogue.
3929. The method of claim 3901 wherein the agent is a pyrimidine analogue.
3930. The method of claim 3901 wherein the agent is a fluoropyrimidine analogue.
3931. The method of claim 3901 wherein the agent is a purine analogue.
3932. The method of claim 3901 wherein the agent is a nitrogen mustard or an analogue or derivative thereof. 2005/051871
3933. The method of claim 3901 wherein the agent is a hydroxyurea.
3934. The method of claim 3901 wherein the agent is a mytomicin or an analogue or derivative thereof.
3935. The method of claim 3901 wherein the agent is an alkyl sulfonate.
3936. The method of claim 3901 wherein the agent is a benzamide or an analogue or derivative thereof.
3937. The method of claim 3901 wherein the agent is a nicotinamide or an analogue or derivative thereof.
3938. The method of claim 3901 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
3939. The method of claim 3901 wherein the agent is a DNA alkylating agent.
3940. The method of claim 3901 wherein the agent is an antimicrotubule agent.
3941. The method of claim 3901 wherein the agent is a topoisomerase inhibitor.
3942. The method of claim 3901 wherein the agent is a DNA cleaving agent.
3943. The method of claim 3901 wherein the agent is an antimetabolite. 2005/051871
3944. The method of claim 3901 wherein the agent inhibits adenosine deaminase.
3945. The method of claim 3901 wherein the agent inhibits purine ring synthesis.
3946. The method of claim 3901 wherein the agent is a nucleotide interconversion inhibitor.
3947. The method of claim 3901 wherein the agent inhibits dihydrofolate reduction.
3948. The method of claim 3901 wherein the agent blocks thymidine monophosphate.
3949. The method of claim 3901 wherein the agent causes DNA damage.
3950. The method of claim 3901 wherein the agent is a DNA intercalation agent.
3951. The method of claim 3901 wherein the agent is a RNA synthesis inhibitor.
3952. The method of claim 3901 wherein the agent is a pyrimidine synthesis inhibitor.
3953. The method of claim 3901 wherein the agent inhibits ribonucleotide synthesis or function.
3954. The method of claim 3901 wherein the agent inhibits thymidine monophosphate synthesis or function. 2005/051871
3955. The method of claim 3901 wherein the agent inhibits DNA synthesis.
3956. The method of claim 3901 wherein the agent causes DNA adduct formation.
3957. The method of claim 3901 wherein the agent inhibits protein synthesis.
3958. The method of claim 3901 wherein the agent inhibits microtubule function.
3959. The method of claim 3901 wherein the agent is a cyclin dependent protein kinase inhibitor.
3960. The method of claim 3901 wherein the agent is an epidermal growth factor kinase inhibitor.
3961. The method of claim 3901 wherein the agent is an elastase inhibitor.
3962. The method of claim 3901 wherein the agent is a factor Xa inhibitor.
3963. The method of claim 3901 wherein the agent is a farnesyltransferase inhibitor.
3964. The method of claim 3901 wherein the agent is a fibrinogen antagonist.
3965. The method of claim 3901 wherein the agent is a guanylate cyclase stimulant.
3966. The method of claim 3901 wherein the agent is a heat shock protein 90 antagonist.
3967. The method of claim 3901 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
3968. The method of claim 3901 wherein the agent is a guanylate cyclase stimulant
3969. The method of claim 3901 wherein the agent is a HMGCoA reductase inhibitor.
3970. The method of claim 3901 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
3971. The method of claim 3901 wherein the agent is a hydroorotate dehydrogenase inhibitor.
3972. The method of claim 3901 wherein the agent is an IKK2 inhibitor.
3973. The method of claim 3901 wherein the agent is an IL-1 antagonist.
3974. The method of claim 3901 wherein the agent is an ICE antagonist.
3975. The method of claim 3901 wherein the agent is an IRAK antagonist.
3976. The method of claim 3901 wherein the agent is an IL-4 agonist.
3977. The method of claim 3901 wherein the agent is an immunomodulatory agent.
3978. The method of claim 3901 wherein the agent is sirolimus or an analogue or derivative thereof.
3979. The method of claim 3901 wherein the agent is not sirolimus.
3980. The method of claim 3901 wherein the agent is everolimus or an analogue or derivative thereof.
3981. The method of claim 3901 wherein the agent is tacrolimus or an analogue or derivative thereof.
3982. The method of claim 3901 wherein the agent is not tacrolimus.
3983. The method of claim 3901 wherein the agent is biolmus or an analogue or derivative thereof.
3984. The method of claim 3901 wherein the agent is tresperimus or an analogue or derivative thereof.
3985. The method of claim 3901 wherein the agent is auranofin or an analogue or derivative thereof.
3986. The method of claim 3901 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof. 2005/051871
.
3987. The method of claim 3901 wherein the agent is gusperimus or an analogue or derivative thereof.
3988. The method of claim 3901 wherein the agent is pimecrolimus or an analogue or derivative thereof.
3989. The method of claim 3901 wherein the agent is ABT-578 or an analogue or derivative thereof.
3990. The method of claim 3901 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
3991. The method of claim 3901 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
3992. The method of claim 3901 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
3993. The method of claim 3901 wherein the agent is a leukotriene inhibitor.
3994. The method of claim 3901 wherein the agent is a MCP-1 antagonist.
3995. The method of claim 3901 wherein the agent is a MMP inhibitor.
3996. The method of claim 3901 wherein the agent is an NF kappa B inhibitor.
3997. The method of claim 3901 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
3998. The method of claim 3901 wherein the agent is an NO antagonist.
3999. The method of claim 3901 wherein the agent is a p38 MAP kinase inhibitor.
4000. The method of claim 3901 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
4001. The method of claim 3901 wherein the agent is a phosphodiesterase inhibitor.
4002. The method of claim 3901 wherein the agent is a TGF beta inhibitor.
4003. The method of claim 3901 wherein the agent is a thromboxane A2 antagonist.
4004. The method of claim 3901 wherein the agent is a TNF alpha antagonist.
4005. The method of claim 3901 wherein the agent is a TACE inhibitor.
4006. The method of claim 3901 wherein the agent is a tyrosine kinase inhibitor.
4007. The method of claim 3901 wherein the agent is a vitronectin inhibitor.
4008. The method of claim 3901 wherein the agent is a fibroblast growth factor inhibitor.
4009. The method of claim 3901 wherein the agent is a protein kinase inhibitor.
4010. The method of claim 3901 wherein the agent is a PDGF receptor kinase inhibitor.
4011. The method of claim 3901 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
4012. The method of claim 3901 wherein the agent is a retinoic acid receptor antagonist.
4013. The method of claim 3901 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
4014. The method of claim 3901 wherein the agent is a fibrinogen antagonist
4015. The method of claim 3901 wherein the agent is an antimycotic agent.
4016. The method of claim 3901 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
4017. The method of claim 3901 wherein the agent is a bisphosphonate.
4018. The method of claim 3901 wherein the agent is a phospholipase A1 inhibitor.
4019. The method of claim 3901 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
4020. The method of claim 3901 wherein the agent is a macrolide antibiotic.
4021. The method of claim 3901 wherein the agent is a GPIIb/llla receptor antagonist.
4022. The method of claim 3901 wherein the agent is an endothelin receptor antagonist.
4023. The method of claim 3901 wherein the agent is a peroxisome proliferator-activated receptor agonist.
4024. The method of claim 3901 wherein the agent is an estrogen receptor agent.
4025. The method of claim 3901 wherein the agent is a somastostatin analogue.
4026. The method of claim 3901 wherein the agent is a neurokinin 1 antagonist.
4027. The method of claim 3901 wherein the agent is a neurokinin 3 antagonist.
4028. The method of claim 3901 wherein the agent is a VLA-4 antagonist.
4029. The method of claim 3901 wherein the agent is an osteoclast inhibitor.
4030. The method of claim 3901 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
4031. The method of claim 3901 wherein the agent is an angiotensin I converting enzyme inhibitor.
4032. The method of claim 3901 wherein the agent is an angiotensin II antagonist.
4033. The method of claim 3901 wherein the agent is an enkephalinase inhibitor.
4034. The method of claim 3901 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
4035. The method of claim 3901 wherein the agent is a protein kinase C inhibitor.
4036. The method of claim 3901 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
4037. The method of claim 3901 wherein the agent is a CXCR3 inhibitor.
4038. The method of claim 3901 wherein the agent is an Itk inhibitor.
4039. The method of claim 3901 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
4040. The method of claim 3901 wherein the agent is a PPAR agonist.
4041. The method of claim 3901 wherein the agent is an immunosuppressant
4042. The method of claim 3901 wherein the agent is an Erb inhibitor.
4043. The method of claim 3901 wherein the agent is an apoptosis agonist
4044. The method of claim 3901 wherein the agent is a lipocortin agonist.
4045. The method of claim 3901 wherein the agent is a VCAM-1 antagonist.
4046. The method of claim 3901 wherein the agent is a collagen antagonist.
4047. The method of claim 3901 wherein the agent is an alpha 2 integrin antagonist.
4048. The method of claim 3901 wherein the agent is a TNF alpha inhibitor.
4049. The method of claim 3901 wherein the agent is a nitric oxide inhibitor
4050. The method of claim 3901 wherein the agent is a cathepsin inhibitor.
4051. The method of claim 3901 wherein the agent is not an anti- inflammatory agent.
4052. The method of claim 3901 wherein the agent is not a steroid.
4053. The method of claim 3901 wherein the agent is not a glucocorticosteroid.
4054. The method of claim 3901 wherein the agent is not dexamethasone.
4055. The method of claim 3901 wherein the agent is not beclomethasone.
4056. The method of claim 3901 wherein the agent is not dipropionate.
4057. The method of claim 3901 wherein the agent is not an anti- infective agent.
4058. The method of claim 3901 wherein the agent is not an antibiotic.
4059. The method of claim 3901 wherein the agent is not an anti- fungal agent.
4060. The method of claim 3901 , wherein the composition comprises a polymer.
4061. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a copolymer.
4062. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a block copolymer.
4063. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a random copolymer.
4064. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a biodegradable polymer.
4065. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a non-biodegradable polymer.
4066. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophilic polymer.
4067. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophobic polymer.
4068. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophilic domains.
4069. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophobic domains.
4070. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a non-conductive polymer.
4071. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, an elastomer.
4072. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrogel.
4073. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a silicone polymer.
4074. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrocarbon polymer.
4075. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a styrene-derived polymer.
4076. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a butadiene-derived polymer.
4077. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a macromer.
4078. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, a poly(ethylene glycol) polymer.
4079. The method of claim 3901 , wherein the composition comprises a polymer, and the polymer is, or comprises, an amorphous polymer.
4080. The method of claim 3901, wherein the composition further comprises a second pharmaceutically active agent.
4081. The method of claim 3901 , wherein the composition further comprises an anti-inflammatory agent.
4082. The method of claim 3901 , wherein the composition further comprises an agent that inhibits infection.
4083. The method of claim 3901 , wherein the composition further comprises an anthracycline.
4084. The method of claim 3901 , wherein the composition further comprises doxorubicin.
4085. The method of claim 3901 wherein the composition further comprises mitoxantrone.
4086. The method of claim 3901 wherein the composition further comprises a fluoropyrimidine.
4087. The method of claim 3901 , wherein the composition further comprises 5-fluorouracil (5-FU).
4088. The method of claim 3901 , wherein the composition further comprises a folic acid antagonist.
4089. The method of claim 3901 , wherein the composition further comprises methotrexate.
4090. The method of claim 3901 , wherein the composition further comprises a podophylotoxin.
4091. The method of claim 3901 , wherein the composition further comprises etoposide.
4092. The method of claim 3901 , wherein the composition further comprises camptothecin.
4093. The method of claim 3901 , wherein the composition further comprises a hydroxyurea.
4094. The method of claim 3901 , wherein the composition further comprises a platinum complex.
4095. The method of claim 3901 , wherein the composition further comprises cisplatin.
4096. The method of claim 3901 wherein the composition further comprises an anti-thrombotic agent.
4097. The method of claim 3901 , wherein the composition further comprises a visualization agent.
4098. The method of claim 3901 , wherein the composition further comprises a visualization agent, and the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
4099. The method of claim 3901 , wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, barium, tantalum, ortechnetium.
4100. The method of claim 3901 , wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, an MRI responsive material. 2005/051871
4101. The method of claim 3901 , wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a gadolinium chelate.
4102. The method of claim 3901 , wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron, magnesium, manganese, copper, or chromium.
4103. The method of claim 3901 , wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron oxide compound.
4104. The method of claim 3901 , wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a dye, pigment or colorant.
4105. The method of claim 3901 wherein the agent is released in effective concentrations from the composition comprising the agent by diffusion over a period ranging from the time of administration to about 90 days.
4106. The method of claim 3901 wherein the agent is released in effective concentrations from the composition comprising the agent by erosion of the composition over a period ranging from the time of administration to about 90 days.
4107. The method of claim 3901 wherein the composition further comprises an inflammatory cytokine.
4108. The method of claim 3901 wherein the composition further comprises an agent that stimulates cell proliferation.
4109. The method of claim 3901 wherein the composition further comprises a polymeric carrier.
4110. The method of claim 3901 wherein the composition is in the form of a gel, paste, or spray.
4111. The method of claim 3901 wherein the sensor is partially constructed with the agent or the composition.
4112. The method of claim 3901 wherein the sensor is impregnated with the agent or the composition.
4113. The method of claim 3901 , wherein the agent or the composition forms a coating, and the coating directly contacts the sensor.
4114. The method of claim 3901 , wherein the agent or the composition forms a coating, and the coating indirectly contacts the sensor.
4115. The method of claim 3901 wherein the agent or the composition forms a coating, and the coating partially covers the sensor.
4116. The method of claim 3901 , wherein the agent or the composition forms a coating, and the coating completely covers the sensor.
4117. The method of claim 3901 wherein the agent or the composition is located within pores or holes of the sensor.
4118. The method of claim 3901 wherein the agent or the composition is located within a channel, lumen, or divet of the sensor.
4119. The method of claim 3901 wherein the sensor further comprises an echogenic material.
4120. The method of claim 3901 wherein the sensor further comprises an echogenic material, wherein the echogenic material is in the form of a coating.
4121. The method of claim 3901 wherein the sensor is sterile.
4122. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor.
4123. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is connective tissue.
4124. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is muscle tissue.
4125. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is nerve tissue.
4126. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is epithelium tissue.
4127. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from the time of deployment of the sensor to about 1 year.
4128. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 month to 6 months.
4129. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 - 90 days.
4130. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a constant rate.
4131. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at an increasing rate.
4132. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a decreasing rate.
4133. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the sensor comprises about 0.01 μg to about 10 μg of the agent.
4134. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 μg to about 10 mg of the agent.
4135. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 mg to about 250 mg of the agent.
4136. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the sensor comprises about 250 mg to about 1000 mg of the agent
4137. The method of claim 3901 wherein the agent is delivered from the sensor, wherein the sensor comprises about 1000 mg to about 2500 mg of the agent.
4138. The method of claim 3901 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises less than 0.01 μg of the agent per mm2 of sensor surface to which the agent is applied.
4139. The method of claim 3901 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 0.01 μg to about 1 μg of the agent per mm2 of sensor surface to which the agent is applied.
4140. The method of claim 3901 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1 μg to about 10 μg of the agent per mm2 of sensor surface to which the agent is applied.
4141. The method of claim 3901 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 10 μg to about 250 μg of the agent per mm2 of sensor surface to which the agent is applied.
4142. The method of claim 3901 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 250 μg to about 1000 μg of the agent per mm2 of sensor surface to which the agent is applied. 2005/051871
4143. The method of claim 3901 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1000 μg to about 2500 μg of the agent per mm2 of sensor surface to which the agent is applied.
4144. The method of claim 3901 , wherein the sensor further comprises a coating, and the coating is a uniform coating.
4145. The method of claim 3901 , wherein the sensor further comprises a coating, and the coating is a non-uniform coating.
4146. The method of claim 3901 , wherein the sensor further comprises a coating, and the coating is a discontinuous coating.
4147. The method of claim 3901, wherein the sensor further comprises a coating, and the coating is a patterned coating.
4148. The method of claim 3901, wherein the sensor further comprises a coating, and the coating has a thickness of 100 μm or less.
4149. The method of claim 3901 , wherein the sensor further comprises a coating, and the coating has a thickness of 10 μm or less.
4150. The method of claim 3901 , wherein the sensor further comprises a coating, and the coating adheres to the surface of the sensor upon deployment of the sensor.
4151. The method of claim 3901 , wherein the sensor further comprises a coating, and the coating is stable at room temperature for a period of at least 1 year.
4152. The method of claim 3901 , wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 0.0001% to about 1 % by weight
4153. The method of claim 3901 , wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
4154. The method of claim 3901 , wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
4155. The method of claim 3901 , wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
4156. The method of claim 3901 , wherein the sensor further comprises a coating, and the coating comprises a polymer.
4157. The method of claim 3901 , wherein the sensor comprises a first coating having a first composition and a second coating having a second composition.
4158. The method of claim 3901 , wherein the sensor comprises a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
4159. The method of claim 3901 wherein the agent or the composition is affixed to the sensor.
4160. The method of claim 3901 wherein the agent or the composition is covalently attached to the sensor.
4161. The method of claim 3901 wherein the agent or the composition is non-covalently attached to the sensor.
4162. The method of claim 3901 wherein the sensor comprises a coating that absorbs the agent or the composition.
4163. The method of claim 3901 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
4164. The method of claim 3901 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
4165. The method of claim 3901 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
4166. The method of claim 3901 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
4167. The method of claim 3901 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
4168. The method of claim 3901 wherein the sensor is linked to a pump.
4169. The method of claim 3901 wherein the agent or the composition is applied to the sensor surface prior to to the placing of the sensor into the host. 2005/051871
4170. The method of claim 3901 wherein the agent or the composition is applied to the sensor surface during the placing of the sensor into the host.
4171. The method of claim 3901 wherein the agent or the composition is applied to the sensor surface immediately after the placing of the sensor into the host.
4172. The method of claim 3901 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor prior to to the placing of the sensor into the host.
4173. The method of claim 3901 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor during the placing of the sensor into the host.
4174. The method of claim 3901 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor immediately after the placing of the sensor into the host.
4175. The method of claim 3901 wherein the agent or the composition is topically applied into the anatomical space where the sensor is placed.
4176. The method of claim 3901 wherein the agent or the composition is percutaneously injected into the tissue in the host surrounding the sensor.
4177. The method of any one of claims 3901 -4176 wherein the sensor is deliverable to the vascular system transluminally using a catheter on a stent platform. 2005/051871
4178. The method of any one of claims 3901-4176 wherein the sensor is composed of glucose sensitive living cells that monitor blood glucose levels and produce a detectable electrical or optical signal in response to changes in glucose concentrations.
4179. The method of any one of claims 3901-4176 wherein the sensor is an electrode composed of an analyte responsive enzyme.
4180. The method of any one of claims 3901-4176 wherein the sensor is a closed loop insulin delivery system that comprises a sensing means that detects the host's blood glucose level and stimulates an insulin pump to supply insulin.
4181. The method of any one of claims 3901-4176 wherein the sensor is a closed loop insulin delivery system that comprises a sensing means that detects the host's blood glucose level and stimulates the pancreas to supply insulin.
4182. A method for inhibiting scarring comprising placing a pressure or stress sensor and an anti-scarring agent or a composition comprising an anti-scarring agent into an animal host, wherein the agent inhibits scarring.
4183. The method of claim 4182 wherein the agent inhibits cell regeneration.
4184. The method of claim 4182 wherein the agent inhibits angiogenesis.
4185. The method of claim 4182 wherein the agent inhibits fibroblast migration.
4186. The method of claim 4182 wherein the agent inhibits fibroblast proliferation.
4187. The method of claim 4182 wherein the agent inhibits deposition of extracellular matrix.
4188. The method of claim 4182 wherein the agent inhibits tissue remodeling.
4189. The method of claim 4182 wherein the agent is an angiogenesis inhibitor.
4190. The method of claim 4182 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
4191. The method of claim 4182 wherein the agent is a chemokine receptor antagonist.
4192. The method of claim 4182 wherein the agent is a cell cycle inhibitor.
4193. The method of claim 4182 wherein the agent is a taxane.
4194. The method of claim 4182 wherein the agent is an antimicrotubule agent.
4195. The method of claim 4182 wherein the agent is paclitaxel.
4196. The method of claim 4182 wherein the agent is not paclitaxel.
4197. The method of claim 4182 wherein the agent is an analogue or derivative of paclitaxel.
4198. The method of claim 4182 wherein the agent is a vinca alkaloid.
4199. The method of claim 4182 wherein the agent is camptothecin or an analogue or derivative thereof.
4200. The method of claim 4182 wherein the agent is a podophyllotoxin.
4201. The method of claim 4182 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
4202. The method of claim 4182 wherein the agent is an anthracycline.
4203. The method of claim 4182 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
4204. The method of claim 4182 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
4205. The method of claim 4182 wherein the agent is a platinum compound.
4206. The method of claim 4182 wherein the agent is a nitrosourea.
4207. The method of claim 4182 wherein the agent is a nitroimidazole.
4208. The method of claim 4182 wherein the agent is a folic acid antagonist.
4209. The method of claim 4182 wherein the agent is a cytidine analogue.
4210. The method of claim 4182 wherein the agent is a pyrimidine analogue.
4211. The method of claim 4182 wherein the agent is a fluoropyrimidine analogue.
4212. The method of claim 4182 wherein the agent is a purine analogue.
4213. The method of claim 4182 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
4214. The method of claim 4182 wherein the agent is a hydroxyurea.
4215. The method of claim 4182 wherein the agent is a mytomicin or an analogue or derivative thereof.
4216. The method of claim 4182 wherein the agent is an alkyl sulfonate.
4217. The method of claim 4182 wherein the agent is a benzamide or an analogue or derivative thereof.
4218. The method of claim 4182 wherein the agent is a nicotinamide or an analogue or derivative thereof.
4219. The method of claim 4182 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
4220. The method of claim 4182 wherein the agent is a DNA alkylating agent.
4221. The method of claim 4182 wherein the agent is an antimicrotubule agent.
4222. The method of claim 4182 wherein the agent is a topoisomerase inhibitor.
4223. The method of claim 4182 wherein the agent is a DNA cleaving agent.
4224. The method of claim 4182 wherein the agent is an antimetabolite.
4225. The method of claim 4182 wherein the agent inhibits adenosine deaminase.
4226. The method of claim 4182 wherein the agent inhibits purine ring synthesis.
4227. The method of claim 4182 wherein the agent is a nucleotide interconversion inhibitor.
4228. The method of claim 4182 wherein the agent inhibits dihydrofolate reduction.
4229. The method of claim 4182 wherein the agent blocks thymidine monophosphate.
4230. The method of claim 4182 wherein the agent causes DNA damage.
4231. The method of claim 4182 wherein the agent is a DNA intercalation agent.
4232. The method of claim 4182 wherein the agent is a RNA synthesis inhibitor.
4233. The method of claim 4182 wherein the agent is a pyrimidine synthesis inhibitor.
4234. The method of claim 4182 wherein the agent inhibits ribonucleotide synthesis or function.
4235. The method of claim 4182 wherein the agent inhibits thymidine monophosphate synthesis or function.
4236. The method of claim 4182 wherein the agent inhibits DNA synthesis.
4237. The method of claim 4182 wherein the agent causes DNA adduct formation.
4238. The method of claim 4182 wherein the agent inhibits protein synthesis.
4239. The method of claim 4182 wherein the agent inhibits microtubule function.
4240. The method of claim 4182 wherein the agent is a cyclin dependent protein kinase inhibitor.
4241. The method of claim 4182 wherein the agent is an epidermal growth factor kinase inhibitor.
4242. The method of claim 4182 wherein the agent is an elastase inhibitor.
4243. The method of claim 4182 wherein the agent is a factor Xa inhibitor.
4244. The method of claim 4182 wherein the agent is a farnesyltransferase inhibitor.
4245. The method of claim 4182 wherein the agent is a fibrinogen antagonist
4246. The method of claim 4182 wherein the agent is a guanylate cyclase stimulant.
4247. The method of claim 4182 wherein the agent is a heat shock protein 90 antagonist.
4248. The method of claim 4182 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
4249. The method of claim 4182 wherein the agent is a guanylate cyclase stimulant.
4250. The method of claim 4182 wherein the agent is a HMGCoA reductase inhibitor.
4251. The method of claim 4182 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
4252. The method of claim 4182 wherein the agent is a hydroorotate dehydrogenase inhibitor.
4253. The method of claim 4182 wherein the agent is an IKK2 inhibitor.
4254. The method of claim 4182 wherein the agent is an IL-1 antagonist.
4255. The method of claim 4182 wherein the agent is an ICE antagonist.
4256. The method of claim 4182 wherein the agent is an IRAK antagonist.
4257. The method of claim 4182 wherein the agent is an IL-4 agonist.
4258. The method of claim 4182 wherein the agent is an immunomodulatory agent.
4259. The method of claim 4182 wherein the agent is sirolimus or an analogue or derivative thereof.
4260. The method of claim 4182 wherein the agent is not sirolimus.
4261. The method of claim 4182 wherein the agent is everolimus or an analogue or derivative thereof.
4262. The method of claim 4182 wherein the agent is tacrolimus or an analogue or derivative thereof.
4263. The method of claim 4182 wherein the agent is not tacrolimus.
4264. The method of claim 4182 wherein the agent is biolmus or an analogue or derivative thereof.
4265. The method of claim 4182 wherein the agent is tresperimus or an analogue or derivative thereof.
4266. The method of claim 4182 wherein the agent is auranofin or an analogue or derivative thereof.
4267. The method of claim 4182 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
4268. The method of claim 4182 wherein the agent is gusperimus or an analogue or derivative thereof.
4269. The method of claim 4182 wherein the agent is pimecrolimus or an analogue or derivative thereof.
4270. The method of claim 4182 wherein the agent is ABT-578 or an analogue or derivative thereof.
4271. The method of claim 4182 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
4272. The method of claim 4182 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
4273. The method of claim 4182 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
4274. The method of claim 4182 wherein the agent is a leukotriene inhibitor.
4275. The method of claim 4182 wherein the agent is a MCP-1 antagonist.
4276. The method of claim 4182 wherein the agent is a MMP inhibitor.
4277. The method of claim 4182 wherein the agent is an NF kappa B inhibitor.
4278. The method of claim 4182 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
4279. The method of claim 4182 wherein the agent is an NO antagonist.
4280. The method of claim 4182 wherein the agent is a p38 MAP kinase inhibitor.
4281. The method of claim 4182 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
4282. The method of claim 4182 wherein the agent is a phosphodiesterase inhibitor.
4283. The method of claim 4182 wherein the agent is a TGF beta inhibitor.
4284. The method of claim 4182 wherein the agent is a thromboxane A2 antagonist.
4285. The method of claim 4182 wherein the agent is a TNF alpha antagonist
4286. The method of claim 4182 wherein the agent is a TACE inhibitor.
4287. The method of claim 4182 wherein the agent is a tyrosine kinase inhibitor.
4288. The method of claim 4182 wherein the agent is a vitronectin inhibitor.
4289. The method of claim 4182 wherein the agent is a fibroblast growth factor inhibitor.
4290. The method of claim 4182 wherein the agent is a protein kinase inhibitor.
4291. The method of claim 4182 wherein the agent is a PDGF receptor kinase inhibitor.
4292. The method of claim 4182 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
4293. The method of claim 4182 wherein the agent is a retinoic acid receptor antagonist.
4294. The method of claim 4182 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
4295. The method of claim 4182 wherein the agent is a fibrinogen antagonist.
4296. The method of claim 4182 wherein the agent is an antimycotic agent.
4297. The method of claim 4182 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
4298. The method of claim 4182 wherein the agent is a bisphosphonate.
4299. The method of claim 4182 wherein the agent is a phospholipase A1 inhibitor.
4300. The method of claim 4182 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
4301. The method of claim 4182 wherein the agent is a macrolide antibiotic.
4302. The method of claim 4182 wherein the agent is a GPIIb/llla receptor antagonist.
4303. The method of claim 4182 wherein the agent is an endothelin receptor antagonist
4304. The method of claim 4182 wherein the agent is a peroxisome proliferator-activated receptor agonist.
4305. The method of claim 4182 wherein the agent is an estrogen receptor agent.
4306. The method of claim 4182 wherein the agent is a somastostatin analogue.
4307. The method of claim 4182 wherein the agent is a neurokinin 1 antagonist.
4308. The method of claim 4182 wherein the agent is a neurokinin 3 antagonist.
4309. The method of claim 4182 wherein the agent is a VLA-4 antagonist.
4310. The method of claim 4182 wherein the agent is an osteoclast inhibitor.
4311. The method of claim 4182 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
4312. The method of claim 4182 wherein the agent is an angiotensin I converting enzyme inhibitor.
4313. The method of claim 4182 wherein the agent is an angiotensin II antagonist.
4314. The method of claim 4182 wherein the agent is an enkephalinase inhibitor.
4315. The method of claim 4182 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
4316. The method of claim 4182 wherein the agent is a protein kinase C inhibitor.
4317. The method of claim 4182 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
4318. The method of claim 4182 wherein the agent is a CXCR3 inhibitor.
4319. The method of claim 4182 wherein the agent is an Itk inhibitor.
4320. The method of claim 4182 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
4321. The method of claim 4182 wherein the agent is a PPAR agonist.
4322. The method of claim 4182 wherein the agent is an immunosuppressant.
4323. The method of claim 4182 wherein the agent is an Erb inhibitor.
4324. The method of claim 4182 wherein the agent is an apoptosis agonist.
4325. The method of claim 4182 wherein the agent is a lipocortin agonist.
4326. The method of claim 4182 wherein the agent is a VCAM-1 antagonist.
4327. The method of claim 4182 wherein the agent is a collagen antagonist.
4328. The method of claim 4182 wherein the agent is an alpha 2 integrin antagonist.
4329. The method of claim 4182 wherein the agent is a TNF alpha inhibitor.
4330. The method of claim 4182 wherein the agent is a nitric oxide inhibitor
4331. The method of claim 4182 wherein the agent is a cathepsin inhibitor.
4332. The method of claim 4182 wherein the agent is not an anti- inflammatory agent.
4333. The method of claim 4182 wherein the agent is not a steroid.
4334. The method of claim 4182 wherein the agent is not a glucocorticosteroid.
4335. The method of claim 4182 wherein the agent is not dexamethasone.
4336. The method of claim 4182 wherein the agent is not beclomethasone.
4337. The method of claim 4182 wherein the agent is not dipropionate.
4338. The method of claim 4182 wherein the agent is not an anti- infective agent.
4339. The method of claim 4182 wherein the agent is not an antibiotic.
4340. The method of claim 4182 wherein the agent is not an anti- fungal agent.
4341. The method of claim 4182, wherein the composition comprises a polymer.
4342. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a copolymer.
4343. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a block copolymer.
4344. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a random copolymer.
4345. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a biodegradable polymer.
4346. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-biodegradable polymer.
4347. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophilic polymer.
4348. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophobic polymer.
4349. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophilic domains.
4350. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophobic domains.
4351. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-conductive polymer.
4352. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, an elastomer.
4353. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrogel.
4354. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a silicone polymer.
4355. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrocarbon polymer.
4356. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a styrene-derived polymer.
4357. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a butadiene-derived polymer.
4358. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a macromer.
4359. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, a poly(ethylene glycol) polymer.
4360. The method of claim 4182, wherein the composition comprises a polymer, and the polymer is, or comprises, an amorphous polymer.
4361. The method of claim 4182, wherein the composition further comprises a second pharmaceutically active agent.
4362. The method of claim 4182, wherein the composition further comprises an anti-inflammatory agent.
4363. The method of claim 4182, wherein the composition further comprises an agent that inhibits infection.
4364. The method of claim 4182, wherein the composition further comprises an anthracycline.
4365. The method of claim 4182, wherein the composition further comprises doxorubicin.
4366. The method of claim 4182 wherein the composition further comprises mitoxantrone.
4367. The method of claim 4182 wherein the composition further comprises a fluoropyrimidine.
4368. The method of claim 4182, wherein the composition further comprises 5-fluorouracil (5-FU).
4369. The method of claim 4182, wherein the composition further comprises a folic acid antagonist.
4370. The method of claim 4182, wherein the composition further comprises methotrexate.
4371. The method of claim 4182, wherein the composition further comprises a podophylotoxin.
4372. The method of claim 4182, wherein the composition further comprises etoposide.
4373. The method of claim 4182, wherein the composition further comprises camptothecin.
4374. The method of claim 4182, wherein the composition further comprises a hydroxyurea.
4375. The method of claim 4182, wherein the composition further comprises a platinum complex.
4376. The method of claim 4182, wherein the composition further comprises cisplatin.
4377. The method of claim 4182 wherein the composition further comprises an anti-thrombotic agent.
4378. The method of claim 4182, wherein the composition further comprises a visualization agent.
4379. The method of claim 4182, wherein the composition further comprises a visualization agent, and the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
4380. The method of claim 4182, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, barium, tantalum, or technetium.
4381. The method of claim 4182, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, an MRI responsive material.
4382. The method of claim 4182, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a gadolinium chelate.
4383. The method of claim 4182, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron, magnesium, manganese, copper, or chromium.
4384. The method of claim 4182, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron oxide compound.
4385. The method of claim 4182, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a dye, pigment, or colorant.
4386. The method of claim 4182 wherein the agent is released in effective concentrations from the composition comprising the agent by diffusion over a period ranging from the time of administration to about 90 days.
4387. The method of claim 4182 wherein the agent is released in effective concentrations from the composition comprising the agent by erosion of the composition over a period ranging from the time of administration to about 90 days.
4388. The method of claim 4182 wherein the composition further comprises an inflammatory cytokine.
4389. The method of claim 4182 wherein the composition further comprises an agent that stimulates cell proliferation.
4390. The method of claim 4182 wherein the composition further comprises a polymeric carrier.
4391. The method of claim 4182 wherein the composition is in the form of a gel, paste, or spray.
4392. The method of claim 4182 wherein the sensor is partially constructed with the agent or the composition.
4393. The method of claim 4182 wherein the sensor is impregnated with the agent or the composition.
4394. The method of claim 4182, wherein the agent or the composition forms a coating, and the coating directly contacts the sensor.
4395. The method of claim 4182, wherein the agent or the composition forms a coating, and the coating indirectly contacts the sensor.
4396. The method of claim 4182 wherein the agent or the composition forms a coating, and the coating partially covers the sensor.
4397. The method of claim 4182, wherein the agent or the composition forms a coating, and the coating completely covers the sensor.
4398. The method of claim 4182 wherein the agent or the composition is located within pores or holes of the sensor.
4399. The method of claim 4182 wherein the agent or the composition is located within a channel, lumen, or divet of the sensor.
4400. The method of claim 4182 wherein the sensor further comprises an echogenic material.
4401. The method of claim 4182 wherein the sensor further comprises an echogenic material, wherein the echogenic material is in the form of a coating.
4402. The method of claim 4182 wherein the sensor is sterile.
4403. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor.
4404. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is connective tissue.
4405. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is muscle tissue.
4406. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is nerve tissue.
4407. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is epithelium tissue.
4408. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from the time of deployment of the sensor to about 1 year.
4409. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 month to 6 months.
4410. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 - 90 days.
4411. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a constant rate.
4412. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at an increasing rate.
4413. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a decreasing rate.
4414. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the sensor comprises about 0.01 μg to about 10 μg of the agent.
4415. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 μg to about 10 mg of the agent.
4416. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 mg to about 250 mg of the agent
4417. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the sensor comprises about 250 mg to about 1000 mg of the agent.
4418. The method of claim 4182 wherein the agent is delivered from the sensor, wherein the sensor comprises about 1000 mg to about 2500 mg of the agent.
4419. The method of claim 4182 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises less than 0.01 μg of the agent per mm2 of sensor surface to which the agent is applied.
4420. The method of claim 4182 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 0.01 μg to about 1 μg of the agent per mm2 of sensor surface to which the agent is applied.
4421. The method of claim 4182 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1 μg to about 10 μg of the agent per mm2 of sensor surface to which the agent is applied.
4422. The method of claim 4182 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 10 μg to about 250 μg of the agent per mm2 of sensor surface to which the agent is applied.
4423. The method of claim 4182 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 250 μg to about 1000 μg of the agent per mm2 of sensor surface to which the agent is applied.
4424. The method of claim 4182 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1000 μg to about 2500 μg of the agent per mm2 of sensor surface to which the agent is applied.
4425. The method of claim 4182, wherein the sensor further comprises a coating, and the coating is a uniform coating.
4426. The method of claim 4182, wherein the sensor further comprises a coating, and the coating is a non-uniform coating.
4427. The method of claim 4182, wherein the sensor further comprises a coating, and the coating is a discontinuous coating.
4428. The method of claim 4182, wherein the sensor further comprises a coating, and the coating is a patterned coating.
4429. The method of claim 4182, wherein the sensor further comprises a coating, and the coating has a thickness of 100 μm or less.
4430. The method of claim 4182, wherein the sensor further comprises a coating, and the coating has a thickness of 10 μm or less.
4431. The method of claim 4182, wherein the sensor further comprises a coating, and the coating adheres to the surface of the sensor < upon deployment of the sensor.
4432. The method of claim 4182, wherein the sensor further comprises a coating, and the coating is stable at room temperature for a period of at least 1 year.
4433. The method of claim 4182, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 0.0001 % to about 1% by weight.
4434. The method of claim 4182, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
4435. The method of claim 4182, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
4436. The method of claim 4182, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
4437. The method of claim 4182, wherein the sensor further comprises a coating, and the coating comprises a polymer.
4438. The method of claim 4182, wherein the sensor comprises a first coating having a first composition and a second coating having a second composition.
4439. The method of claim 4182, wherein the sensor comprises a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
4440. The method of claim 4182 wherein the agent or the composition is affixed to the sensor.
4441. The method of claim 4182 wherein the agent or the composition is covalently attached to the sensor.
4442. The method of claim 4182 wherein the agent or the composition is non-covalently attached to the sensor.
4443. The method of claim 4182 wherein the sensor comprises a coating that absorbs the agent or the composition.
4444. The method of claim 4182 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
4445. The method of claim 4182 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
4446. The method of claim 4182 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
4447. The method of claim 4182 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
4448. The method of claim 4182 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
4449. The method of claim 4182 wherein the sensor is linked to a pump.
4450. The method of claim 4182 wherein the agent or the composition is applied to the sensor surface prior to to the placing of the sensor into the host.
4451. The method of claim 4182 wherein the agent or the composition is applied to the sensor surface during the placing of the sensor into the host.
4452. The method of claim 4182 wherein the agent or the composition is applied to the sensor surface immediately after the placing of the sensor into the host.
4453. The method of claim 4182 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor prior to to the placing of the sensor into the host.
4454. The method of claim 4182 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor during the placing of the sensor into the host.
4455. The method of claim 4182 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor immediately after the placing of the sensor into the host.
4456. The method of claim 4182 wherein the agent or the composition is topically applied into the anatomical space where the sensor is placed.
4457. The method of claim 4182 wherein the agent or the composition is percutaneously injected into the tissue in the host surrounding the sensor.
4458. The method of any one of claims 4182-4457 wherein the sensor monitors blood pressure.
4459. The method of any one of claims 4182-4457 wherein the sensor monitors fluid flow.
4460. The method of any one of claims 4182-4457 wherein the sensor monitors pressure within an aneurysm sac.
4461. The method of any one of claims 4182-4457 wherein the sensor monitors intracranial pressure.
4462. The method of any one of claims 4182-4457 wherein the sensor monitors mechanical pressure associated with a bone fracture.
4463. The method of any one of claims 4182-4457 wherein the sensor monitors barometric pressure.
4464. The method of any one of claims 4182-4457 wherein the sensor monitors eye tremors.
4465. The method of any one of claims 4182-4457 wherein the sensor monitors the depth of a corneal implant.
4466. The method of any one of claims 4182-4457 wherein the sensor monitors intraocular pressure.
4467. The method of any one of claims 4182-4457 wherein the sensor is a passive sensor with an inductor-capacitor circuit.
4468. The method of any one of claims 4182-4457 wherein the sensor is a self-powered strain sensing system.
4469. The method of any one of claims 4182-4457 wherein the sensor comprises a lead, a sensor module, a sensor circuit and means for providing voltage.
4470. A method for inhibiting scarring comprising placing a cardiac sensor and an anti-scarring agent or a composition comprising an anti- scarring agent into an animal host, wherein the agent inhibits scarring.
4471. The method of claim 4470 wherein the agent inhibits cell regeneration.
4472. The method of claim 4470 wherein the agent inhibits angiogenesis.
4473. The method of claim 4470 wherein the agent inhibits fibroblast migration.
4474. The method of claim 4470 wherein the agent inhibits fibroblast proliferation.
4475. The method of claim 4470 wherein the agent inhibits deposition of extracellular matrix.
4476. The method of claim 4470 wherein the agent inhibits tissue remodeling.
4477. The method of claim 4470 wherein the agent is an angiogenesis inhibitor.
4478. The method of claim 4470 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
4479. The method of claim 4470 wherein the agent is a chemokine receptor antagonist.
4480. The method of claim 4470 wherein the agent is a cell cycle inhibitor.
4481. The method of claim 4470 wherein the agent is a taxane.
4482. The method of claim 4470 wherein the agent is an antimicrotubule agent.
4483. The method of claim 4470 wherein the agent is paclitaxel.
4484. The method of claim 4470 wherein the agent is not paclitaxel.
4485. The method of claim 4470 wherein the agent is an analogue or derivative of paclitaxel.
4486. The method of claim 4470 wherein the agent is a vinca alkaloid.
4487. The method of claim 4470 wherein the agent is camptothecin or an analogue or derivative thereof.
4488. The method of claim 4470 wherein the agent is a podophyllotoxin.
4489. The method of claim 4470 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
4490. The method of claim 4470 wherein the agent is an anthracycline.
4491. The method of claim 4470 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
4492. The method of claim 4470 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
4493. The method of claim 4470 wherein the agent is a platinum compound.
4494. The method of claim 4470 wherein the agent is a nitrosourea.
4495. The method of claim 4470 wherein the agent is a nitroimidazole.
4496. The method of claim 4470 wherein the agent is a folic acid antagonist.
4497. The method of claim 4470 wherein the agent is a cytidine analogue.
4498. The method of claim 4470 wherein the agent is a pyrimidine analogue.
4499. The method of claim 4470 wherein the agent is a fluoropyrimidine analogue.
4500. The method of claim 4470 wherein the agent is a purine analogue.
4501. The method of claim 4470 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
4502. The method of claim 4470 wherein the agent is a hydroxyurea.
4503. The method of claim 4470 wherein the agent is a mytomicin or an analogue or derivative thereof.
4504. The method of claim 4470 wherein the agent is an alkyl sulfonate.
4505. The method of claim 4470 wherein the agent is a benzamide or an analogue or derivative thereof.
4506. The method of claim 4470 wherein the agent is a nicotinamide or an analogue or derivative thereof.
4507. The method of claim 4470 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
4508. The method of claim 4470 wherein the agent is a DNA alkylating agent.
4509. The method of claim 4470 wherein the agent is an antimicrotubule agent.
4510. The method of claim 4470 wherein the agent is a topoisomerase inhibitor.
4511. The method of claim 4470 wherein the agent is a DNA cleaving agent.
4512. The method of claim 4470 wherein the agent is an antimetabolite.
4513. The method of claim 4470 wherein the agent inhibits adenosine deaminase.
4514. The method of claim 4470 wherein the agent inhibits purine ring synthesis.
4515. The method of claim 4470 wherein the agent is a nucleotide interconversion inhibitor.
4516. The method of claim 4470 wherein the agent inhibits dihydrofolate reduction.
4517. The method of claim 4470 wherein the agent blocks thymidine monophosphate.
4518. The method of claim 4470 wherein the agent causes DNA damage.
4519. The method of claim 4470 wherein the agent is a DNA intercalation agent.
4520. The method of claim 4470 wherein the agent is a RNA synthesis inhibitor.
4521. The method of claim 4470 wherein the agent is a pyrimidine synthesis inhibitor.
4522. The method of claim 4470 wherein the agent inhibits ribonucleotide synthesis or function.
4523. The method of claim 4470 wherein the agent inhibits thymidine monophosphate synthesis or function. 2005/051871
4524. The method of claim 4470 wherein the agent inhibits DNA synthesis.
4525. The method of claim 4470 wherein the agent causes DNA adduct formation.
4526. The method of claim 4470 wherein the agent inhibits protein synthesis.
4527. The method of claim 4470 wherein the agent inhibits microtubule function.
4528. The method of claim 4470 wherein the agent is a cyclin dependent protein kinase inhibitor.
4529. The method of claim 4470 wherein the agent is an epidermal growth factor kinase inhibitor.
4530. The method of claim 4470 wherein the agent is an elastase inhibitor.
4531. The method of claim 4470 wherein the agent is a factor Xa inhibitor.
4532. The method of claim 4470 wherein the agent is a farnesyltransferase inhibitor.
4533. The method of claim 4470 wherein the agent is a fibrinogen antagonist.
4534. The method of claim 4470 wherein the agent is a guanylate cyclase stimulant. 2005/051871
4535. The method of claim 4470 wherein the agent is a heat shock protein 90 antagonist.
4536. The method of claim 4470 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
4537. The method of claim 4470 wherein the agent is a guanylate cyclase stimulant.
4538. The method of claim 4470 wherein the agent is a HMGCoA reductase inhibitor.
4539. The method of claim 4470 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
4540. The method of claim 4470 wherein the agent is a hydroorotate dehydrogenase inhibitor.
4541. The method of claim 4470 wherein the agent is an IKK2 inhibitor.
4542. The method of claim 4470 wherein the agent is an IL-1 antagonist.
4543. The method of claim 4470 wherein the agent is an ICE antagonist.
4544. The method of claim 4470 wherein the agent is an IRAK antagonist. 2005/051871
4545. The method of claim 4470 wherein the agent is an IL-4 agonist.
4546. The method of claim 4470 wherein the agent is an immunomodulatory agent.
4547. The method of claim 4470 wherein the agent is sirolimus or an analogue or derivative thereof.
4548. The method of claim 4470 wherein the agent is not sirolimus.
4549. The method of claim 4470 wherein the agent is everolimus or an analogue or derivative thereof.
4550. The method of claim 4470 wherein the agent is tacrolimus or an analogue or derivative thereof.
4551. The method of claim 4470 wherein the agent is not tacrolimus.
4552. The method of claim 4470 wherein the agent is biolmus or an analogue or derivative thereof.
4553. The method of claim 4470 wherein the agent is tresperimus or an analogue or derivative thereof.
4554. The method of claim 4470 wherein the agent is auranofin or an analogue or derivative thereof.
4555. The method of claim 4470 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof. 2005/051871
4556. The method of claim 4470 wherein the agent is gusperimus or an analogue or derivative thereof.
4557. The method of claim 4470 wherein the agent is pimecrolimus or an analogue or derivative thereof.
4558. The method of claim 4470 wherein the agent is ABT-578 or an analogue or derivative thereof.
4559. The method of claim 4470 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
4560. The method of claim 4470 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
4561. The method of claim 4470 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
4562. The method of claim 4470 wherein the agent is a leukotriene inhibitor.
4563. The method of claim 4470 wherein the agent is a MCP-1 antagonist.
4564. The method of claim 4470 wherein the agent is a MMP inhibitor.
4565. The method of claim 4470 wherein the agent is an NF kappa B inhibitor. 2005/051871
4566. The method of claim 4470 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
4567. The method of claim 4470 wherein the agent is an NO antagonist
4568. The method of claim 4470 wherein the agent is a p38 MAP kinase inhibitor.
4569. The method of claim 4470 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
4570. The method of claim 4470 wherein the agent is a phosphodiesterase inhibitor.
4571. The method of claim 4470 wherein the agent is a TGF beta inhibitor.
4572. The method of claim 4470 wherein the agent is a thromboxane A2 antagonist.
4573. The method of claim 4470 wherein the agent is a TNF alpha antagonist.
4574. The method of claim 4470 wherein the agent is a TACE inhibitor.
4575. The method of claim 4470 wherein the agent is a tyrosine kinase inhibitor.
4576. The method of claim 4470 wherein the agent is a vitronectin inhibitor.
4577. The method of claim 4470 wherein the agent is a fibroblast growth factor inhibitor.
4578. The method of claim 4470 wherein the agent is a protein kinase inhibitor.
4579. The method of claim 4470 wherein the agent is a PDGF receptor kinase inhibitor.
4580. The method of claim 4470 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
4581. The method of claim 4470 wherein the agent is a retinoic acid receptor antagonist.
4582. The method of claim 4470 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
4583. The method of claim 4470 wherein the agent is a fibrinogen antagonist.
4584. The method of claim 4470 wherein the agent is an antimycotic agent.
4585. The method of claim 4470 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
4586. The method of claim 4470 wherein the agent is a bisphosphonate.
4587. The method of claim 4470 wherein the agent is a phospholipase A1 inhibitor.
4588. The method of claim 4470 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
4589. The method of claim 4470 wherein the agent is a macrolide antibiotic.
4590. The method of claim 4470 wherein the agent is a GPIIb/llla receptor antagonist.
4591. The method of claim 4470 wherein the agent is an endothelin receptor antagonist.
4592. The method of claim 4470 wherein the agent is a peroxisome proliferator-activated receptor agonist
4593. The method of claim 4470 wherein the agent is an estrogen receptor agent.
4594. The method of claim 4470 wherein the agent is a somastostatin analogue.
4595. The method of claim 4470 wherein the agent is a neurokinin 1 antagonist.
4596. The method of claim 4470 wherein the agent is a neurokinin 3 antagonist
4597. The method of claim 4470 wherein the agent is a VLA-4 antagonist.
4598. The method of claim 4470 wherein the agent is an osteoclast inhibitor.
4599. The method of claim 4470 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
4600. The method of claim 4470 wherein the agent is an angiotensin I converting enzyme inhibitor.
4601. The method of claim 4470 wherein the agent is an angiotensin II antagonist
4602. The method of claim 4470 wherein the agent is an enkephalinase inhibitor.
4603. The method of claim 4470 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
4604. The method of claim 4470 wherein the agent is a protein kinase C inhibitor.
4605. The method of claim 4470 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
4606. The method of claim 4470 wherein the agent is a CXCR3 inhibitor.
4607. The method of claim 4470 wherein the agent is an Itk inhibitor.
4608. The method of claim 4470 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
4609. The method of claim 4470 wherein the agent is a PPAR agonist.
4610. The method of claim 4470 wherein the agent is an immunosuppressant
4611. The method of claim 4470 wherein the agent is an Erb inhibitor.
4612. The method of claim 4470 wherein the agent is an apoptosis agonist.
4613. The method of claim 4470 wherein the agent is a lipocortin agonist.
4614. The method of claim 4470 wherein the agent is a VCAM-1 antagonist
4615. The method of claim 4470 wherein the agent is a collagen antagonist.
4616. The method of claim 4470 wherein the agent is an alpha 2 integrin antagonist
4617. The method of claim 4470 wherein the agent is a TNF alpha inhibitor.
4618. The method of claim 4470 wherein the agent is a nitric oxide inhibitor
4619. The method of claim 4470 wherein the agent is a cathepsin inhibitor.
4620. The method of claim 4470 wherein the agent is not an anti- inflammatory agent.
4621. The method of claim 4470 wherein the agent is not a steroid.
4622. The method of claim 4470 wherein the agent is not a glucocorticosteroid.
4623. The method of claim 4470 wherein the agent is not dexamethasone.
4624. The method of claim 4470 wherein the agent is not beclomethasone.
4625. The method of claim 4470 wherein the agent is not dipropionate.
4626. The method of claim 4470 wherein the agent is not an anti- infective agent.
4627. The method of claim 4470 wherein the agent is not an antibiotic.
4628. The method of claim 4470 wherein the agent is not an anti- fungal agent
4629. The method of claim 4470, wherein the composition comprises a polymer.
4630. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a copolymer.
4631. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a block copolymer.
4632. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a random copolymer.
4633. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a biodegradable polymer.
4634. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-biodegradable polymer.
4635. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophilic polymer.
4636. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophobic polymer.
4637. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophilic domains.
4638. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophobic domains.
4639. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-conductive polymer.
4640. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, an elastomer.
4641. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrogel.
4642. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a silicone polymer.
4643. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrocarbon polymer.
4644. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a styrene-derived polymer.
4645. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a butadiene-derived polymer.
4646. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a macromer.
4647. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, a poly( ethylene glycol) polymer.
4648. The method of claim 4470, wherein the composition comprises a polymer, and the polymer is, or comprises, an amorphous polymer.
4649. The method of claim 4470, wherein the composition further comprises a second pharmaceutically active agent.
4650. The method of claim 4470, wherein the composition further comprises an anti-inflammatory agent.
4651. The method of claim 4470, wherein the composition further comprises an agent that inhibits infection.
4652. The method of claim 4470, wherein the composition further comprises an anthracycline.
4653. The method of claim 4470, wherein the composition further comprises doxorubicin.
4654. The method of claim 4470 wherein the composition further comprises mitoxantrone.
4655. The method of claim 4470 wherein the composition further comprises a fluoropyrimidine.
4656. The method of claim 4470, wherein the composition further comprises 5-fluorouracil (5-FU).
4657. The method of claim 4470, wherein the composition further comprises a folic acid antagonist.
4658. The method of claim 4470, wherein the composition further comprises methotrexate.
4659. The method of claim 4470, wherein the composition further comprises a podophylotoxin.
4660. The method of claim 4470, wherein the composition further comprises etoposide.
4661. The method of claim 4470, wherein the composition further comprises camptothecin.
4662. The method of claim 4470, wherein the composition further comprises a hydroxyurea.
4663. The method of claim 4470, wherein the composition further comprises a platinum complex.
4664. The method of claim 4470, wherein the composition further comprises cisplatin.
4665. The method of claim 4470 wherein the composition further comprises an anti-thrombotic agent.
4666. The method of claim 4470, wherein the composition further comprises a visualization agent.
4667. The method of claim 4470, wherein the composition further comprises a visualization agent, and the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
4668. The method of claim 4470, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, barium, tantalum, or technetium.
4669. The method of claim 4470, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, an MRI responsive material.
4670. The method of claim 4470, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a gadolinium chelate.
4671. The method of claim 4470, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron, magnesium, manganese, copper, or chromium.
4672. The method of claim 4470, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron oxide compound.
4673. The method of claim 4470, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a dye, pigment, or colorant.
4674. The method of claim 4470 wherein the agent is released in effective concentrations from the composition comprising the agent by diffusion over a period ranging from the time of administration to about 90 days.
4675. The method of claim 4470 wherein the agent is released in effective concentrations from the composition comprising the agent by erosion of the composition over a period ranging from the time of administration to about 90 days.
4676. The method of claim 4470 wherein the composition further comprises an inflammatory cytokine.
4677. The method of claim 4470 wherein the composition further comprises an agent that stimulates cell proliferation.
4678. The method of claim 4470 wherein the composition further comprises a polymeric carrier.
4679. The method of claim 4470 wherein the composition is in the form of a gel, paste, or spray.
4680. The method of claim 4470 wherein the sensor is partially constructed with the agent or the composition.
4681. The method of claim 4470 wherein the sensor is impregnated with the agent or the composition.
4682. The method of claim 4470, wherein the agent or the composition forms a coating, and the coating directly contacts the sensor.
4683. The method of claim 4470, wherein the agent or the composition forms a coating, and the coating indirectly contacts the sensor.
4684. The method of claim 4470 wherein the agent or the composition forms a coating, and the coating partially covers the sensor.
4685. The method of claim 4470, wherein the agent or the composition forms a coating, and the coating completely covers the sensor.
4686. The method of claim 4470 wherein the agent or the composition is located within pores or holes of the sensor.
4687. The method of claim 4470 wherein the agent or the composition is located within a channel, lumen, or divet of the sensor.
4688. The method of claim 4470 wherein the sensor further comprises an echogenic material.
4689. The method of claim 4470 wherein the sensor further comprises an echogenic material, wherein the echogenic material is in the form of a coating.
4690. The method of claim 4470 wherein the sensor is sterile.
4691. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor.
4692. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is connective tissue.
4693. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is muscle tissue.
4694. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is nerve tissue.
4695. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is epithelium tissue.
4696. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from the time of deployment of the sensor to about 1 year.
4697. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 month to 6 months.
4698. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 - 90 days.
4699. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a constant rate.
4700. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at an increasing rate.
4701. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a decreasing rate.
4702. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the sensor comprises about 0.01 μg to about 10 μg of the agent.
4703. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 μg to about 10 mg of the agent.
4704. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 mg to about 250 mg of the agent.
4705. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the sensor comprises about 250 mg to about 1000 mg of the agent.
4706. The method of claim 4470 wherein the agent is delivered from the sensor, wherein the sensor comprises about 1000 mg to about 2500 mg of the agent
4707. The method of claim 4470 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises less than 0.01 μg of the agent per mm2 of sensor surface to which the agent is applied.
4708. The method of claim 4470 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 0.01 μg to about 1 μg of the agent per mm2 of sensor surface to which the agent is applied.
4709. The method of claim 4470 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1 μg to about 10 μg of the agent per mm2 of sensor surface to which the agent is applied.
4710. The method of claim 4470 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 10 μg to about 250 μg of the agent per mm2 of sensor surface to which the agent is applied.
4711. The method of claim 4470 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 250 μg to about 1000 μg of the agent per mm2 of sensor surface to which the agent is applied.
4712. The method of claim 4470 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1000 μg to about 2500 μg of the agent per mm2 of sensor surface to which the agent is applied.
4713. The method of claim 4470, wherein the sensor further comprises a coating, and the coating is a uniform coating.
4714. The method of claim 4470, wherein the sensor further comprises a coating, and the coating is a non-uniform coating.
4715. The method of claim 4470, wherein the sensor further comprises a coating, and the coating is a discontinuous coating.
4716. The method of claim 4470, wherein the sensor further comprises a coating, and the coating is a patterned coating.
4717. The method of claim 4470, wherein the sensor further comprises a coating, and the coating has a thickness of 100 μm or less.
4718. The method of claim 4470, wherein the sensor further comprises a coating, and the coating has a thickness of 10 μm or less.
4719. The method of claim 4470, wherein the sensor further comprises a coating, and the coating adheres to the surface of the sensor upon deployment of the sensor.
4720. The method of claim 4470, wherein the sensor further comprises a coating, and the coating is stable at room temperature for a period of at least 1 year.
4721. The method of claim 4470, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 0.0001% to about 1% by weight.
4722. The method of claim 4470, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 1 % to about 10% by weight
4723. The method of claim 4470, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
4724. The method of claim 4470, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
4725. The method of claim 4470, wherein the sensor further comprises a coating, and the coating comprises a polymer.
4726. The method of claim 4470, wherein the sensor comprises a first coating having a first composition and a second coating having a second composition.
4727. The method of claim 4470, wherein the sensor comprises a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
4728. The method of claim 4470 wherein the agent or the composition is affixed to the sensor.
4729. The method of claim 4470 wherein the agent or the composition is covalently attached to the sensor.
4730. The method of claim 4470 wherein the agent or the composition is non-covalently attached to the sensor.
4731. The method of claim 4470 wherein the sensor comprises a coating that absorbs the agent or the composition.
4732. The method of claim 4470 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
4733. The method of claim 4470 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
4734. The method of claim 4470 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
4735. The method of claim 4470 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
4736. The method of claim 4470 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
4737. The method of claim 4470 wherein the sensor is linked to a pump.
4738. The method of claim 4470 wherein the agent or the composition is applied to the sensor surface prior to to the placing of the sensor into the host.
4739. The method of claim 4470 wherein the agent or the composition is applied to the sensor surface during the placing of the sensor into the host
4740. The method of claim 4470 wherein the agent or the composition is applied to the sensor surface immediately after the placing of the sensor into the host.
4741. The method of claim 4470 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor prior to to the placing of the sensor into the host.
4742. The method of claim 4470 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor during the placing of the sensor into the host.
4743. The method of claim 4470 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor immediately after the placing of the sensor into the host.
4744. The method of claim 4470 wherein the agent or the composition is topically applied into the anatomical space where the sensor is placed.
4745. The method of claim 4470 wherein the agent or the composition is percutaneously injected into the tissue in the host surrounding the sensor.
4746. The method of any one of claims 4470-4745 wherein the sensor monitors cardiac output.
4747. The method of any one of claims 4470-4745 wherein the sensor monitors ejection fraction.
4748. The method of any one of claims 4470-4745 wherein the sensor monitors blood pressure in a heart chamber.
4749. The method of any one of claims 4470-4745 wherein the sensor monitors ventricular wall motions.
4750. The method of any one of claims 4470-4745 wherein the sensor monitors blood flow to a transplanted organ.
4751. The method of any one of claims 4470-4745 wherein the sensor monitors heart rate.
4752. A method for inhibiting scarring comprising placing a respiratory sensor and an anti-scarring agent or a composition comprising an anti-scarring agent into an animal host, wherein the agent inhibits scarring.
4753. The method of claim 4752 wherein the agent inhibits cell regeneration.
4754. The method of claim 4752 wherein the agent inhibits angiogenesis.
4755. The method of claim 4752 wherein the agent inhibits fibroblast migration.
4756. The method of claim 4752 wherein the agent inhibits fibroblast proliferation.
4757. The method of claim 4752 wherein the agent inhibits deposition of extracellular matrix.
4758. The method of claim 4752 wherein the agent inhibits tissue remodeling.
4759. The method of claim 4752 wherein the agent is an angiogenesis inhibitor.
4760. The method of claim 4752 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
4761. The method of claim 4752 wherein the agent is a chemokine receptor antagonist.
4762. The method of claim 4752 wherein the agent is a cell cycle inhibitor.
4763. The method of claim 4752 wherein the agent is a taxane.
4764. The method of claim 4752 wherein the agent is an antimicrotubule agent.
4765. The method of claim 4752 wherein the agent is paclitaxel.
4766. The method of claim 4752 wherein the agent is not paclitaxel.
4767. The method of claim 4752 wherein the agent is an analogue or derivative of paclitaxel.
4768. The method of claim 4752 wherein the agent is a vinca alkaloid.
4769. The method of claim 4752 wherein the agent is camptothecin or an analogue or derivative thereof.
4770. The method of claim 4752 wherein the agent is a podophyllotoxin.
4771. The method of claim 4752 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
4772. The method of claim 4752 wherein the agent is an anthracycline.
4773. The method of claim 4752 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
4774. The method of claim 4752 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
4775. The method of claim 4752 wherein the agent is a platinum compound.
4776. The method of claim 4752 wherein the agent is a nitrosourea.
4777. The method of claim 4752 wherein the agent is a nitroimidazole.
4778. The method of claim 4752 wherein the agent is a folic acid antagonist.
4779. The method of claim 4752 wherein the agent is a cytidine analogue.
4780. The method of claim 4752 wherein the agent is a pyrimidine analogue.
4781. The method of claim 4752 wherein the agent is a fluoropyrimidine analogue.
4782. The method of claim 4752 wherein the agent is a purine analogue.
4783. The method of claim 4752 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
4784. The method of claim 4752 wherein the agent is a hydroxyurea.
4785. The method of claim 4752 wherein the agent is a mytomicin or an analogue or derivative thereof.
4786. The method of claim 4752 wherein the agent is an alkyl sulfonate.
4787. The method of claim 4752 wherein the agent is a benzamide or an analogue or derivative thereof.
4788. The method of claim 4752 wherein the agent is a nicotinamide or an analogue or derivative thereof.
4789. The method of claim 4752 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
4790. The method of claim 4752 wherein the agent is a DNA alkylating agent.
4791. The method of claim 4752 wherein the agent is an antimicrotubule agent.
4792. The method of claim 4752 wherein the agent is a topoisomerase inhibitor.
4793. The method of claim 4752 wherein the agent is a DNA cleaving agent.
4794. The method of claim 4752 wherein the agent is an antimetabolite.
4795. The method of claim 4752 wherein the agent inhibits adenosine deaminase.
4796. The method of claim 4752 wherein the agent inhibits purine ring synthesis.
4797. The method of claim 4752 wherein the agent is a nucleotide interconversion inhibitor.
4798. The method of claim 4752 wherein the agent inhibits dihydrofolate reduction.
4799. The method of claim 4752 wherein the agent blocks thymidine monophosphate.
4800. The method of claim 4752 wherein the agent causes DNA damage.
4801. The method of claim 4752 wherein the agent is a DNA intercalation agent
4802. The method of claim 4752 wherein the agent is a RNA synthesis inhibitor.
4803. The method of claim 4752 wherein the agent is a pyrimidine synthesis inhibitor.
4804. The method of claim 4752 wherein the agent inhibits ribonucleotide synthesis or function.
4805. The method of claim 4752 wherein the agent inhibits thymidine monophosphate synthesis or function.
4806. The method of claim 4752 wherein the agent inhibits DNA synthesis.
4807. The method of claim 4752 wherein the agent causes DNA adduct formation.
4808. The method of claim 4752 wherein the agent inhibits protein synthesis.
4809. The method of claim 4752 wherein the agent inhibits microtubule function.
4810. The method of claim 4752 wherein the agent is a cyclin dependent protein kinase inhibitor.
4811. The method of claim 4752 wherein the agent is an epidermal growth factor kinase inhibitor.
4812. The method of claim 4752 wherein the agent is an elastase inhibitor.
4813. The method of claim 4752 wherein the agent is a factor Xa inhibitor.
4814. The method of claim 4752 wherein the agent is a farnesyltransferase inhibitor.
4815. The method of claim 4752 wherein the agent is a fibrinogen antagonist.
4816. The method of claim 4752 wherein the agent is a guanylate cyclase stimulant.
4817. The method of claim 4752 wherein the agent is a heat shock protein 90 antagonist.
4818. The method of claim 4752 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
4819. The method of claim 4752 wherein the agent is a guanylate cyclase stimulant.
4820. The method of claim 4752 wherein the agent is a HMGCoA reductase inhibitor.
4821. The method of claim 4752 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
4822. The method of claim 4752 wherein the agent is a hydroorotate dehydrogenase inhibitor.
4823. The method of claim 4752 wherein the agent is an IKK2 inhibitor.
4824. The method of claim 4752 wherein the agent is an IL-1 antagonist.
4825. The method of claim 4752 wherein the agent is an ICE antagonist.
4826. The method of claim 4752 wherein the agent is an IRAK antagonist.
4827. The method of claim 4752 wherein the agent is an IL-4 agonist.
4828. The method of claim 4752 wherein the agent is an immunomodulatory agent.
4829. The method of claim 4752 wherein the agent is sirolimus or an analogue or derivative thereof.
4830. The method of claim 4752 wherein the agent is not sirolimus.
4831. The method of claim 4752 wherein the agent is everolimus or an analogue or derivative thereof.
4832. The method of claim 4752 wherein the agent is tacrolimus or an analogue or derivative thereof.
4833. The method of claim 4752 wherein the agent is not tacrolimus.
4834. The method of claim 4752 wherein the agent is biolmus or an analogue or derivative thereof.
4835. The method of claim 4752 wherein the agent is tresperimus or an analogue or derivative thereof.
4836. The method of claim 4752 wherein the agent is auranofin or an analogue or derivative thereof.
4837. The method of claim 4752 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
4838. The method of claim 4752 wherein the agent is gusperimus or an analogue or derivative thereof.
4839. The method of claim 4752 wherein the agent is pimecrolimus or an analogue or derivative thereof.
4840. The method of claim 4752 wherein the agent is ABT-578 or an analogue or derivative thereof.
4841. The method of claim 4752 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
4842. The method of claim 4752 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
4843. The method of claim 4752 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
4844. The method of claim 4752 wherein the agent is a leukotriene inhibitor.
4845. The method of claim 4752 wherein the agent is a MCP-1 antagonist
4846. The method of claim 4752 wherein the agent is a MMP inhibitor.
4847. The method of claim 4752 wherein the agent is an NF kappa B inhibitor.
4848. The method of claim 4752 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
4849. The method of claim 4752 wherein the agent is an NO antagonist.
4850. The method of claim 4752 wherein the agent is a p38 MAP kinase inhibitor.
4851. The method of claim 4752 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
4852. The method of claim 4752 wherein the agent is a phosphodiesterase inhibitor.
4853. The method of claim 4752 wherein the agent is a TGF beta inhibitor.
4854. The method of claim 4752 wherein the agent is a thromboxane A2 antagonist.
4855. The method of claim 4752 wherein the agent is a TNF alpha antagonist.
4856. The method of claim 4752 wherein the agent is a TACE inhibitor.
4857. The method of claim 4752 wherein the agent is a tyrosine kinase inhibitor.
4858. The method of claim 4752 wherein the agent is a vitronectin inhibitor.
4859. The method of claim 4752 wherein the agent is a fibroblast growth factor inhibitor.
4860. The method of claim 4752 wherein the agent is a protein kinase inhibitor.
4861. The method of claim 4752 wherein the agent is a PDGF receptor kinase inhibitor.
4862. The method of claim 4752 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
4863. The method of claim 4752 wherein the agent is a retinoic acid receptor antagonist.
4864. The method of claim 4752 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
4865. The method of claim 4752 wherein the agent is a fibrinogen antagonist.
4866. The method of claim 4752 wherein the agent is an antimycotic agent
4867. The method of claim 4752 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
4868. The method of claim 4752 wherein the agent is a bisphosphonate.
4869. The method of claim 4752 wherein the agent is a phospholipase A1 inhibitor.
4870. The method of claim 4752 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
4871. The method of claim 4752 wherein the agent is a macrolide antibiotic.
4872. The method of claim 4752 wherein the agent is a GPIIb/llla receptor antagonist.
4873. The method of claim 4752 wherein the agent is an endothelin receptor antagonist.
4874. The method of claim 4752 wherein the agent is a peroxisome proliferator-activated receptor agonist.
4875. The method of claim 4752 wherein the agent is an estrogen receptor agent.
4876. The method of claim 4752 wherein the agent is a somastostatin analogue.
4877. The method of claim 4752 wherein the agent is a neurokinin 1 antagonist.
4878. The method of claim 4752 wherein the agent is a neurokinin 3 antagonist.
4879. The method of claim 4752 wherein the agent is a VLA-4 antagonist.
4880. The method of claim 4752 wherein the agent is an osteoclast inhibitor.
4881. The method of claim 4752 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
4882. The method of claim 4752 wherein the agent is an angiotensin I converting enzyme inhibitor.
4883. The method of claim 4752 wherein the agent is an angiotensin II antagonist.
4884. The method of claim 4752 wherein the agent is an enkephalinase inhibitor.
4885. The method of claim 4752 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
4886. The method of claim 4752 wherein the agent is a protein kinase C inhibitor.
4887. The method of claim 4752 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
4888. The method of claim 4752 wherein the agent is a CXCR3 inhibitor,
4889. The method of claim 4752 wherein the agent is an Itk inhibitor.
4890. The method of claim 4752 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
4891. The method of claim 4752 wherein the agent is a PPAR agonist.
4892. The method of claim 4752 wherein the agent is an immunosuppressant.
4893. The method of claim 4752 wherein the agent is an Erb inhibitor.
4894. The method of claim 4752 wherein the agent is an apoptosis agonist.
4895. The method of claim 4752 wherein the agent is a lipocortin agonist.
4896. The method of claim 4752 wherein the agent is a VCAM-1 antagonist.
4897. The method of claim 4752 wherein the agent is a collagen antagonist.
4898. The method of claim 4752 wherein the agent is an alpha 2 integrin antagonist.
4899. The method of claim 4752 wherein the agent is a TNF alpha inhibitor.
4900. The method of claim 4752 wherein the agent is a nitric oxide inhibitor
4901. The method of claim 4752 wherein the agent is a cathepsin inhibitor.
4902. The method of claim 4752 wherein the agent is not an anti- inflammatory agent
4903. The method of claim 4752 wherein the agent is not a steroid.
4904. The method of claim 4752 wherein the agent is not a glucocorticosteroid.
4905. The method of claim 4752 wherein the agent is not dexamethasone.
4906. The method of claim 4752 wherein the agent is not beclomethasone.
4907. The method of claim 4752 wherein the agent is not dipropionate.
4908. The method of claim 4752 wherein the agent is not an anti- infective agent.
4909. The method of claim 4752 wherein the agent is not an antibiotic.
4910. The method of claim 4752 wherein the agent is not an anti- fungal agent.
4911. The method of claim 4752, wherein the composition comprises a polymer.
4912. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a copolymer.
4913. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a block copolymer.
4914. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a random copolymer.
4915. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a biodegradable polymer.
4916. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-biodegradable polymer.
4917. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophilic polymer.
4918. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophobic polymer.
4919. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophilic domains.
4920. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophobic domains.
4921. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-conductive polymer.
4922. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, an elastomer.
4923. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrogel.
4924. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a silicone polymer.
4925. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrocarbon polymer.
4926. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a styrene-derived polymer.
4927. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a butadiene-derived polymer.
4928. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a macromer.
4929. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, a poly( ethylene glycol) polymer.
4930. The method of claim 4752, wherein the composition comprises a polymer, and the polymer is, or comprises, an amorphous polymer.
4931. The method of claim 4752, wherein the composition further comprises a second pharmaceutically active agent.
4932. The method of claim 4752, wherein the composition further comprises an anti-inflammatory agent.
4933. The method of claim 4752, wherein the composition further comprises an agent that inhibits infection.
4934. The method of claim 4752, wherein the composition further comprises an anthracycline.
4935. The method of claim 4752, wherein the composition further comprises doxorubicin.
4936. The method of claim 4752 wherein the composition further comprises mitoxantrone.
4937. The method of claim 4752 wherein the composition further comprises a fluoropyrimidine.
4938. The method of claim 4752, wherein the composition further comprises 5-fluorouracil (5-FU).
4939. The method of claim 4752, wherein the composition further comprises a folic acid antagonist.
4940. The method of claim 4752, wherein the composition further comprises methotrexate.
4941. The method of claim 4752, wherein the composition further comprises a podophylotoxin.
4942. The method of claim 4752, wherein the composition further comprises etoposide.
4943. The method of claim 4752, wherein the composition further comprises camptothecin.
4944. The method of claim 4752, wherein the composition further comprises a hydroxyurea.
4945. The method of claim 4752, wherein the composition further comprises a platinum complex.
4946. The method of claim 4752, wherein the composition further comprises cisplatin.
4947. The method of claim 4752 wherein the composition further comprises an anti-thrombotic agent.
4948. The method of claim 4752, wherein the composition further comprises a visualization agent.
4949. The method of claim 4752, wherein the composition further comprises a visualization agent, and the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
4950. The method of claim 4752, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, barium, tantalum, or technetium.
4951. The method of claim 4752, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, an MRI responsive material.
4952. The method of claim 4752, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a gadolinium chelate.
4953. The method of claim 4752, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron, magnesium, manganese, copper, or chromium.
4954. The method of claim 4752, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron oxide compound.
4955. The method of claim 4752, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a dye, pigment, or colorant
4956. The method of claim 4752 wherein the agent is released in effective concentrations from the composition comprising the agent by diffusion over a period ranging from the time of administration to about 90 days.
4957. The method of claim 4752 wherein the agent is released in effective concentrations from the composition comprising the agent by erosion of the composition over a period ranging from the time of administration to about 90 days.
4958. The method of claim 4752 wherein the composition further comprises an inflammatory cytokine.
4959. The method of claim 4752 wherein the composition further comprises an agent that stimulates cell proliferation.
4960. The method of claim 4752 wherein the composition further comprises a polymeric carrier.
4961. The method of claim 4752 wherein the composition is in the form of a gel, paste, or spray.
4962. The method of claim 4752 wherein the sensor is partially constructed with the agent or the composition.
4963. The method of claim 4752 wherein the sensor is impregnated with the agent or the composition.
4964. The method of claim 4752, wherein the agent or the composition forms a coating, and the coating directly contacts the sensor.
4965. The method of claim 4752, wherein the agent or the composition forms a coating, and the coating indirectly contacts the sensor.
4966. The method of claim 4752 wherein the agent or the composition forms a coating, and the coating partially covers the sensor.
4967. The method of claim 4752, wherein the agent or the composition forms a coating, and the coating completely covers the sensor.
4968. The method of claim 4752 wherein the agent or the composition is located within pores or holes of the sensor.
4969. The method of claim 4752 wherein the agent or the composition is located within a channel, lumen, or divet of the sensor.
4970. The method of claim 4752 wherein the sensor further comprises an echogenic material.
4971. The method of claim 4752 wherein the sensor further comprises an echogenic material, wherein the echogenic material is in the form of a coating.
4972. The method of claim 4752 wherein the sensor is sterile.
4973. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor.
4974. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is connective tissue.
4975. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is muscle tissue.
4976. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is nerve tissue.
4977. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is epithelium tissue.
4978. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from the time of deployment of the sensor to about 1 year.
4979. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 month to 6 months.
4980. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 - 90 days.
4981. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a constant rate.
4982. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at an increasing rate.
4983. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a decreasing rate.
4984. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the sensor comprises about 0.01 μg to about 10 μg of the agent.
4985. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 μg to about 10 mg of the agent.
4986. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 mg to about 250 mg of the agent.
4987. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the sensor comprises about 250 mg to about 1000 mg of the agent.
4988. The method of claim 4752 wherein the agent is delivered from the sensor, wherein the sensor comprises about 1000 mg to about 2500 mg of the agent.
4989. The method of claim 4752 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises less than 0.01 μg of the agent per mm2 of sensor surface to which the agent is applied.
4990. The method of claim 4752 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 0.01 μg to about 1 μg of the agent per mm2 of sensor surface to which the agent is applied.
4991. The method of claim 4752 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1 μg to about 10 μg of the agent per mm2 of sensor surface to which the agent is applied.
4992. The method of claim 4752 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 10 μg to about 250 μg of the agent per mm2 of sensor surface to which the agent is applied.
4993. The method of claim 4752 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 250 μg to about 1000 μg of the agent per mm2 of sensor surface to which the agent is applied.
4994. The method of claim 4752 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1000 μg to about 2500 μg of the agent per mm2 of sensor surface to which the agent is applied.
4995. The method of claim 4752, wherein the sensor further comprises a coating, and the coating is a uniform coating.
4996. The method of claim 4752, wherein the sensor further comprises a coating, and the coating is a non-uniform coating.
4997. The method of claim 4752, wherein the sensor further comprises a coating, and the coating is a discontinuous coating.
4998. The method of claim 4752, wherein the sensor further comprises a coating, and the coating is a patterned coating.
4999. The method of claim 4752, wherein the sensor further comprises a coating, and the coating has a thickness of 100 μm or less.
5000. The method of claim 4752, wherein the sensor further comprises a coating, and the coating has a thickness of 10 μm or less.
5001. The method of claim 4752, wherein the sensor further comprises a coating, and the coating adheres to the surface of the sensor upon deployment of the sensor.
5002. The method of claim 4752, wherein the sensor further comprises a coating, and the coating is stable at room temperature for a period of at least 1 year.
5003. The method of claim 4752, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 0.0001 % to about 1 % by weight.
5004. The method of claim 4752, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
5005. The method of claim 4752, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
5006. The method of claim 4752, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
5007. The method of claim 4752, wherein the sensor further comprises a coating, and the coating comprises a polymer.
5008. The method of claim 4752, wherein the sensor comprises a first coating having a first composition and a second coating having a second composition.
5009. The method of claim 4752, wherein the sensor comprises a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
5010. The method of claim 4752 wherein the agent or the composition is affixed to the sensor.
5011. The method of claim 4752 wherein the agent or the composition is covalently attached to the sensor.
5012. The method of claim 4752 wherein the agent or the composition is non-covalently attached to the sensor.
5013. The method of claim 4752 wherein the sensor comprises a coating that absorbs the agent or the composition.
5014. The method of claim 4752 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
5015. The method of claim 4752 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
5016. The method of claim 4752 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
5017. The method of claim 4752 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
5018. The method of claim 4752 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
5019. The method of claim 4752 wherein the sensor is linked to a pump.
5020. The method of claim 4752 wherein the agent or the composition is applied to the sensor surface prior to to the placing of the sensor into the host.
5021. The method of claim 4752 wherein the agent or the composition is applied to the sensor surface during the placing of the sensor into the host.
5022. The method of claim 4752 wherein the agent or the composition is applied to the sensor surface immediately after the placing of the sensor into the host.
5023. The method of claim 4752 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor prior to to the placing of the sensor into the host.
5024. The method of claim 4752 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor during the placing of the sensor into the host.
5025. The method of claim 4752 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor immediately after the placing of the sensor into the host.
5026. The method of claim 4752 wherein the agent or the composition is topically applied into the anatomical space where the sensor is placed.
5027. The method of claim 4752 wherein the agent or the composition is percutaneously injected into the tissue in the host surrounding the sensor.
5028. The method of claims 4752-5027 wherein the sensor monitors pulmonary functions.
5029. A method for inhibiting scarring comprising placing an auditory sensor and an anti-scarring agent or a composition comprising an anti-scarring agent into an animal host, wherein the agent inhibits scarring.
5030. The method of claim 5029 wherein the agent inhibits cell regeneration.
5031. The method of claim 5029 wherein the agent inhibits angiogenesis.
5032. The method of claim 5029 wherein the agent inhibits fibroblast migration.
5033. The method of claim 5029 wherein the agent inhibits fibroblast proliferation.
5034. The method of claim 5029 wherein the agent inhibits deposition of extracellular matrix.
5035. The method of claim 5029 wherein the agent inhibits tissue remodeling.
5036. The method of claim 5029 wherein the agent is an angiogenesis inhibitor.
5037. The method of claim 5029 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
5038. The method of claim 5029 wherein the agent is a chemokine receptor antagonist.
5039. The method of claim 5029 wherein the agent is a cell cycle inhibitor.
5040. The method of claim 5029 wherein the agent is a taxane. ' iC ' ' •■ '' 'U1 ^ ιuι """" "*' "Ά' ' "!,i ":r il ,π' ,v'
5041. The method of claim 5029 wherein the agent is an antimicrotubule agent.
5042. The method of claim 5029 wherein the agent is paclitaxel.
5043. The method of claim 5029 wherein the agent is not paclitaxel.
5044. The method of claim 5029 wherein the agent is an analogue or derivative of paclitaxel.
5045. The method of claim 5029 wherein the agent is a vinca alkaloid.
5046. The method of claim 5029 wherein the agent is camptothecin or an analogue or derivative thereof.
5047. The method of claim 5029 wherein the agent is a podophyllotoxin.
5048. The method of claim 5029 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
5049. The method of claim 5029 wherein the agent is an anthracycline.
5050. The method of claim 5029 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof. : T . U b; ϋ i!<* -* ""iii «,»iι ,uι ■''
5051. The method of claim 5029 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
5052. The method of claim 5029 wherein the agent is a platinum compound.
5053. The method of claim 5029 wherein the agent is a nitrosourea.
5054. The method of claim 5029 wherein the agent is a nitroimidazole.
5055. The method of claim 5029 wherein the agent is a folic acid antagonist.
5056. The method of claim 5029 wherein the agent is a cytidine analogue.
5057. The method of claim 5029 wherein the agent is a pyrimidine analogue.
5058. The method of claim 5029 wherein the agent is a fluoropyrimidine analogue.
5059. The method of claim 5029 wherein the agent is a purine analogue.
5060. The method of claim 5029 wherein the agent is a nitrogen mustard or an analogue or derivative thereof. : { r ■ yι \ \j >' 'ii" .■■ .T -.;;» *•• •» »-» ••■
5061. The method of claim 5029 wherein the agent is a hydroxyurea.
5062. The method of claim 5029 wherein the agent is a mytomicin or an analogue or derivative thereof.
5063. The method of claim 5029 wherein the agent is an alkyl sulfonate.
5064. The method of claim 5029 wherein the agent is a benzamide or an analogue or derivative thereof.
5065. The method of claim 5029 wherein the agent is a nicotinamide or an analogue or derivative thereof.
5066. The method of claim 5029 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
5067. The method of claim 5029 wherein the agent is a DNA alkylating agent
5068. The method of claim 5029 wherein the agent is an antimicrotubule agent.
5069. The method of claim 5029 wherein the agent is a topoisomerase inhibitor.
5070. The method of claim 5029 wherein the agent is a DNA cleaving agent.
5071. The method of claim 5029 wherein the agent is an antimetabolite. C T / 8 if -Λ ii jt '''ii' ••' -3ι "ϋi1 -H 'i ,* >ι-
5072. The method of claim 5029 wherein the agent inhibits adenosine deaminase.
5073. The method of claim 5029 wherein the agent inhibits purine ring synthesis.
5074. The method of claim 5029 wherein the agent is a nucleotide interconversion inhibitor.
5075. The method of claim 5029 wherein the agent inhibits dihydrofolate reduction.
5076. The method of claim 5029 wherein the agent blocks thymidine monophosphate.
5077. The method of claim 5029 wherein the agent causes DNA damage.
5078. The method of claim 5029 wherein the agent is a DNA intercalation agent.
5079. The method of claim 5029 wherein the agent is a RNA synthesis inhibitor.
5080. The method of claim 5029 wherein the agent is a pyrimidine synthesis inhibitor.
5081. The method of claim 5029 wherein the agent inhibits ribonucleotide synthesis or function.
5082. The method of claim 5029 wherein the agent inhibits thymidine monophosphate synthesis or function.
5083. The method of claim 5029 wherein the agent inhibits DNA synthesis.
5084. The method of claim 5029 wherein the agent causes DNA adduct formation.
5085. The method of claim 5029 wherein the agent inhibits protein synthesis.
5086. The method of claim 5029 wherein the agent inhibits microtubule function.
5087. The method of claim 5029 wherein the agent is a cyclin dependent protein kinase inhibitor.
5088. The method of claim 5029 wherein the agent is an epidermal growth factor kinase inhibitor.
5089. The method of claim 5029 wherein the agent is an elastase inhibitor.
5090. The method of claim 5029 wherein the agent is a factor Xa inhibitor.
5091. The method of claim 5029 wherein the agent is a farnesyltransferase inhibitor.
5092. The method of claim 5029 wherein the agent is a fibrinogen antagonist.
5093. The method of claim 5029 wherein the agent is a guanylate cyclase stimulant. f C I ϋ! ''»»u lui ""ll" ■■' ""''' '■'" ""* 'wι "
5094. The method of claim 5029 wherein the agent is a heat shock protein 90 antagonist.
5095. The method of claim 5029 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
5096. The method of claim 5029 wherein the agent is a guanylate cyclase stimulant.
5097. The method of claim 5029 wherein the agent is a HMGCoA reductase inhibitor.
5098. The method of claim 5029 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
5099. The method of claim 5029 wherein the agent is a hydroorotate dehydrogenase inhibitor.
5100. The method of claim 5029 wherein the agent is an IKK2 inhibitor.
5101. The method of claim 5029 wherein the agent is an IL-1 antagonist.
5102. The method of claim 5029 wherein the agent is an ICE antagonist
5103. The method of claim 5029 wherein the agent is an IRAK antagonist.
5104. The method of claim 5029 wherein the agent is an IL-4 agonist
5105. The method of claim 5029 wherein the agent is an immunomodulatory agent.
5106. The method of claim 5029 wherein the agent is sirolimus or an analogue or derivative thereof.
5107. The method of claim 5029 wherein the agent is not sirolimus.
5108. The method of claim 5029 wherein the agent is everolimus or an analogue or derivative thereof.
5109. The method of claim 5029 wherein the agent is tacrolimus or an analogue or derivative thereof.
5110. The method of claim 5029 wherein the agent is not tacrolimus.
5111. The method of claim 5029 wherein the agent is biolmus or an analogue or derivative thereof.
5112. The method of claim 5029 wherein the agent is tresperimus or an analogue or derivative thereof.
5113. The method of claim 5029 wherein the agent is auranofin or an analogue or derivative thereof.
5114. The method of claim 5029 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof. " it X i/l ^MUl ''"if',.' „,.,H .^ . .|. -H» ■
5115. The method of claim 5029 wherein the agent is gusperimus or an analogue or derivative thereof.
5116. The method of claim 5029 wherein the agent is pimecrolimus or an analogue or derivative thereof.
5117. The method of claim 5029 wherein the agent is ABT-578 or an analogue or derivative thereof.
5118. The method of claim 5029 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
5119. The method of claim 5029 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
5120. The method of claim 5029 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
5121. The method of claim 5029 wherein the agent is a leukotriene inhibitor.
5122. The method of claim 5029 wherein the agent is a MCP-1 antagonist
5123. The method of claim 5029 wherein the agent is a MMP inhibitor.
5124. The method of claim 5029 wherein the agent is an NF kappa B inhibitor.
5125. The method of claim 5029 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
5126. The method of claim 5029 wherein the agent is an NO antagonist.
5127. The method of claim 5029 wherein the agent is a p38 MAP kinase inhibitor.
5128. The method of claim 5029 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
5129. The method of claim 5029 wherein the agent is a phosphodiesterase inhibitor.
5130. The method of claim 5029 wherein the agent is a TGF beta inhibitor.
5131. The method of claim 5029 wherein the agent is a thromboxane A2 antagonist.
5132. The method of claim 5029 wherein the agent is a TNF alpha antagonist.
5133. The method of claim 5029 wherein the agent is a TACE inhibitor.
5134. The method of claim 5029 wherein the agent is a tyrosine kinase inhibitor.
5135. The method of claim 5029 wherein the agent is a vitronectin inhibitor.
5136. The method of claim 5029 wherein the agent is a fibroblast growth factor inhibitor.
5137. The method of claim 5029 wherein the agent is a protein kinase inhibitor.
5138. The method of claim 5029 wherein the agent is a PDGF receptor kinase inhibitor.
5139. The method of claim 5029 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
5140. The method of claim 5029 wherein the agent is a retinoic acid receptor antagonist.
5141. The method of claim 5029 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
5142. The method of claim 5029 wherein the agent is a fibrinogen antagonist.
5143. The method of claim 5029 wherein the agent is an antimycotic agent.
5144. The method of claim 5029 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
5145. The method of claim 5029 wherein the agent is a bisphosphonate.
5146. The method of claim 5029 wherein the agent is a phospholipase A1 inhibitor.
5147. The method of claim 5029 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
5148. The method of claim 5029 wherein the agent is a macrolide antibiotic.
5149. The method of claim 5029 wherein the agent is a GPIIb/llla receptor antagonist
5150. The method of claim 5029 wherein the agent is an endothelin receptor antagonist.
5151. The method of claim 5029 wherein the agent is a peroxisome proliferator-activated receptor agonist.
5152. The method of claim 5029 wherein the agent is an estrogen receptor agent.
5153. The method of claim 5029 wherein the agent is a somastostatin analogue.
5154. The method of claim 5029 wherein the agent is a neurokinin 1 antagonist.
5155. The method of claim 5029 wherein the agent is a neurokinin 3 antagonist.
5156. The method of claim 5029 wherein the agent is a VLA-4 antagonist.
5157. The method of claim 5029 wherein the agent is an osteoclast inhibitor.
5158. The method of claim 5029 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
5159. The method of claim 5029 wherein the agent is an angiotensin I converting enzyme inhibitor.
5160. The method of claim 5029 wherein the agent is an angiotensin II antagonist.
5161. The method of claim 5029 wherein the agent is an enkephalinase inhibitor.
5162. The method of claim 5029 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
5163. The method of claim 5029 wherein the agent is a protein kinase C inhibitor.
5164. The method of claim 5029 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
5165. The method of claim 5029 wherein the agent is a CXCR3 inhibitor.
5166. The method of claim 5029 wherein the agent is an Itk inhibitor.
5167. The method of claim 5029 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
5168. The method of claim 5029 wherein the agent is a PPAR agonist.
5169. The method of claim 5029 wherein the agent is an immunosuppressant.
5170. The method of claim 5029 wherein the agent is an Erb inhibitor.
5171. The method of claim 5029 wherein the agent is an apoptosis agonist.
5172. The method of claim 5029 wherein the agent is a lipocortin agonist.
5173. The method of claim 5029 wherein the agent is a VCAM-1 antagonist.
5174. The method of claim 5029 wherein the agent is a collagen antagonist
5175. The method of claim 5029 wherein the agent is an alpha 2 integrin antagonist.
5176. The method of claim 5029 wherein the agent is a TNF alpha inhibitor.
5177. The method of claim 5029 wherein the agent is a nitric oxide inhibitor
5178. The method of claim 5029 wherein the agent is a cathepsin inhibitor.
5179. The method of claim 5029 wherein the agent is not an anti- inflammatory agent.
5180. The method of claim 5029 wherein the agent is not a steroid.
5181. The method of claim 5029 wherein the agent is not a glucocorticosteroid.
5182. The method of claim 5029 wherein the agent is not dexamethasone.
5183. The method of claim 5029 wherein the agent is not beclomethasone.
5184. The method of claim 5029 wherein the agent is not dipropionate.
5185. The method of claim 5029 wherein the agent is not an anti- infective agent
5186. The method of claim 5029 wherein the agent is not an antibiotic.
5187. The method of claim 5029 wherein the agent is not an anti- fungal agent.
5188. The method of claim 5029, wherein the composition comprises a polymer.
5189. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a copolymer.
5190. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a block copolymer.
5191. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a random copolymer.
5192. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a biodegradable polymer.
5193. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-biodegradable polymer.
5194. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophilic polymer.
5195. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophobic polymer.
5196. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophilic domains.
5197. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophobic domains.
5198. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-conductive polymer.
5199. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, an elastomer.
5200. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrogel.
5201. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a silicone polymer.
5202. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrocarbon polymer.
5203. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a styrene-derived polymer.
5204. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a butadiene-derived polymer.
5205. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a macromer.
5206. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, a poly(ethylene glycol) polymer.
5207. The method of claim 5029, wherein the composition comprises a polymer, and the polymer is, or comprises, an amorphous polymer.
5208. The method of claim 5029, wherein the composition further comprises a second pharmaceutically active agent.
5209. The method of claim 5029, wherein the composition further comprises an anti-inflammatory agent.
5210. The method of claim 5029, wherein the composition further comprises an agent that inhibits infection.
5211. The method of claim 5029, wherein the composition further comprises an anthracycline.
5212. The method of claim 5029, wherein the composition further comprises doxorubicin.
5213. The method of claim 5029 wherein the composition further comprises mitoxantrone.
5214. The method of claim 5029 wherein the composition further comprises a fluoropyrimidine.
5215. The method of claim 5029, wherein the composition further comprises 5-fluorouracil (5-FU).
5216. The method of claim 5029, wherein the composition further comprises a folic acid antagonist.
5217. The method of claim 5029, wherein the composition further comprises methotrexate.
5218. The method of claim 5029, wherein the composition further comprises a podophylotoxin.
5219. The method of claim 5029, wherein the composition further comprises etoposide.
5220. The method of claim 5029, wherein the composition further comprises camptothecin.
5221. The method of claim 5029, wherein the composition further comprises a hydroxyurea.
5222. The method of claim 5029, wherein the composition further comprises a platinum complex.
5223. The method of claim 5029, wherein the composition further comprises cisplatin.
5224. The method of claim 5029 wherein the composition further comprises an anti-thrombotic agent.
5225. The method of claim 5029, wherein the composition further comprises a visualization agent
5226. The method of claim 5029, wherein the composition further comprises a visualization agent, and the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
5227. The method of claim 5029, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, barium, tantalum, or technetium.
5228. The method of claim 5029, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, an MRI responsive material.
5229. The method of claim 5029, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a gadolinium chelate.
5230. The method of claim 5029, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron, magnesium, manganese, copper, or chromium.
5231. The method of claim 5029, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron oxide compound.
5232. The method of claim 5029, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a dye, pigment, or colorant.
5233. The method of claim 5029 wherein the agent is released in effective concentrations from the composition comprising the agent by diffusion over a period ranging from the time of administration to about 90 days.
5234. The method of claim 5029 wherein the agent is released in effective concentrations from the composition comprising the agent by erosion of the composition over a period ranging from the time of administration to about 90 days.
5235. The method of claim 5029 wherein the composition further comprises an inflammatory cytokine.
5236. The method of claim 5029 wherein the composition further comprises an agent that stimulates cell proliferation.
5237. The method of claim 5029 wherein the composition further comprises a polymeric carrier.
5238. The method of claim 5029 wherein the composition is in the form of a gel, paste, or spray.
5239. The method of claim 5029 wherein the sensor is partially constructed with the agent or the composition.
5240. The method of claim 5029 wherein the sensor is impregnated with the agent or the composition.
5241. The method of claim 5029, wherein the agent or the composition forms a coating, and the coating directly contacts the sensor.
5242. The method of claim 5029, wherein the agent or the composition forms a coating, and the coating indirectly contacts the sensor.
5243. The method of claim 5029 wherein the agent or the composition forms a coating, and the coating partially covers the sensor.
5244. The method of claim 5029, wherein the agent or the composition forms a coating, and the coating completely covers the sensor.
5245. The method of claim 5029 wherein the agent or the composition is located within pores or holes of the sensor.
5246. The method of claim 5029 wherein the agent or the composition is located within a channel, lumen, or divet of the sensor.
5247. The method of claim 5029 wherein the sensor further comprises an echogenic material. 2005/051871
5248. The method of claim 5029 wherein the sensor further comprises an echogenic material, wherein the echogenic material is in the form of a coating.
5249. The method of claim 5029 wherein the sensor is sterile.
5250. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor.
5251. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is connective tissue.
5252. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is muscle tissue.
5253. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is nerve tissue.
5254. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is epithelium tissue.
5255. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from the time of deployment of the sensor to about 1 year.
5256. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 month to 6 months.
5257. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 - 90 days.
5258. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a constant rate.
5259. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at an increasing rate.
5260. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a decreasing rate.
5261. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the sensor comprises about 0.01 μg to about 10 μg of the agent.
5262. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 μg to about 10 mg of the agent.
5263. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 mg to about 250 mg of the agent.
5264. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the sensor comprises about 250 mg to about 1000 mg of the agent.
5265. The method of claim 5029 wherein the agent is delivered from the sensor, wherein the sensor comprises about 1000 mg to about 2500 mg of the agent.
5266. The method of claim 5029 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises less than 0.01 μg of the agent per mm2 of sensor surface to which the agent is applied.
5267. The method of claim 5029 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 0.01 μg to about 1 μg of the agent per mm2 of sensor surface to which the agent is applied.
5268. The method of claim 5029 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1 μg to about 10 μg of the agent per mm2 of sensor surface to which the agent is applied.
5269. The method of claim 5029 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 10 μg to about 250 μg of the agent per mm2 of sensor surface to which the agent is applied.
5270. The method of claim 5029 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 250 μg to about 1000 μg of the agent per mm2 of sensor surface to which the agent is applied.
5271. The method of claim 5029 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1000 μg to about 2500 μg of the agent per mm2 of sensor surface to which the agent is applied.
5272. The method of claim 5029, wherein the sensor further comprises a coating, and the coating is a uniform coating.
5273. The method of claim 5029, wherein the sensor further comprises a coating, and the coating is a non-uniform coating.
5274. The method of claim 5029, wherein the sensor further comprises a coating, and the coating is a discontinuous coating.
5275. The method of claim 5029, wherein the sensor further comprises a coating, and the coating is a patterned coating.
5276. The method of claim 5029, wherein the sensor further comprises a coating, and the coating has a thickness of 100 μm or less.
5277. The method of claim 5029, wherein the sensor further comprises a coating, and the coating has a thickness of 10 μm or less.
5278. The method of claim 5029, wherein the sensor further comprises a coating, and the coating adheres to the surface of the sensor upon deployment of the sensor.
5279. The method of claim 5029, wherein the sensor further comprises a coating, and the coating is stable at room temperature for a period of at least 1 year.
5280. The method of claim 5029, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 0.0001 % to about 1 % by weight.
5281. The method of claim 5029, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
5282. The method of claim 5029, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
5283. The method of claim 5029, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
5284. The method of claim 5029, wherein the sensor further comprises a coating, and the coating comprises a polymer.
5285. The method of claim 5029, wherein the sensor comprises a first coating having a first composition and a second coating having a second composition.
5286. The method of claim 5029, wherein the sensor comprises a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
5287. The method of claim 5029 wherein the agent or the composition is affixed to the sensor.
5288. The method of claim 5029 wherein the agent or the composition is covalently attached to the sensor.
5289. The method of claim 5029 wherein the agent or the composition is non-covalently attached to the sensor.
5290. The method of claim 5029 wherein the sensor comprises a coating that absorbs the agent or the composition.
5291. The method of claim 5029 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
5292. The method of claim 5029 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
5293. The method of claim 5029 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
5294. The method of claim 5029 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
5295. The method of claim 5029 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
5296. The method of claim 5029 wherein the sensor is linked to a pump.
5297. The method of claim 5029 wherein the agent or the composition is applied to the sensor surface prior to to the placing of the sensor into the host.
5298. The method of claim 5029 wherein the agent or the composition is applied to the sensor surface during the placing of the sensor into the host.
5299. The method of claim 5029 wherein the agent or the composition is applied to the sensor surface immediately after the placing of the sensor into the host.
5300. The method of claim 5029 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor prior to to the placing of the sensor into the host.
5301. The method of claim 5029 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor during the placing of the sensor into the host.
5302. The method of claim 5029 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor immediately after the placing of the sensor into the host.
5303. The method of claim 5029 wherein the agent or the composition is topically applied into the anatomical space where the sensor is placed.
5304. The method of claim 5029 wherein the agent or the composition is percutaneously injected into the tissue in the host surrounding the sensor.
5305. The method of any one of claims 5029-5304 wherein the sensor is adapted for delivering an electrical signal to an implantable electromechanical transducer that acts on the middle or inner ear.
5306. The method of any one of claims 5029-5304 wherein the sensor generates an electrical audio signal.
5307. The method of any one of claims 5029-5304 wherein the sensor is a capacitive sensor that is coupled to a vibrating auditory element.
5308. The method of any one of claims 5029-5304 wherein the sensor is an electromagnetic sensor.
5309. A method for inhibiting scarring comprising placing an electrolyte or metabolite sensor and an anti-scarring agent or a composition comprising an anti-scarring agent into an animal host, wherein the agent inhibits scarring.
5310. The method of claim 5309 wherein the agent inhibits cell regeneration.
5311. The method of claim 5309 wherein the agent inhibits angiogenesis.
5312. The method of claim 5309 wherein the agent inhibits fibroblast migration.
5313. The method of claim 5309 wherein the agent inhibits fibroblast proliferation.
5314. The method of claim 5309 wherein the agent inhibits deposition of extracellular matrix.
5315. The method of claim 5309 wherein the agent inhibits tissue remodeling.
5316. The method of claim 5309 wherein the agent is an angiogenesis inhibitor.
5317. The method of claim 5309 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
5318. The method of claim 5309 wherein the agent is a chemokine receptor antagonist.
5319. The method of claim 5309 wherein the agent is a cell cycle inhibitor.
5320. The method of claim 5309 wherein the agent is a taxane.
5321. The method of claim 5309 wherein the agent is an antimicrotubule agent.
5322. The method of claim 5309 wherein the agent is paclitaxel.
5323. The method of claim 5309 wherein the agent is not paclitaxel.
5324. The method of claim 5309 wherein the agent is an analogue or derivative of paclitaxel.
5325. The method of claim 5309 wherein the agent is a vinca alkaloid.
5326. The method of claim 5309 wherein the agent is camptothecin or an analogue or derivative thereof.
5327. The method of claim 5309 wherein the agent is a podophyllotoxin.
5328. The method of claim 5309 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
5329. The method of claim 5309 wherein the agent is an anthracycline.
5330. The method of claim 5309 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
5331. The method of claim 5309 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
5332. The method of claim 5309 wherein the agent is a platinum compound.
5333. The method of claim 5309 wherein the agent is a nitrosourea.
5334. The method of claim 5309 wherein the agent is a nitroimidazole.
5335. The method of claim 5309 wherein the agent is a folic acid antagonist.
5336. The method of claim 5309 wherein the agent is a cytidine analogue.
5337. The method of claim 5309 wherein the agent is a pyrimidine analogue.
5338. The method of claim 5309 wherein the agent is a fluoropyrimidine analogue.
5339. The method of claim 5309 wherein the agent is a purine analogue.
5340. The method of claim 5309 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
5341. The method of claim 5309 wherein the agent is a hydroxyurea.
5342. The method of claim 5309 wherein the agent is a mytomicin or an analogue or derivative thereof.
.
5343. The method of claim 5309 wherein the agent is an alkyl sulfonate.
5344. The method of claim 5309 wherein the agent is a benzamide or an analogue or derivative thereof.
5345. The method of claim 5309 wherein the agent is a nicotinamide or an analogue or derivative thereof.
5346. The method of claim 5309 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
5347. The method of claim 5309 wherein the agent is a DNA alkylating agent.
5348. The method of claim 5309 wherein the agent is an antimicrotubule agent.
5349. The method of claim 5309 wherein the agent is a topoisomerase inhibitor.
5350. The method of claim 5309 wherein the agent is a DNA cleaving agent
5351. The method of claim 5309 wherein the agent is an antimetabolite.
5352. The method of claim 5309 wherein the agent inhibits adenosine deaminase.
5353. The method of claim 5309 wherein the agent inhibits purine ring synthesis.
5354. The method of claim 5309 wherein the agent is a nucleotide interconversion inhibitor.
5355. The method of claim 5309 wherein the agent inhibits dihydrofolate reduction.
5356. The method of claim 5309 wherein the agent blocks thymidine monophosphate.
5357. The method of claim 5309 wherein the agent causes DNA damage.
5358. The method of claim 5309 wherein the agent is a DNA intercalation agent.
5359. The method of claim 5309 wherein the agent is a RNA synthesis inhibitor.
5360. The method of claim 5309 wherein the agent is a pyrimidine synthesis inhibitor.
5361. The method of claim 5309 wherein the agent inhibits ribonucleotide synthesis or function.
5362. The method of claim 5309 wherein the agent inhibits thymidine monophosphate synthesis or function.
5363. The method of claim 5309 wherein the agent inhibits DNA synthesis.
5364. The method of claim 5309 wherein the agent causes DNA adduct formation.
5365. The method of claim 5309 wherein the agent inhibits protein synthesis.
5366. The method of claim 5309 wherein the agent inhibits microtubule function.
5367. The method of claim 5309 wherein the agent is a cyclin dependent protein kinase inhibitor.
5368. The method of claim 5309 wherein the agent is an epidermal growth factor kinase inhibitor.
5369. The method of claim 5309 wherein the agent is an elastase inhibitor.
5370. The method of claim 5309 wherein the agent is a factor Xa inhibitor.
5371. The method of claim 5309 wherein the agent is a farnesyltransferase inhibitor.
5372. The method of claim 5309 wherein the agent is a fibrinogen antagonist.
5373. The method of claim 5309 wherein the agent is a guanylate cyclase stimulant.
5374. The method of claim 5309 wherein the agent is a heat shock protein 90 antagonist.
5375. The method of claim 5309 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
5376. The method of claim 5309 wherein the agent is a guanylate cyclase stimulant.
5377. The method of claim 5309 wherein the agent is a HMGCoA reductase inhibitor.
5378. The method of claim 5309 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
5379. The method of claim 5309 wherein the agent is a hydroorotate dehydrogenase inhibitor.
5380. The method of claim 5309 wherein the agent is an IKK2 inhibitor.
5381. The method of claim 5309 wherein the agent is an IL-1 antagonist.
5382. The method of claim 5309 wherein the agent is an ICE antagonist.
5383. The method of claim 5309 wherein the agent is an IRAK antagonist.
5384. The method of claim 5309 wherein the agent is an IL-4 agonist.
5385. The method of claim 5309 wherein the agent is an immunomodulatory agent.
5386. The method of claim 5309 wherein the agent is sirolimus or an analogue or derivative thereof.
5387. The method of claim 5309 wherein the agent is not sirolimus.
5388. The method of claim 5309 wherein the agent is everolimus or an analogue or derivative thereof.
5389. The method of claim 5309 wherein the agent is tacrolimus or an analogue or derivative thereof.
5390. The method of claim 5309 wherein the agent is not tacrolimus.
5391. The method of claim 5309 wherein the agent is biolmus or an analogue or derivative thereof.
5392. The method of claim 5309 wherein the agent is tresperimus or an analogue or derivative thereof.
5393. The method of claim 5309 wherein the agent is auranofin or an analogue or derivative thereof.
5394. The method of claim 5309 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
5395. The method of claim 5309 wherein the agent is gusperimus or an analogue or derivative thereof.
5396. The method of claim 5309 wherein the agent is pimecrolimus or an analogue or derivative thereof.
5397. The method of claim 5309 wherein the agent is ABT-578 or an analogue or derivative thereof.
5398. The method of claim 5309 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
5399. The method of claim 5309 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
5400. The method of claim 5309 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
5401. The method of claim 5309 wherein the agent is a leukotriene inhibitor.
5402. The method of claim 5309 wherein the agent is a MCP-1 antagonist
5403. The method of claim 5309 wherein the agent is a MMP inhibitor.
5404. The method of claim 5309 wherein the agent is an NF kappa B inhibitor.
5405. The method of claim 5309 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
5406. The method of claim 5309 wherein the agent is an NO antagonist.
5407. The method of claim 5309 wherein the agent is a p38 MAP kinase inhibitor.
5408. The method of claim 5309 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
5409. The method of claim 5309 wherein the agent is a phosphodiesterase inhibitor.
5410. The method of claim 5309 wherein the agent is a TGF beta inhibitor.
5411. The method of claim 5309 wherein the agent is a thromboxane A2 antagonist.
5412. The method of claim 5309 wherein the agent is a TNF alpha antagonist.
5413. The method of claim 5309 wherein the agent is a TACE inhibitor.
5414. The method of claim 5309 wherein the agent is a tyrosine kinase inhibitor.
5415. The method of claim 5309 wherein the agent is a vitronectin inhibitor.
5416. The method of claim 5309 wherein the agent is a fibroblast growth factor inhibitor.
5417. The method of claim 5309 wherein the agent is a protein kinase inhibitor.
5418. The method of claim 5309 wherein the agent is a PDGF receptor kinase inhibitor.
5419. The method of claim 5309 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
5420. The method of claim 5309 wherein the agent is a retinoic acid receptor antagonist.
5421. The method of claim 5309 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
5422. The method of claim 5309 wherein the agent is a fibrinogen antagonist.
5423. The method of claim 5309 wherein the agent is an antimycotic agent.
5424. The method of claim 5309 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
5425. The method of claim 5309 wherein the agent is a bisphosphonate.
5426. The method of claim 5309 wherein the agent is a phospholipase A1 inhibitor.
5427. The method of claim 5309 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
5428. The method of claim 5309 wherein the agent is a macrolide antibiotic.
5429. The method of claim 5309 wherein the agent is a GPIIb/llla receptor antagonist.
5430. The method of claim 5309 wherein the agent is an endothelin receptor antagonist.
5431. The method of claim 5309 wherein the agent is a peroxisome proliferator-activated receptor agonist
5432. The method of claim 5309 wherein the agent is an estrogen receptor agent.
5433. The method of claim 5309 wherein the agent is a somastostatin analogue.
5434. The method of claim 5309 wherein the agent is a neurokinin 1 antagonist.
5435. The method of claim 5309 wherein the agent is a neurokinin 3 antagonist.
5436. The method of claim 5309 wherein the agent is a VLA-4 antagonist.
5437. The method of claim 5309 wherein the agent is an osteoclast inhibitor.
5438. The method of claim 5309 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
5439. The method of claim 5309 wherein the agent is an angiotensin I converting enzyme inhibitor.
5440. The method of claim 5309 wherein the agent is an angiotensin II antagonist.
5441. The method of claim 5309 wherein the agent is an enkephalinase inhibitor.
5442. The method of claim 5309 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
5443. The method of claim 5309 wherein the agent is a protein kinase C inhibitor.
5444. The method of claim 5309 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
5445. The method of claim 5309 wherein the agent is a CXCR3 inhibitor.
5446. The method of claim 5309 wherein the agent is an Itk inhibitor.
5447. The method of claim 5309 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
5448. The method of claim 5309 wherein the agent is a PPAR agonist.
5449. The method of claim 5309 wherein the agent is an immunosuppressant
5450. The method of claim 5309 wherein the agent is an Erb inhibitor.
5451. The method of claim 5309 wherein the agent is an apoptosis agonist.
5452. The method of claim 5309 wherein the agent is a lipocortin agonist.
5453. The method of claim 5309 wherein the agent is a VCAM-1 antagonist.
5454. The method of claim 5309 wherein the agent is a collagen antagonist.
5455. The method of claim 5309 wherein the agent is an alpha 2 integrin antagonist.
5456. The method of claim 5309 wherein the agent is a TNF alpha inhibitor.
5457. The method of claim 5309 wherein the agent is a nitric oxide inhibitor
5458. The method of claim 5309 wherein the agent is a cathepsin inhibitor.
5459. The method of claim 5309 wherein the agent is not an anti- inflammatory agent
5460. The method of claim 5309 wherein the agent is not a steroid.
5461. The method of claim 5309 wherein the agent is not a glucocorticosteroid.
5462. The method of claim 5309 wherein the agent is not dexamethasone.
5463. The method of claim 5309 wherein the agent is not beclomethasone.
5464. The method of claim 5309 wherein the agent is not dipropionate.
5465. The method of claim 5309 wherein the agent is not an anti- infective agent.
5466. The method of claim 5309 wherein the agent is not an antibiotic.
5467. The method of claim 5309 wherein the agent is not an anti- fungal agent.
5468. The method of claim 5309, wherein the composition comprises a polymer.
5469. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a copolymer.
5470. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a block copolymer.
5471. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a random copolymer.
5472. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a biodegradable polymer.
5473. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-biodegradable polymer.
5474. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophilic polymer.
5475. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophobic polymer.
5476. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophilic domains.
5477. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophobic domains.
5478. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-conductive polymer.
5479. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, an elastomer.
5480. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrogel.
5481. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a silicone polymer.
5482. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrocarbon polymer.
5483. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a styrene-derived polymer.
5484. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a butadiene-derived polymer.
5485. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a macromer.
5486. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, a poly( ethylene glycol) polymer.
5487. The method of claim 5309, wherein the composition comprises a polymer, and the polymer is, or comprises, an amorphous polymer.
5488. The method of claim 5309, wherein the composition further comprises a second pharmaceutically active agent.
5489. The method of claim 5309, wherein the composition further comprises an anti-inflammatory agent
5490. The method of claim 5309, wherein the composition further comprises an agent that inhibits infection.
5491. The method of claim 5309, wherein the composition further comprises an anthracycline.
5492. The method of claim 5309, wherein the composition further comprises doxorubicin.
5493. The method of claim 5309 wherein the composition further comprises mitoxantrone.
5494. The method of claim 5309 wherein the composition further comprises a fluoropyrimidine.
5495. The method of claim 5309, wherein the composition further comprises 5-fluorouracil (5-FU).
5496. The method of claim 5309, wherein the composition further comprises a folic acid antagonist
5497. The method of claim 5309, wherein the composition further comprises methotrexate.
5498. The method of claim 5309, wherein the composition further comprises a podophylotoxin.
5499. The method of claim 5309, wherein the composition further comprises etoposide.
5500. The method of claim 5309, wherein the composition further comprises camptothecin.
5501. The method of claim 5309, wherein the composition further comprises a hydroxyurea.
5502. The method of claim 5309, wherein the composition further comprises a platinum complex.
5503. The method of claim 5309, wherein the composition further comprises cisplatin.
5504. The method of claim 5309 wherein the composition further comprises an anti-thrombotic agent
5505. The method of claim 5309, wherein the composition further comprises a visualization agent.
5506. The method of claim 5309, wherein the composition further comprises a visualization agent, and the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
5507. The method of claim 5309, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, barium, tantalum, or technetium.
5508. The method of claim 5309, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, an MRI responsive material.
5509. The method of claim 5309, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a gadolinium chelate.
5510. The method of claim 5309, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron, magnesium, manganese, copper, or chromium.
5511. The method of claim 5309, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron oxide compound.
5512. The method of claim 5309, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a dye, pigment, or colorant.
5513. The method of claim 5309 wherein the agent is released in effective concentrations from the composition comprising the agent by diffusion over a period ranging from the time of administration to about 90 days.
5514. The method of claim 5309 wherein the agent is released in effective concentrations from the composition comprising the agent by erosion of the composition over a period ranging from the time of administration to about 90 days.
5515. The method of claim 5309 wherein the composition further comprises an inflammatory cytokine.
5516. The method of claim 5309 wherein the composition further comprises an agent that stimulates cell proliferation.
5517. The method of claim 5309 wherein the composition further comprises a polymeric carrier.
5518. The method of claim 5309 wherein the composition is in the form of a gel, paste, or spray.
5519. The method of claim 5309 wherein the sensor is partially constructed with the agent or the composition.
5520. The method of claim 5309 wherein the sensor is impregnated with the agent or the composition.
5521. The method of claim 5309, wherein the agent or the composition forms a coating, and the coating directly contacts the sensor.
5522. The method of claim 5309, wherein the agent or the composition forms a coating, and the coating indirectly contacts the sensor.
5523. The method of claim 5309 wherein the agent or the composition forms a coating, and the coating partially covers the sensor.
5524. The method of claim 5309, wherein the agent or the composition forms a coating, and the coating completely covers the sensor.
5525. The method of claim 5309 wherein the agent or the composition is located within pores or holes of the sensor.
5526. The method of claim 5309 wherein the agent or the composition is located within a channel, lumen, or divet of the sensor.
5527. The method of claim 5309 wherein the sensor further comprises an echogenic material.
5528. The method of claim 5309 wherein the sensor further comprises an echogenic material, wherein the echogenic material is in the form of a coating.
5529. The method of claim 5309 wherein the sensor is sterile.
5530. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor.
5531. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is connective tissue.
5532. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is muscle tissue.
5533. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is nerve tissue.
5534. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the agent is released into tissue in the vicinity of the sensor after deployment of the sensor, wherein the tissue is epithelium tissue.
5535. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from the time of deployment of the sensor to about 1 year.
5536. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 month to 6 months.
5537. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor over a period ranging from about 1 - 90 days.
5538. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a constant rate.
5539. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at an increasing rate.
5540. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the agent is released in effective concentrations from the sensor at a decreasing rate.
5541. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the sensor comprises about 0.01 μg to about 10 μg of the agent.
5542. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 μg to about 10 mg of the agent.
5543. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the sensor comprises about 10 mg to about 250 mg of the agent.
5544. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the sensor comprises about 250 mg to about 1000 mg of the agent.
5545. The method of claim 5309 wherein the agent is delivered from the sensor, wherein the sensor comprises about 1000 mg to about 2500 mg of the agent.
5546. The method of claim 5309 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises less than 0.01 μg of the agent per mm2 of sensor surface to which the agent is applied.
5547. The method of claim 5309 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 0.01 μg to about 1 μg of the agent per mm2 of sensor surface to which the agent is applied.
5548. The method of claim 5309 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1 μg to about 10 μg of the agent per mm2 of sensor surface to which the agent is applied.
5549. The method of claim 5309 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 10 μg to about 250 μg of the agent per mm2 of sensor surface to which the agent is applied.
5550. The method of claim 5309 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 250 μg to about 1000 μg of the agent per mm2 of sensor surface to which the agent is applied.
5551. The method of claim 5309 wherein the agent is delivered from the sensor, wherein a surface of the sensor comprises about 1000 μg to about 2500 μg of the agent per mm2 of sensor surface to which the agent is applied.
5552. The method of claim 5309, wherein the sensor further comprises a coating, and the coating is a uniform coating.
5553. The method of claim 5309, wherein the sensor further comprises a coating, and the coating is a non-uniform coating.
5554. The method of claim 5309, wherein the sensor further comprises a coating, and the coating is a discontinuous coating.
5555. The method of claim 5309, wherein the sensor further comprises a coating, and the coating is a patterned coating.
5556. The method of claim 5309, wherein the sensor further comprises a coating, and the coating has a thickness of 100 μm or less.
5557. The method of claim 5309, wherein the sensor further comprises a coating, and the coating has a thickness of 10 μm or less.
5558. The method of claim 5309, wherein the sensor further comprises a coating, and the coating adheres to the surface of the sensor upon deployment of the sensor.
5559. The method of claim 5309, wherein the sensor further comprises a coating, and the coating is stable at room temperature for a period of at least 1 year.
5560. The method of claim 5309, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 0.0001% to about 1 % by weight.
5561. The method of claim 5309, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
5562. The method of claim 5309, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
5563. The method of claim 5309, wherein the sensor further comprises a coating, and the agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
5564. The method of claim 5309, wherein the sensor further comprises a coating, and the coating comprises a polymer.
5565. The method of claim 5309, wherein the sensor comprises a first coating having a first composition and a second coating having a second composition.
5566. The method of claim 5309, wherein the sensor comprises a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
5567. The method of claim 5309 wherein the agent or the composition is affixed to the sensor.
5568. The method of claim 5309 wherein the agent or the composition is covalently attached to the sensor.
5569. The method of claim 5309 wherein the agent or the composition is non-covalently attached to the sensor.
5570. The method of claim 5309 wherein the sensor comprises a coating that absorbs the agent or the composition.
5571. The method of claim 5309 wherein the sensor is interweaved with a thread composed of, or coated with, the agent or the composition.
5572. The method of claim 5309 wherein a portion of the sensor is covered with a sleeve that contains the agent or the composition.
5573. The method of claim 5309 wherein the sensor is completely covered with a sleeve that contains the agent or the composition.
5574. The method of claim 5309 wherein a portion of the sensor is covered with a mesh that contains the agent or the composition.
5575. The method of claim 5309 wherein the sensor is completely covered with a mesh that contains the agent or the composition.
5576. The method of claim 5309 wherein the sensor is linked to a pump.
5577. The method of claim 5309 wherein the agent or the composition is applied to the sensor surface prior to to the placing of the sensor into the host.
5578. The method of claim 5309 wherein the agent or the composition is applied to the sensor surface during the placing of the sensor into the host.
5579. The method of claim 5309 wherein the agent or the composition is applied to the sensor surface immediately after the placing of the sensor into the host.
5580. The method of claim 5309 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor prior to to the placing of the sensor into the host.
5581. The method of claim 5309 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor during the placing of the sensor into the host.
5582. The method of claim 5309 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the sensor immediately after the placing of the sensor into the host.
5583. The method of claim 5309 wherein the agent or the composition is topically applied into the anatomical space where the sensor is placed.
5584. The method of claim 5309 wherein the agent or the composition is percutaneously injected into the tissue in the host surrounding the sensor.
5585. The method of any one of claims 5309-5584 wherein the sensor emits a source of radiation directed towards blood to interact with a plurality of detectors that provide an output signal.
5586. The method of any one of claims 5309-5584 wherein the sensor is a biosensing transponder composed of a dye that has optical properties that change in response to changes in the environment, a photosensor to sense the optical changes, and a transponder for transmitting data to a remote reader.
5587. The method of any one of claims 5309-5584 wherein the sensor is a monolithic bioelectronic device for detecting at least one analyte within the host.
5588. A method for inhibiting scarring comprising placing a pump and an anti-scarring agent or a composition comprising an anti-scarring agent into an animal host, wherein the agent inhibits scarring.
5589. The method of claim 5588 wherein the agent inhibits cell regeneration.
5590. The method of claim 5588 wherein the agent inhibits angiogenesis.
5591. The method of claim 5588 wherein the agent inhibits fibroblast migration.
5592. The method of claim 5588 wherein the agent inhibits fibroblast proliferation.
5593. The method of claim 5588 wherein the agent inhibits deposition of extracellular matrix.
5594. The method of claim 5588 wherein the agent inhibits tissue remodeling.
5595. The method of claim 5588 wherein the agent is an angiogenesis inhibitor.
5596. The method of claim 5588 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
5597. The method of claim 5588 wherein the agent is a chemokine receptor antagonist.
5598. The method of claim 5588 wherein the agent is a cell cycle inhibitor.
5599. The method of claim 5588 wherein the agent is a taxane.
5600. The method of claim 5588 wherein the agent is an antimicrotubule agent.
5601. The method of claim 5588 wherein the agent is paclitaxel.
5602. The method of claim 5588 wherein the agent is not paclitaxel.
5603. The method of claim 5588 wherein the agent is an analogue or derivative of paclitaxel.
5604. The method of claim 5588 wherein the agent is a vinca alkaloid.
5605. The method of claim 5588 wherein the agent is camptothecin or an analogue or derivative thereof.
5606. The method of claim 5588 wherein the agent is a podophyllotoxin.
5607. The method of claim 5588 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
5608. The method of claim 5588 wherein the agent is an anthracycline.
5609. The method of claim 5588 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
5610. The method of claim 5588 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
5611. The method of claim 5588 wherein the agent is a platinum compound.
5612. The method of claim 5588 wherein the agent is a nitrosourea.
5613. The method of claim 5588 wherein the agent is a nitroimidazole.
5614. The method of claim 5588 wherein the agent is a folic acid antagonist.
5615. The method of claim 5588 wherein the agent is a cytidine analogue.
5616. The method of claim 5588 wherein the agent is a pyrimidine analogue.
5617. The method of claim 5588 wherein the agent is a fluoropyrimidine analogue.
5618. The method of claim 5588 wherein the agent is a purine analogue.
5619. The method of claim 5588 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
5620. The method of claim 5588 wherein the agent is a hydroxyurea.
5621. The method of claim 5588 wherein the agent is a mytomicin or an analogue or derivative thereof.
5622. The method of claim 5588 wherein the agent is an alkyl sulfonate.
5623. The method of claim 5588 wherein the agent is a benzamide or an analogue or derivative thereof.
5624. The method of claim 5588 wherein the agent is a nicotinamide or an analogue or derivative thereof.
5625. The method of claim 5588 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
5626. The method of claim 5588 wherein the agent is a DNA alkylating agent.
5627. The method of claim 5588 wherein the agent is an antimicrotubule agent.
5628. The method of claim 5588 wherein the agent is a topoisomerase inhibitor.
5629. The method of claim 5588 wherein the agent is a DNA cleaving agent.
5630. The method of claim 5588 wherein the agent is an antimetabolite.
5631. The method of claim 5588 wherein the agent inhibits adenosine deaminase.
5632. The method of claim 5588 wherein the agent inhibits purine ring synthesis.
5633. The method of claim 5588 wherein the agent is a nucleotide interconversion inhibitor.
5634. The method of claim 5588 wherein the agent inhibits dihydrofolate reduction.
5635. The method of claim 5588 wherein the agent blocks thymidine monophosphate.
5636. The method of claim 5588 wherein the agent causes DNA damage.
5637. The method of claim 5588 wherein the agent is a DNA intercalation agent
5638. The method of claim 5588 wherein the agent is a RNA synthesis inhibitor.
5639. The method of claim 5588 wherein the agent is a pyrimidine synthesis inhibitor.
5640. The method of claim 5588 wherein the agent inhibits ribonucleotide synthesis or function.
5641. The method of claim 5588 wherein the agent inhibits thymidine monophosphate synthesis or function.
5642. The method of claim 5588 wherein the agent inhibits DNA synthesis.
5643. The method of claim 5588 wherein the agent causes DNA adduct formation.
5644. The method of claim 5588 wherein the agent inhibits protein synthesis.
5645. The method of claim 5588 wherein the agent inhibits microtubule function.
5646. The method of claim 5588 wherein the agent is a cyclin dependent protein kinase inhibitor.
5647. The method of claim 5588 wherein the agent is an epidermal growth factor kinase inhibitor.
5648. The method of claim 5588 wherein the agent is an elastase inhibitor.
5649. The method of claim 5588 wherein the agent is a factor Xa inhibitor.
5650. The method of claim 5588 wherein the agent is a farnesyltransferase inhibitor.
5651. The method of claim 5588 wherein the agent is a fibrinogen antagonist.
5652. The method of claim 5588 wherein the agent is a guanylate cyclase stimulant.
5653. The method of claim 5588 wherein the agent is a heat shock protein 90 antagonist.
5654. The method of claim 5588 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
5655. The method of claim 5588 wherein the agent is a guanylate cyclase stimulant.
5656. The method of claim 5588 wherein the agent is a HMGCoA reductase inhibitor.
5657. The method of claim 5588 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
5658. The method of claim 5588 wherein the agent is a hydroorotate dehydrogenase inhibitor.
5659. The method of claim 5588 wherein the agent is an IKK2 inhibitor.
5660. The method of claim 5588 wherein the agent is an IL-1 antagonist.
5661. The method of claim 5588 wherein the agent is an ICE antagonist.
5662. The method of claim 5588 wherein the agent is an IRAK antagonist.
5663. The method of claim 5588 wherein the agent is an IL-4 agonist.
5664. The method of claim 5588 wherein the agent is an immunomodulatory agent.
5665. The method of claim 5588 wherein the agent is sirolimus or an analogue or derivative thereof.
5666. The method of claim 5588 wherein the agent is not sirolimus.
5667. The method of claim 5588 wherein the agent is everolimus or an analogue or derivative thereof.
5668. The method of claim 5588 wherein the agent is tacrolimus or an analogue or derivative thereof.
5669. The method of claim 5588 wherein the agent is not tacrolimus.
5670. The method of claim 5588 wherein the agent is biolmus or an analogue or derivative thereof.
5671. The method of claim 5588 wherein the agent is tresperimus or an analogue or derivative thereof.
5672. The method of claim 5588 wherein the agent is auranofin or an analogue or derivative thereof.
5673. The method of claim 5588 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
5674. The method of claim 5588 wherein the agent is gusperimus or an analogue or derivative thereof.
5675. The method of claim 5588 wherein the agent is pimecrolimus or an analogue or derivative thereof.
5676. The method of claim 5588 wherein the agent is ABT-578 or an analogue or derivative thereof.
5677. The method of claim 5588 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
5678. The method of claim 5588 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
5679. The method of claim 5588 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
5680. The method of claim 5588 wherein the agent is a leukotriene inhibitor.
5681. The method of claim 5588 wherein the agent is a MCP-1 antagonist.
5682. The method of claim 5588 wherein the agent is a MMP inhibitor.
5683. The method of claim 5588 wherein the agent is an NF kappa B inhibitor.
5684. The method of claim 5588 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
5685. The method of claim 5588 wherein the agent is an NO antagonist.
5686. The method of claim 5588 wherein the agent is a p38 MAP kinase inhibitor.
5687. The method of claim 5588 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
5688. The method of claim 5588 wherein the agent is a phosphodiesterase inhibitor.
5689. The method of claim 5588 wherein the agent is a TGF beta inhibitor.
5690. The method of claim 5588 wherein the agent is a thromboxane A2 antagonist.
5691. The method of claim 5588 wherein the agent is a TNF alpha antagonist.
5692. The method of claim 5588 wherein the agent is a TACE inhibitor.
5693. The method of claim 5588 wherein the agent is a tyrosine kinase inhibitor.
5694. The method of claim 5588 wherein the agent is a vitronectin inhibitor.
5695. The method of claim 5588 wherein the agent is a fibroblast growth factor inhibitor.
5696. The method of claim 5588 wherein the agent is a protein kinase inhibitor.
5697. The method of claim 5588 wherein the agent is a PDGF receptor kinase inhibitor.
5698. The method of claim 5588 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
5699. The method of claim 5588 wherein the agent is a retinoic acid receptor antagonist.
5700. The method of claim 5588 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
5701. The method of claim 5588 wherein the agent is a fibrinogen antagonist.
5702. The method of claim 5588 wherein the agent is an antimycotic agent.
5703. The method of claim 5588 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
5704. The method of claim 5588 wherein the agent is a bisphosphonate.
5705. The method of claim 5588 wherein the agent is a phospholipase A1 inhibitor.
5706. The method of claim 5588 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
5707. The method of claim 5588 wherein the agent is a macrolide antibiotic.
5708. The method of claim 5588 wherein the agent is a GPIIb/llla receptor antagonist.
5709. The method of claim 5588 wherein the agent is an endothelin receptor antagonist.
5710. The method of claim 5588 wherein the agent is a peroxisome proliferator-activated receptor agonist.
5711. The method of claim 5588 wherein the agent is an estrogen receptor agent.
5712. The method of claim 5588 wherein the agent is a somastostatin analogue.
5713. The method of claim 5588 wherein the agent is a neurokinin 1 antagonist.
5714. The method of claim 5588 wherein the agent is a neurokinin 3 antagonist.
5715. The method of claim 5588 wherein the agent is a VLA-4 antagonist.
5716. The method of claim 5588 wherein the agent is an osteoclast inhibitor.
5717. The method of claim 5588 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
5718. The method of claim 5588 wherein the agent is an angiotensin I converting enzyme inhibitor.
5719. The method of claim 5588 wherein the agent is an angiotensin II antagonist.
5720. The method of claim 5588 wherein the agent is an enkephalinase inhibitor.
5721. The method of claim 5588 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
5722. The method of claim 5588 wherein the agent is a protein kinase C inhibitor.
5723. The method of claim 5588 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
5724. The method of claim 5588 wherein the agent is a CXCR3 inhibitor.
5725. The method of claim 5588 wherein the agent is an Itk inhibitor.
5726. The method of claim 5588 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
5727. The method of claim 5588 wherein the agent is a PPAR agonist.
5728. The method of claim 5588 wherein the agent is an immunosuppressant
5729. The method of claim 5588 wherein the agent is an Erb inhibitor.
5730. The method of claim 5588 wherein the agent is an apoptosis agonist.
5731. The method of claim 5588 wherein the agent is a lipocortin agonist.
5732. The method of claim 5588 wherein the agent is a VCAM-1 antagonist.
5733. The method of claim 5588 wherein the agent is a collagen antagonist.
5734. The method of claim 5588 wherein the agent is an alpha 2 integrin antagonist
5735. The method of claim 5588 wherein the agent is a TNF alpha inhibitor.
5736. The method of claim 5588 wherein the agent is a nitric oxide inhibitor
5737. The method of claim 5588 wherein the agent is a cathepsin inhibitor.
5738. The method of claim 5588 wherein the agent is not an anti- inflammatory agent.
5739. The method of claim 5588 wherein the agent is not a steroid.
5740. The method of claim 5588 wherein the agent is not a glucocorticosteroid.
5741. The method of claim 5588 wherein the agent is not dexamethasone.
5742. The method of claim 5588 wherein the agent is not beclomethasone.
5743. The method of claim 5588 wherein the agent is not dipropionate.
5744. The method of claim 5588 wherein the agent is not an anti- infective agent.
5745. The method of claim 5588 wherein the agent is not an antibiotic.
5746. The method of claim 5588 wherein the agent is not an anti- fungal agent
5747. The method of claim 5588, wherein the composition comprises a polymer.
5748. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a copolymer.
5749. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a block copolymer.
5750. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a random copolymer.
5751. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a biodegradable polymer.
5752. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-biodegradable polymer.
5753. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophilic polymer.
5754. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophobic polymer.
5755. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophilic domains.
5756. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophobic domains.
5757. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-conductive polymer.
5758. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, an elastomer.
5759. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrogel.
5760. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a silicone polymer.
5761. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrocarbon polymer.
5762. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a styrene-derived polymer.
5763. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a butadiene-derived polymer.
5764. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a macromer.
5765. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, a poly(ethylene glycol) polymer.
5766. The method of claim 5588, wherein the composition comprises a polymer, and the polymer is, or comprises, an amorphous polymer.
5767. The method of claim 5588, wherein the composition further comprises a second pharmaceutically active agent.
5768. The method of claim 5588, wherein the composition further comprises an anti-inflammatory agent.
5769. The method of claim 5588, wherein the composition further comprises an agent that inhibits infection.
5770. The method of claim 5588, wherein the composition further comprises an anthracycline.
5771. The method of claim 5588, wherein the composition further comprises doxorubicin.
5772. The method of claim 5588 wherein the composition further comprises mitoxantrone.
5773. The method of claim 5588 wherein the composition further comprises a fluoropyrimidine.
5774. The method of claim 5588, wherein the composition further comprises 5-fluorouracil (5-FU).
5775. The method of claim 5588, wherein the composition further comprises a folic acid antagonist.
5776. The method of claim 5588, wherein the composition further comprises methotrexate.
5777. The method of claim 5588, wherein the composition further comprises a podophylotoxin.
5778. The method of claim 5588, wherein the composition further comprises etoposide.
5779. The method of claim 5588, wherein the composition further comprises camptothecin.
5780. The method of claim 5588, wherein the composition further comprises a hydroxyurea.
5781. The method of claim 5588, wherein the composition further comprises a platinum complex.
5782. The method of claim 5588, wherein the composition further comprises cisplatin.
5783. The method of claim 5588 wherein the composition further comprises an anti-thrombotic agent.
5784. The method of claim 5588, wherein the composition further comprises a visualization agent
5785. The method of claim 5588, wherein the composition further comprises a visualization agent, and the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
5786. The method of claim 5588, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, barium, tantalum, or technetium.
5787. The method of claim 5588, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, an MRI responsive material.
5788. The method of claim 5588, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a gadolinium chelate.
5789. The method of claim 5588, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron, magnesium, manganese, copper, or chromium.
5790. The method of claim 5588, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron oxide compound.
5791. The method of claim 5588, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a dye, pigment, or colorant.
5792. The method of claim 5588 wherein the agent is released in effective concentrations from the composition comprising the agent by diffusion over a period ranging from the time of administration to about 90 days.
5793. The method of claim 5588 wherein the agent is released in effective concentrations from the composition comprising the agent by erosion of the composition over a period ranging from the time of administration to about 90 days.
5794. The method of claim 5588 wherein the composition further comprises an inflammatory cytokine.
5795. The method of claim 5588 wherein the composition further comprises an agent that stimulates cell proliferation.
5796. The method of claim 5588 wherein the composition further comprises a polymeric carrier.
5797. The method of claim 5588 wherein the composition is in the form of a gel, paste, or spray.
5798. The method of claim 5588 wherein the pump is partially constructed with the agent or the composition.
5799. The method of claim 5588 wherein the pump is impregnated with the agent or the composition.
5800. The method of claim 5588, wherein the agent or the composition forms a coating, and the coating directly contacts the pump.
5801. The method of claim 5588, wherein the agent or the composition forms a coating, and the coating indirectly contacts the pump.
5802. The method of claim 5588 wherein the agent or the composition forms a coating, and the coating partially covers the pump.
5803. The method of claim 5588, wherein the agent or the composition forms a coating, and the coating completely covers the pump.
5804. The method of claim 5588 wherein the agent or the composition is located within pores or holes of the pump.
5805. The method of claim 5588 wherein the agent or the composition is located within a channel, lumen, or divet of the pump.
5806. The method of claim 5588 wherein the pump further comprises an echogenic material.
5807. The method of claim 5588 wherein the pump further comprises an echogenic material, wherein the echogenic material is in the form of a coating.
5808. The method of claim 5588 wherein the pump is sterile.
5809. The method of claim 5588 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump.
5810. The method of claim 5588 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is connective tissue.
5811. The method of claim 5588 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is muscle tissue.
5812. The method of claim 5588 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is nerve tissue.
5813. The method of claim 5588 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is epithelium tissue.
5814. The method of claim 5588 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from the time of deployment of the pump to about 1 year.
5815. The method of claim 5588 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from about 1 month to 6 months.
5816. The method of claim 5588 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from about 1 - 90 days.
5817. The method of claim 5588 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at a constant rate.
5818. The method of claim 5588 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at an increasing rate.
5819. The method of claim 5588 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at a decreasing rate.
5820. The method of claim 5588 wherein the agent is delivered from the pump, wherein the pump comprises about 0.01 μg to about 10 μg of the agent.
5821. The method of claim 5588 wherein the agent is delivered from the pump, wherein the pump comprises about 10 μg to about 10 mg of the agent.
5822. The method of claim 5588 wherein the agent is delivered from the pump, wherein the pump comprises about 10 mg to about 250 mg of the agent.
5823. The method of claim 5588 wherein the agent is delivered from the pump, wherein the pump comprises about 250 mg to about 1000 mg of the agent.
5824. The method of claim 5588 wherein the agent is delivered from the pump, wherein the pump comprises about 1000 mg to about 2500 mg of the agent.
5825. The method of claim 5588 wherein the agent is delivered from the pump, wherein a surface of the pump comprises less than 0.01 μg of the agent per mm2 of pump surface to which the agent is applied.
5826. The method of claim 5588 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 0.01 μg to about 1 μg of the agent per mm2 of pump surface to which the agent is applied.
5827. The method of claim 5588 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 1 μg to about 10 μg of the agent per mm2 of pump surface to which the agent is applied.
5828. The method of claim 5588 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 10 μg to about 250 μg of the agent per mm2 of pump surface to which the agent is applied.
5829. The method of claim 5588 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 250 μg to about 1000 μg of the agent per mm2 of pump surface to which the agent is applied.
5830. The method of claim 5588 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 1000 μg to about 2500 μg of the agent per mm2 of pump surface to which the agent is applied.
5831. The method of claim 5588, wherein the pump further comprises a coating, and the coating is a uniform coating.
5832. The method of claim 5588, wherein the pump further comprises a coating, and the coating is a non-uniform coating.
5833. The method of claim 5588, wherein the pump further comprises a coating, and the coating is a discontinuous coating.
5834. The method of claim 5588, wherein the pump further comprises a coating, and the coating is a patterned coating.
5835. The method of claim 5588, wherein the pump further comprises a coating, and the coating has a thickness of 100 μm or less.
5836. The method of claim 5588, wherein the pump further comprises a coating, and the coating has a thickness of 10 μm or less.
5837. The method of claim 5588, wherein the pump further comprises a coating, and the coating adheres to the surface of the pump upon deployment of the pump.
5838. The method of claim 5588, wherein the pump further comprises a coating, and the coating is stable at room temperature for a period of at least 1 year.
5839. The method of claim 5588, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 0.0001 % to about 1% by weight.
5840. The method of claim 5588, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
5841. The method of claim 5588, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
5842. The method of claim 5588, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
5843. The method of claim 5588, wherein the pump further comprises a coating, and the coating comprises a polymer.
5844. The method of claim 5588, wherein the pump comprises a first coating having a first composition and a second coating having a second composition.
5845. The method of claim 5588, wherein the pump comprises a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
5846. The method of claim 5588 wherein the agent or the composition is affixed to the pump.
5847. The method of claim 5588 wherein the agent or the composition is covalently attached to the pump.
5848. The method of claim 5588 wherein the agent or the composition is non-covalently attached to the pump.
5849. The method of claim 5588 wherein the pump comprises a coating that absorbs the agent or the composition.
5850. The method of claim 5588 wherein the pump is interweaved with a thread composed of, or coated with, the agent or the composition.
5851. The method of claim 5588 wherein a portion of the pump is covered with a sleeve that contains the agent or the composition.
5852. The method of claim 5588 wherein the pump is completely covered with a sleeve that contains the agent or the composition.
5853. The method of claim 5588 wherein a portion of the pump is covered with a mesh that contains the agent or the composition.
5854. The method of claim 5588 wherein the pump is completely covered with a mesh that contains the agent or the composition.
5855. The method of claim 5588 wherein the pump is linked to a sensor.
5856. The method of claim 5588 wherein the agent or the composition is applied to the pump surface prior to to the placing of the pump into the host
5857. The method of claim 5588 wherein the agent or the composition is applied to the pump surface during the placing of the pump into the host.
5858. The method of claim 5588 wherein the agent or the composition is applied to the pump surface immediately after the placing of the pump into the host.
5859. The method of claim 5588 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump prior to to the placing of the pump into the host.
5860. The method of claim 5588 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump during the placing of the pump into the host
5861. The method of claim 5588 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump immediately after the placing of the pump into the host.
5862. The method of claim 5588 wherein the agent or the composition is topically applied into the anatomical space where the pump is placed.
5863. The method of claim 5588 wherein the agent or the composition is percutaneously injected into the tissue in the host surrounding the pump.
5864. The method of any one of claims 5588-5863 wherein the pump is adapted for delivering insulin.
5865. The method of any one of claims 5588-5863 wherein the pump is adapted for delivering a narcotic.
5866. The method of any one of claims 5588-5863 wherein the pump is adapted for delivering a chemotherapeutic agent.
5867. The method of any one of claims 5588-5863 wherein the pump is adapted for delivering an anti-arrhythmic drug.
5868. The method of any one of claims 5588-5863 wherein the pump is adapted for delivering an anti-spasmotic drug.
5869. The method of any one of claims 5588-5863 wherein the pump is adapted for delivering an anti-spastic agent
5870. The method of any one of claims 5588-5863 wherein the pump is adapted for delivering an antibiotic.
5871. The method of any one of claims 5588-5863 wherein the pump is adapted for delivering a drug only when changes in the host are detected.
5872. The method of any one of claims 5588-5863 wherein the pump is adapted for delivering a drug as a continuous slow release.
5873. The method of any one of claims 5588-5863 wherein the pump is adapted for delivering a drug at prescribed dosages in a pulsatile manner.
5874. The method of any one of claims 5588-5863 wherein the pump is a programmable drug delivery pump.
5875. The method of any one of claims 5588-5863 wherein the pump is adapted for intraocularly delivering a drug.
5876. The method of any one of claims 5588-5863 wherein the pump is adapted for intrathecally delivering a drug.
5877. The method of any one of claims 5588-5863 wherein the pump is adapted for intraperitoneally delivering a drug.
5878. The method of any one of claims 5588-5863 wherein the pump is adapted for intra-arterially delivering a drug.
5879. The method of any one of claims 5588-5863 wherein the pump is adapted for intracardiac delivery of a drug.
5880. The method of any one of claims 5588-5863 wherein the pump is an implantable osmotic pump.
5881. The method of any one of claims 5588-5863 wherein the pump is an ocular drug delivery pump.
5882. The method of any one of claims 5588-5863 wherein the pump is metering system.
5883. The method of any one of claims 5588-5863 wherein the pump is a peristaltic (roller) pump.
5884. The method of any one of claims 5588-5863 wherein the pump is an electronically driven pump.
5885. The method of any one of claims 5588-5863 wherein the pump is an elastomeric pump.
5886. The method of any one of claims 5588-5863 wherein the pump is a spring contraction pump.
5887. The method of any one of claims 5588-5863 wherein the pump is a gas-driven pump.
5888. The method of any one of claims 5588-5863 wherein the pump is a hydraulic pump.
5889. The method of any one of claims 5588-5863 wherein the pump is a piston-dependent pump.
5890. The method of any one of claims 5588-5863 wherein the pump is a non-piston-dependent pump.
5891. The method of any one of claims 5588-5863 wherein the pump is a dispensing chamber.
5892. The method of any one of claims 5588-5863 wherein the pump is an infusion pump.
5893. The method of any one of claims 5588-5863 wherein the pump is a passive pump.
5894. A method for inhibiting scarring comprising placing an implantable insulin pump and an anti-scarring agent or a composition comprising an anti-scarring agent into an animal host, wherein the agent inhibits scarring.
5895. The method of claim 5894 wherein the agent inhibits cell regeneration.
5896. The method of claim 5894 wherein the agent inhibits angiogenesis.
5897. The method of claim 5894 wherein the agent inhibits fibroblast migration.
5898. The method of claim 5894 wherein the agent inhibits fibroblast proliferation.
5899. The method of claim 5894 wherein the agent inhibits deposition of extracellular matrix.
5900. The method of claim 5894 wherein the agent inhibits tissue remodeling.
5901. The method of claim 5894 wherein the agent is an angiogenesis inhibitor.
5902. The method of claim 5894 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
5903. The method of claim 5894 wherein the agent is a chemokine receptor antagonist.
5904. The method of claim 5894 wherein the agent is a cell cycle inhibitor.
5905. The method of claim 5894 wherein the agent is a taxane.
5906. The method of claim 5894 wherein the agent is an antimicrotubule agent.
5907. The method of claim 5894 wherein the agent is paclitaxel.
5908. The method of claim 5894 wherein the agent is not paclitaxel.
5909. The method of claim 5894 wherein the agent is an analogue or derivative of paclitaxel.
5910. The method of claim 5894 wherein the agent is a vinca alkaloid.
5911. The method of claim 5894 wherein the agent is camptothecin or an analogue or derivative thereof.
5912. The method of claim 5894 wherein the agent is a podophyllotoxin.
5913. The method of claim 5894 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
5914. The method of claim 5894 wherein the agent is an anthracycline.
5915. The method of claim 5894 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
5916. The method of claim 5894 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
5917. The method of claim 5894 wherein the agent is a platinum compound.
5918. The method of claim 5894 wherein the agent is a nitrosourea.
5919. The method of claim 5894 wherein the agent is a nitroimidazole.
5920. The method of claim 5894 wherein the agent is a folic acid antagonist.
5921. The method of claim 5894 wherein the agent is a cytidine analogue.
5922. The method of claim 5894 wherein the agent is a pyrimidine analogue.
5923. The method of claim 5894 wherein the agent is a fluoropyrimidine analogue.
5924. The method of claim 5894 wherein the agent is a purine analogue.
5925. The method of claim 5894 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
5926. The method of claim 5894 wherein the agent is a hydroxyurea.
5927. The method of claim 5894 wherein the agent is a mytomicin or an analogue or derivative thereof.
5928. The method of claim 5894 wherein the agent is an alkyl sulfonate.
5929. The method of claim 5894 wherein the agent is a benzamide or an analogue or derivative thereof.
5930. The method of claim 5894 wherein the agent is a nicotinamide or an analogue or derivative thereof.
5931. The method of claim 5894 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
5932. The method of claim 5894 wherein the agent is a DNA alkylating agent.
5933. The method of claim 5894 wherein the agent is an antimicrotubule agent.
5934. The method of claim 5894 wherein the agent is a topoisomerase inhibitor.
5935. The method of claim 5894 wherein the agent is a DNA cleaving agent.
5936. The method of claim 5894 wherein the agent is an antimetabolite.
5937. The method of claim 5894 wherein the agent inhibits adenosine deaminase.
5938. The method of claim 5894 wherein the agent inhibits purine ring synthesis.
5939. The method of claim 5894 wherein the agent is a nucleotide interconversion inhibitor.
5940. The method of claim 5894 wherein the agent inhibits dihydrofolate reduction.
5941. The method of claim 5894 wherein the agent blocks thymidine monophosphate.
5942. The method of claim 5894 wherein the agent causes DNA damage.
5943. The method of claim 5894 wherein the agent is a DNA intercalation agent.
5944. The method of claim 5894 wherein the agent is a RNA synthesis inhibitor.
5945. The method of claim 5894 wherein the agent is a pyrimidine synthesis inhibitor.
5946. The method of claim 5894 wherein the agent inhibits ribonucleotide synthesis or function.
5947. The method of claim 5894 wherein the agent inhibits thymidine monophosphate synthesis or function.
5948. The method of claim 5894 wherein the agent inhibits DNA synthesis.
5949. The method of claim 5894 wherein the agent causes DNA adduct formation.
5950. The method of claim 5894 wherein the agent inhibits protein synthesis.
5951. The method of claim 5894 wherein the agent inhibits microtubule function.
5952. The method of claim 5894 wherein the agent is a cyclin dependent protein kinase inhibitor.
5953. The method of claim 5894 wherein the agent is an epidermal growth factor kinase inhibitor.
5954. The method of claim 5894 wherein the agent is an elastase inhibitor.
5955. The method of claim 5894 wherein the agent is a factor Xa inhibitor.
5956. The method of claim 5894 wherein the agent is a farnesyltransferase inhibitor.
5957. The method of claim 5894 wherein the agent is a fibrinogen antagonist.
5958. The method of claim 5894 wherein the agent is a guanylate cyclase stimulant.
5959. The method of claim 5894 wherein the agent is a heat shock protein 90 antagonist.
5960. The method of claim 5894 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
5961. The method of claim 5894 wherein the agent is a guanylate cyclase stimulant.
5962. The method of claim 5894 wherein the agent is a HMGCoA reductase inhibitor.
5963. The method of claim 5894 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
5964. The method of claim 5894 wherein the agent is a hydroorotate dehydrogenase inhibitor.
5965. The method of claim 5894 wherein the agent is an IKK2 inhibitor.
5966. The method of claim 5894 wherein the agent is an IL-1 antagonist.
5967. The method of claim 5894 wherein the agent is an ICE antagonist.
5968. The method of claim 5894 wherein the agent is an IRAK antagonist.
5969. The method of claim 5894 wherein the agent is an IL-4 agonist.
5970. The method of claim 5894 wherein the agent is an immunomodulatory agent.
5971. The method of claim 5894 wherein the agent is sirolimus or an analogue or derivative thereof.
5972. The method of claim 5894 wherein the agent is not sirolimus.
5973. The method of claim 5894 wherein the agent is everolimus or an analogue or derivative thereof.
5974. The method of claim 5894 wherein the agent is tacrolimus or an analogue or derivative thereof.
5975. The method of claim 5894 wherein the agent is not tacrolimus.
5976. The method of claim 5894 wherein the agent is biolmus or an analogue or derivative thereof.
5977. The method of claim 5894 wherein the agent is tresperimus or an analogue or derivative thereof.
5978. The method of claim 5894 wherein the agent is auranofin or an analogue or derivative thereof.
5979. The method of claim 5894 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
5980. The method of claim 5894 wherein the agent is gusperimus or an analogue or derivative thereof.
5981. The method of claim 5894 wherein the agent is pimecrolimus or an analogue or derivative thereof.
5982. The method of claim 5894 wherein the agent is ABT-578 or an analogue or derivative thereof.
5983. The method of claim 5894 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
5984. The method of claim 5894 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
5985. The method of claim 5894 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
5986. The method of claim 5894 wherein the agent is a leukotriene inhibitor.
5987. The method of claim 5894 wherein the agent is a MCP-1 antagonist.
5988. The method of claim 5894 wherein the agent is a MMP inhibitor.
5989. The method of claim 5894 wherein the agent is an NF kappa B inhibitor.
5990. The method of claim 5894 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
5991. The method of claim 5894 wherein the agent is an NO antagonist.
5992. The method of claim 5894 wherein the agent is a p38 MAP kinase inhibitor.
5993. The method of claim 5894 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
5994. The method of claim 5894 wherein the agent is a phosphodiesterase inhibitor.
5995. The method of claim 5894 wherein the agent is a TGF beta inhibitor.
5996. The method of claim 5894 wherein the agent is a thromboxane A2 antagonist.
5997. The method of claim 5894 wherein the agent is a TNF alpha antagonist.
5998. The method of claim 5894 wherein the agent is a TACE inhibitor.
5999. The method of claim 5894 wherein the agent is a tyrosine kinase inhibitor.
6000. The method of claim 5894 wherein the agent is a vitronectin inhibitor.
6001. The method of claim 5894 wherein the agent is a fibroblast growth factor inhibitor.
6002. The method of claim 5894 wherein the agent is a protein kinase inhibitor.
6003. The method of claim 5894 wherein the agent is a PDGF receptor kinase inhibitor.
6004. The method of claim 5894 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
6005. The method of claim 5894 wherein the agent is a retinoic acid receptor antagonist.
6006. The method of claim 5894 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
6007. The method of claim 5894 wherein the agent is a fibrinogen antagonist.
6008. The method of claim 5894 wherein the agent is an antimycotic agent.
6009. The method of claim 5894 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
6010. The method of claim 5894 wherein the agent is a bisphosphonate.
6011. The method of claim 5894 wherein the agent is a phospholipase A1 inhibitor.
6012. The method of claim 5894 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
6013. The method of claim 5894 wherein the agent is a macrolide antibiotic.
6014. The method of claim 5894 wherein the agent is a GPIIb/llla receptor antagonist.
6015. The method of claim 5894 wherein the agent is an endothelin receptor antagonist.
6016. The method of claim 5894 wherein the agent is a peroxisome proliferator-activated receptor agonist.
6017. The method of claim 5894 wherein the agent is an estrogen receptor agent.
6018. The method of claim 5894 wherein the agent is a somastostatin analogue.
6019. The method of claim 5894 wherein the agent is a neurokinin 1 antagonist.
6020. The method of claim 5894 wherein the agent is a neurokinin 3 antagonist.
6021. The method of claim 5894 wherein the agent is a VLA-4 antagonist.
6022. The method of claim 5894 wherein the agent is an osteoclast inhibitor.
6023. The method of claim 5894 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
6024. The method of claim 5894 wherein the agent is an angiotensin I converting enzyme inhibitor.
6025. The method of claim 5894 wherein the agent is an angiotensin II antagonist.
6026. The method of claim 5894 wherein the agent is an enkephalinase inhibitor.
6027. The method of claim 5894 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
6028. The method of claim 5894 wherein the agent is a protein kinase C inhibitor.
6029. The method of claim 5894 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
6030. The method of claim 5894 wherein the agent is a CXCR3 inhibitor.
6031. The method of claim 5894 wherein the agent is an Itk inhibitor.
6032. The method of claim 5894 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
6033. The method of claim 5894 wherein the agent is a PPAR agonist.
6034. The method of claim 5894 wherein the agent is an immunosuppressant.
6035. The method of claim 5894 wherein the agent is an Erb inhibitor.
6036. The method of claim 5894 wherein the agent is an apoptosis agonist
6037. The method of claim 5894 wherein the agent is a lipocortin agonist
6038. The method of claim 5894 wherein the agent is a VCAM-1 antagonist.
6039. The method of claim 5894 wherein the agent is a collagen antagonist.
6040. The method of claim 5894 wherein the agent is an alpha 2 integrin antagonist.
6041. The method of claim 5894 wherein the agent is a TNF alpha inhibitor.
6042. The method of claim 5894 wherein the agent is a nitric oxide inhibitor
6043. The method of claim 5894 wherein the agent is a cathepsin inhibitor.
6044. The method of claim 5894 wherein the agent is not an anti- inflammatory agent.
6045. The method of claim 5894 wherein the agent is not a steroid.
6046. The method of claim 5894 wherein the agent is not a glucocorticosteroid.
6047. The method of claim 5894 wherein the agent is not dexamethasone.
6048. The method of claim 5894 wherein the agent is not beclomethasone.
6049. The method of claim 5894 wherein the agent is not dipropionate.
6050. The method of claim 5894 wherein the agent is not an anti- infective agent.
6051. The method of claim 5894 wherein the agent is not an antibiotic.
6052. The method of claim 5894 wherein the agent is not an anti- fungal agent.
6053. The method of claim 5894, wherein the composition comprises a polymer.
6054. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a copolymer.
6055. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a block copolymer.
6056. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a random copolymer.
6057. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a biodegradable polymer.
6058. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-biodegradable polymer.
6059. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophilic polymer.
6060. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophobic polymer.
6061. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophilic domains.
6062. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophobic domains.
6063. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-conductive polymer.
6064. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, an elastomer.
6065. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrogel.
6066. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a silicone polymer.
6067. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrocarbon polymer.
6068. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a styrene-derived polymer.
6069. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a butadiene-derived polymer.
6070. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a macromer.
,
6071. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, a poly(ethylene glycol) polymer.
6072. The method of claim 5894, wherein the composition comprises a polymer, and the polymer is, or comprises, an amorphous polymer.
6073. The method of claim 5894, wherein the composition further comprises a second pharmaceutically active agent.
6074. The method of claim 5894, wherein the composition further comprises an anti-inflammatory agent.
6075. The method of claim 5894, wherein the composition further comprises an agent that inhibits infection.
6076. The method of claim 5894, wherein the composition further comprises an anthracycline.
6077. The method of claim 5894, wherein the composition further comprises doxorubicin.
6078. The method of claim 5894 wherein the composition further comprises mitoxantrone.
6079. The method of claim 5894 wherein the composition further comprises a fluoropyrimidine.
6080. The method of claim 5894, wherein the composition further comprises 5-fluorouracil (5-FU).
6081. The method of claim 5894, wherein the composition further comprises a folic acid antagonist.
6082. The method of claim 5894, wherein the composition further comprises methotrexate.
6083. The method of claim 5894, wherein the composition further comprises a podophylotoxin.
6084. The method of claim 5894, wherein the composition further comprises etoposide.
6085. The method of claim 5894, wherein the composition further comprises camptothecin.
6086. The method of claim 5894, wherein the composition further comprises a hydroxyurea.
6087. The method of claim 5894, wherein the composition further comprises a platinum complex.
6088. The method of claim 5894, wherein the composition further comprises cisplatin.
6089. The method of claim 5894 wherein the composition further comprises an anti-thrombotic agent
6090. The method of claim 5894, wherein the composition further comprises a visualization agent.
6091. The method of claim 5894, wherein the composition further comprises a visualization agent, and the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
6092. The method of claim 5894, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, barium, tantalum, ortechnetium.
6093. The method of claim 5894, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, an MRI responsive material.
6094. The method of claim 5894, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a gadolinium chelate.
6095. The method of claim 5894, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron, magnesium, manganese, copper, or chromium.
6096. The method of claim 5894, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron oxide compound.
6097. The method of claim 5894, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a dye, pigment, or colorant.
6098. The method of claim 5894 wherein the agent is released in effective concentrations from the composition comprising the agent by diffusion over a period ranging from the time of administration to about 90 days.
6099. The method of claim 5894 wherein the agent is released in effective concentrations from the composition comprising the agent by erosion of the composition over a period ranging from the time of administration to about 90 days.
6100. The method of claim 5894 wherein the composition further comprises an inflammatory cytokine.
6101. The method of claim 5894 wherein the composition further comprises an agent that stimulates cell proliferation.
6102. The method of claim 5894 wherein the composition further comprises a polymeric carrier.
6103. The method of claim 5894 wherein the composition is in the form of a gel, paste, or spray.
6104. The method of claim 5894 wherein the pump is partially constructed with the agent or the composition.
6105. The method of claim 5894 wherein the pump is impregnated with the agent or the composition.
6106. The method of claim 5894, wherein the agent or the composition forms a coating, and the coating directly contacts the pump.
6107. The method of claim 5894, wherein the agent or the composition forms a coating, and the coating indirectly contacts the pump.
6108. The method of claim 5894 wherein the agent or the composition forms a coating, and the coating partially covers the pump.
6109. The method of claim 5894, wherein the agent or the composition forms a coating, and the coating completely covers the pump.
6110. The method of claim 5894 wherein the agent or the composition is located within pores or holes of the pump.
6111. The method of claim 5894 wherein the agent or the composition is located within a channel, lumen, or divet of the pump.
6112. The method of claim 5894 wherein the pump further comprises an echogenic material.
6113. The method of claim 5894 wherein the pump further comprises an echogenic material, wherein the echogenic material is in the form of a coating.
6114. The method of claim 5894 wherein the pump is sterile.
6115. The method of claim 5894 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump.
6116. The method of claim 5894 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is connective tissue.
6117. The method of claim 5894 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is muscle tissue.
6118. The method of claim 5894 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is nerve tissue.
6119. The method of claim 5894 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is epithelium tissue.
6120. The method of claim 5894 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from the time of deployment of the pump to about 1 year.
6121. The method of claim 5894 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from about 1 month to 6 months.
6122. The method of claim 5894 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from about 1 - 90 days.
6123. The method of claim 5894 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at a constant rate.
6124. The method of claim 5894 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at an increasing rate.
6125. The method of claim 5894 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at a decreasing rate.
6126. The method of claim 5894 wherein the agent is delivered from the pump, wherein the pump comprises about 0.01 μg to about 10 μg of the agent.
6127. The method of claim 5894 wherein the agent is delivered from the pump, wherein the pump comprises about 10 μg to about 10 mg of the agent.
6128. The method of claim 5894 wherein the agent is delivered from the pump, wherein the pump comprises about 10 mg to about 250 mg of the agent.
6129. The method of claim 5894 wherein the agent is delivered from the pump, wherein the pump comprises about 250 mg to about 1000 mg of the agent.
6130. The method of claim 5894 wherein the agent is delivered from the pump, wherein the pump comprises about 1000 mg to about 2500 mg of the agent.
6131. The method of claim 5894 wherein the agent is delivered from the pump, wherein a surface of the pump comprises less than 0.01 μg of the agent per mm2 of pump surface to which the agent is applied.
6132. The method of claim 5894 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 0.01 μg to about 1 μg of the agent per mm2 of pump surface to which the agent is applied.
6133. The method of claim 5894 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 1 μg to about 10 μg of the agent per mm2 of pump surface to which the agent is applied.
6134. The method of claim 5894 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 10 μg to about 250 μg of the agent per mm2 of pump surface to which the agent is applied.
6135. The method of claim 5894 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 250 μg to about 1000 μg of the agent per mm2 of pump surface to which the agent is applied.
6136. The method of claim 5894 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 1000 μg to about 2500 μg of the agent per mm2 of pump surface to which the agent is applied.
6137. The method of claim 5894, wherein the pump further comprises a coating, and the coating is a uniform coating.
6138. The method of claim 5894, wherein the pump further comprises a coating, and the coating is a non-uniform coating.
6139. The method of claim 5894, wherein the pump further comprises a coating, and the coating is a discontinuous coating.
6140. The method of claim 5894, wherein the pump further comprises a coating, and the coating is a patterned coating.
6141. The method of claim 5894, wherein the pump further comprises a coating, and the coating has a thickness of 100 μm or less.
6142. The method of claim 5894, wherein the pump further comprises a coating, and the coating has a thickness of 10 μm or less.
6143. The method of claim 5894, wherein the pump further comprises a coating, and the coating adheres to the surface of the pump upon deployment of the pump.
6144. The method of claim 5894, wherein the pump further comprises a coating, and the coating is stable at room temperature for a period of at least 1 year.
6145. The method of claim 5894, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 0.0001 % to about 1 % by weight.
6146. The method of claim 5894, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
6147. The method of claim 5894, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
6148. The method of claim 5894, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
6149. The method of claim 5894, wherein the pump further comprises a coating, and the coating comprises a polymer.
6150. The method of claim 5894, wherein the pump comprises a first coating having a first composition and a second coating having a second composition.
6151. The method of claim 5894, wherein the pump comprises a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
6152. The method of claim 5894 wherein the agent or the composition is affixed to the pump.
6153. The method of claim 5894 wherein the agent or the composition is covalently attached to the pump.
6154. The method of claim 5894 wherein the agent or the composition is non-covalently attached to the pump.
6155. The method of claim 5894 wherein the pump comprises a coating that absorbs the agent or the composition.
6156. The method of claim 5894 wherein the pump is interweaved with a thread composed of, or coated with, the agent or the composition.
6157. The method of claim 5894 wherein a portion of the pump is covered with a sleeve that contains the agent or the composition.
6158. The method of claim 5894 wherein the pump is completely covered with a sleeve that contains the agent or the composition.
6159. The method of claim 5894 wherein a portion of the pump is covered with a mesh that contains the agent or the composition.
6160. The method of claim 5894 wherein the pump is completely covered with a mesh that contains the agent or the composition.
6161. The method of claim 5894 wherein the pump is linked to a sensor.
6162. The method of claim 5894 wherein the agent or the composition is applied to the pump surface prior to to the placing of the pump into the host.
6163. The method of claim 5894 wherein the agent or the composition is applied to the pump surface during the placing of the pump into the host.
6164. The method of claim 5894 wherein the agent or the composition is applied to the pump surface immediately after the placing of the pump into the host
6165. The method of claim 5894 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump prior to to the placing of the pump into the host.
6166. The method of claim 5894 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump during the placing of the pump into the host.
6167. The method of claim 5894 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump immediately after the placing of the pump into the host.
6168. The method of claim 5894 wherein the agent or the composition is topically applied into the anatomical space where the pump is placed.
6169. The method of claim 5894 wherein the agent or the composition is percutaneously injected into the tissue in the host surrounding the pump.
6170. The method of any one of claims 5894-6169 further comprising placing a single channel catheter with a sensor into a vessel to transmit blood chemistry to the implantable insulin pump for dispensing insulin through the catheter.
6171. A method for inhibiting scarring comprising placing an intrathecal pump and an anti-scarring agent or a composition comprising an anti-scarring agent into an animal host, wherein the agent inhibits scarring.
6172. The method of claim 6171 wherein the agent inhibits cell regeneration.
6173. The method of claim 6171 wherein the agent inhibits angiogenesis.
6174. The method of claim 6171 wherein the agent inhibits fibroblast migration.
6175. The method of claim 6171 wherein the agent inhibits fibroblast proliferation.
6176. The method of claim 6171 wherein the agent inhibits deposition of extracellular matrix.
6177. The method of claim 6171 wherein the agent inhibits tissue remodeling.
6178. The method of claim 6171 wherein the agent is an angiogenesis inhibitor.
6179. The method of claim 6171 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
6180. The method of claim 6171 wherein the agent is a chemokine receptor antagonist.
6181. The method of claim 6171 wherein the agent is a cell cycle inhibitor.
6182. The method of claim 6171 wherein the agent is a taxane.
6183. The method of claim 6171 wherein the agent is an antimicrotubule agent.
6184. The method of claim 6171 wherein the agent is paclitaxel.
6185. The method of claim 6171 wherein the agent is not paclitaxel.
6186. The method of claim 6171 wherein the agent is an analogue or derivative of paclitaxel.
6187. The method of claim 6171 wherein the agent is a vinca alkaloid.
6188. The method of claim 6171 wherein the agent is camptothecin or an analogue or derivative thereof.
6189. The method of claim 6171 wherein the agent is a podophyllotoxin.
6190. The method of claim 6171 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
6191. The method of claim 6171 wherein the agent is an anthracycline.
6192. The method of claim 6171 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
6193. The method of claim 6171 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
6194. The method of claim 6171 wherein the agent is a platinum compound.
6195. The method of claim 6171 wherein the agent is a nitrosourea.
6196. The method of claim 6171 wherein the agent is a nitroimidazole.
6197. The method of claim 6171 wherein the agent is a folic acid antagonist.
6198. The method of claim 6171 wherein the agent is a cytidine analogue.
6199. The method of claim 6171 wherein the agent is a pyrimidine analogue.
6200. The method of claim 6171 wherein the agent is a fluoropyrimidine analogue.
6201. The method of claim 6171 wherein the agent is a purine analogue.
6202. The method of claim 6171 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
6203. The method of claim 6171 wherein the agent is a hydroxyurea.
6204. The method of claim 6171 wherein the agent is a mytomicin or an analogue or derivative thereof.
6205. The method of claim 6171 wherein the agent is an alkyl sulfonate.
6206. The method of claim 6171 wherein the agent is a benzamide or an analogue or derivative thereof.
6207. The method of claim 6171 wherein the agent is a nicotinamide or an analogue or derivative thereof.
6208. The method of claim 6171 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
6209. The method of claim 6171 wherein the agent is a DNA alkylating agent.
6210. The method of claim 6171 wherein the agent is an antimicrotubule agent.
6211. The method of claim 6171 wherein the agent is a topoisomerase inhibitor.
6212. The method of claim 6171 wherein the agent is a DNA cleaving agent.
6213. The method of claim 6171 wherein the agent is an antimetabolite.
6214. The method of claim 6171 wherein the agent inhibits adenosine deaminase.
6215. The method of claim 6171 wherein the agent inhibits purine ring synthesis.
6216. The method of claim 6171 wherein the agent is a nucleotide interconversion inhibitor.
6217. The method of claim 6171 wherein the agent inhibits dihydrofolate reduction.
6218. The method of claim 6171 wherein the agent blocks thymidine monophosphate.
6219. The method of claim 6171 wherein the agent causes DNA damage.
6220. The method of claim 6171 wherein the agent is a DNA intercalation agent.
6221. The method of claim 6171 wherein the agent is a RNA synthesis inhibitor.
6222. The method of claim 6171 wherein the agent is a pyrimidine synthesis inhibitor.
6223. The method of claim 6171 wherein the agent inhibits ribonucleotide synthesis or function.
6224. The method of claim 6171 wherein the agent inhibits thymidine monophosphate synthesis or function.
6225. The method of claim 6171 wherein the agent inhibits DNA synthesis.
6226. The method of claim 6171 wherein the agent causes DNA adduct formation.
6227. The method of claim 6171 wherein the agent inhibits protein synthesis.
6228. The method of claim 6171 wherein the agent inhibits microtubule function.
6229. The method of claim 6171 wherein the agent is a cyclin dependent protein kinase inhibitor.
6230. The method of claim 6171 wherein the agent is an epidermal growth factor kinase inhibitor.
6231. The method of claim 6171 wherein the agent is an elastase inhibitor.
6232. The method of claim 6171 wherein the agent is a factor Xa inhibitor.
6233. The method of claim 6171 wherein the agent is a farnesyltransferase inhibitor.
6234. The method of claim 6171 wherein the agent is a fibrinogen antagonist.
6235. The method of claim 6171 wherein the agent is a guanylate cyclase stimulant.
6236. The method of claim 6171 wherein the agent is a heat shock protein 90 antagonist.
6237. The method of claim 6171 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
6238. The method of claim 6171 wherein the agent is a guanylate cyclase stimulant.
6239. The method of claim 6171 wherein the agent is a HMGCoA reductase inhibitor.
6240. The method of claim 6171 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
6241. The method of claim 6171 wherein the agent is a hydroorotate dehydrogenase inhibitor.
6242. The method of claim 6171 wherein the agent is an IKK2 inhibitor.
6243. The method of claim 6171 wherein the agent is an IL-1 antagonist
6244. The method of claim 6171 wherein the agent is an ICE antagonist.
6245. The method of claim 6171 wherein the agent is an IRAK antagonist
6246. The method of claim 6171 wherein the agent is an IL-4 agonist.
6247. The method of claim 6171 wherein the agent is an immunomodulatory agent.
6248. The method of claim 6171 wherein the agent is sirolimus or an analogue or derivative thereof.
6249. The method of claim 6171 wherein the agent is not sirolimus.
6250. The method of claim 6171 wherein the agent is everolimus or an analogue or derivative thereof.
6251. The method of claim 6171 wherein the agent is tacrolimus or an analogue or derivative thereof.
6252. The method of claim 6171 wherein the agent is not tacrolimus.
6253. The method of claim 6171 wherein the agent is biolmus or an analogue or derivative thereof.
6254. The method of claim 6171 wherein the agent is tresperimus or an analogue or derivative thereof.
6255. The method of claim 6171 wherein the agent is auranofin or an analogue or derivative thereof.
6256. The method of claim 6171 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
6257. The method of claim 6171 wherein the agent is gusperimus or an analogue or derivative thereof.
6258. The method of claim 6171 wherein the agent is pimecrolimus or an analogue or derivative thereof.
6259. The method of claim 6171 wherein the agent is ABT-578 or an analogue or derivative thereof.
6260. The method of claim 6171 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
6261. The method of claim 6171 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
6262. The method of claim 6171 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
6263. The method of claim 6171 wherein the agent is a leukotriene inhibitor.
6264. The method of claim 6171 wherein the agent is a MCP-1 antagonist.
6265. The method of claim 6171 wherein the agent is a MMP inhibitor.
6266. The method of claim 6171 wherein the agent is an NF kappa B inhibitor.
6267. The method of claim 6171 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
6268. The method of claim 6171 wherein the agent is an NO antagonist.
6269. The method of claim 6171 wherein the agent is a p38 MAP kinase inhibitor.
6270. The method of claim 6171 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
6271. The method of claim 6171 wherein the agent is a phosphodiesterase inhibitor.
6272. The method of claim 6171 wherein the agent is a TGF beta inhibitor.
6273. The method of claim 6171 wherein the agent is a thromboxane A2 antagonist.
6274. The method of claim 6171 wherein the agent is a TNF alpha antagonist.
6275. The method of claim 6171 wherein the agent is a TACE inhibitor.
6276. The method of claim 6171 wherein the agent is a tyrosine kinase inhibitor.
6277. The method of claim 6171 wherein the agent is a vitronectin inhibitor.
6278. The method of claim 6171 wherein the agent is a fibroblast growth factor inhibitor.
6279. The method of claim 6171 wherein the agent is a protein kinase inhibitor.
6280. The method of claim 6171 wherein the agent is a PDGF receptor kinase inhibitor.
6281. The method of claim 6171 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
6282. The method of claim 6171 wherein the agent is a retinoic acid receptor antagonist.
6283. The method of claim 6171 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
6284. The method of claim 6171 wherein the agent is a fibrinogen antagonist.
6285. The method of claim 6171 wherein the agent is an antimycotic agent.
6286. The method of claim 6171 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
6287. The method of claim 6171 wherein the agent is a bisphosphonate.
6288. The method of claim 6171 wherein the agent is a phospholipase A1 inhibitor.
6289. The method of claim 6171 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
6290. The method of claim 6171 wherein the agent is a macrolide antibiotic.
6291. The method of claim 6171 wherein the agent is a GPIIb/llla receptor antagonist.
6292. The method of claim 6171 wherein the agent is an endothelin receptor antagonist.
6293. The method of claim 6171 wherein the agent is a peroxisome proliferator-activated receptor agonist.
6294. The method of claim 6171 wherein the agent is an estrogen receptor agent.
6295. The method of claim 6171 wherein the agent is a somastostatin analogue.
6296. The method of claim 6171 wherein the agent is a neurokinin 1 antagonist.
6297. The method of claim 6171 wherein the agent is a neurokinin 3 antagonist.
6298. The method of claim 6171 wherein the agent is a VLA-4 antagonist
6299. The method of claim 6171 wherein the agent is an osteoclast inhibitor.
6300. The method of claim 6171 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
6301. The method of claim 6171 wherein the agent is an angiotensin I converting enzyme inhibitor.
6302. The method of claim 6171 wherein the agent is an angiotensin II antagonist.
6303. The method of claim 6171 wherein the agent is an enkephalinase inhibitor.
6304. The method of claim 6171 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
6305. The method of claim 6171 wherein the agent is a protein kinase C inhibitor.
6306. The method of claim 6171 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
6307. The method of claim 6171 wherein the agent is a CXCR3 inhibitor.
6308. The method of claim 6171 wherein the agent is an Itk inhibitor.
6309. The method of claim 6171 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
6310. The method of claim 6171 wherein the agent is a PPAR agonist.
6311. The method of claim 6171 wherein the agent is an immunosuppressant
6312. The method of claim 6171 wherein the agent is an Erb inhibitor.
6313. The method of claim 6171 wherein the agent is an apoptosis agonist.
6314. The method of claim 6171 wherein the agent is a lipocortin agonist.
6315. The method of claim 6171 wherein the agent is a VCAM-1 antagonist.
6316. The method of claim 6171 wherein the agent is a collagen antagonist.
6317. The method of claim 6171 wherein the agent is an alpha 2 integrin antagonist.
6318. The method of claim 6171 wherein the agent is a TNF alpha inhibitor.
6319. The method of claim 6171 wherein the agent is a nitric oxide inhibitor
6320. The method of claim 6171 wherein the agent is a cathepsin inhibitor.
6321. The method of claim 6171 wherein the agent is not an anti- inflammatory agent.
6322. The method of claim 6171 wherein the agent is not a steroid.
6323. The method of claim 6171 wherein the agent is not a glucocorticosteroid.
6324. The method of claim 6171 wherein the agent is not dexamethasone. t
6325. The method of claim 6171 wherein the agent is not beclomethasone.
6326. The method of claim 6171 wherein the agent is not dipropionate.
6327. The method of claim 6171 wherein the agent is not an anti- infective agent.
6328. The method of claim 6171 wherein the agent is not an antibiotic.
6329. The method of claim 6171 wherein the agent is not an anti- fungal agent.
6330. The method of claim 6171 , wherein the composition comprises a polymer.
6331. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a copolymer.
6332. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a block copolymer.
6333. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a random copolymer.
6334. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a biodegradable polymer.
6335. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a non-biodegradable polymer.
6336. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophilic polymer.
6337. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophobic polymer.
6338. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophilic domains.
6339. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophobic domains.
6340. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a non-conductive polymer.
6341. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, an elastomer.
6342. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrogel.
6343. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a silicone polymer.
6344. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrocarbon polymer.
6345. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a styrene-derived polymer.
6346. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a butadiene-derived polymer.
6347. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a macromer.
6348. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, a poly(ethylene glycol) polymer.
6349. The method of claim 6171 , wherein the composition comprises a polymer, and the polymer is, or comprises, an amorphous polymer.
6350. The method of claim 6171 , wherein the composition further comprises a second pharmaceutically active agent.
6351. The method of claim 6171 , wherein the composition further comprises an anti-inflammatory agent.
6352. The method of claim 6171 , wherein the composition further comprises an agent that inhibits infection.
6353. The method of claim 6171 , wherein the composition further comprises an anthracycline.
6354. The method of claim 6171 , wherein the composition further comprises doxorubicin.
6355. The method of claim 6171 wherein the composition further comprises mitoxantrone.
6356. The method of claim 6171 wherein the composition further comprises a fluoropyrimidine.
6357. The method of claim 6171 , wherein the composition further comprises 5-fluorouracil (5-FU).
6358. The method of claim 6171 , wherein the composition further comprises a folic acid antagonist.
6359. The method of claim 6171 , wherein the composition further comprises methotrexate.
6360. The method of claim 6171 , wherein the composition further comprises a podophylotoxin.
6361. The method of claim 6171 , wherein the composition further comprises etoposide.
6362. The method of claim 6171 , wherein the composition further comprises camptothecin.
6363. The method of claim 6171 , wherein the composition further comprises a hydroxyurea.
6364. The method of claim 6171 , wherein the composition further comprises a platinum complex.
6365. The method of claim 6171 , wherein the composition further comprises cisplatin.
6366. The method of claim 6171 wherein the composition further comprises an anti-thrombotic agent.
6367. The method of claim 6171 , wherein the composition further comprises a visualization agent.
6368. The method of claim 6171 , wherein the composition further comprises a visualization agent, and the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
6369. The method of claim 6171 , wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, barium, tantalum, or technetium.
6370. The method of claim 6171 , wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, an MRI responsive material.
6371. The method of claim 6171 , wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a gadolinium chelate.
6372. The method of claim 6171 , wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron, magnesium, manganese, copper, or chromium.
6373. The method of claim 6171 , wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron oxide compound.
6374. The method of claim 6171 , wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a dye, pigment, or colorant.
6375. The method of claim 6171 wherein the agent is released in effective concentrations from the composition comprising the agent by diffusion over a period ranging from the time of administration to about 90 days.
6376. The method of claim 6171 wherein the agent is released in effective concentrations from the composition comprising the agent by erosion of the composition over a period ranging from the time of administration to about 90 days.
6377. The method of claim 6171 wherein the composition further comprises an inflammatory cytokine.
6378. The method of claim 6171 wherein the composition further comprises an agent that stimulates cell proliferation.
6379. The method of claim 6171 wherein the composition further comprises a polymeric carrier.
6380. The method of claim 6171 wherein the composition is in the form of a gel, paste, or spray.
6381. The method of claim 6171 wherein the pump is partially constructed with the agent or the composition.
6382. The method of claim 6171 wherein the pump is impregnated with the agent or the composition.
6383. The method of claim 6171 , wherein the agent or the composition forms a coating, and the coating directly contacts the pump.
6384. The method of claim 6171 , wherein the agent or the composition forms a coating, and the coating indirectly contacts the pump.
6385. The method of claim 6171 wherein the agent or the composition forms a coating, and the coating partially covers the pump.
6386. The method of claim 6171 , wherein the agent or the composition forms a coating, and the coating completely covers the pump.
6387. The method of claim 6171 wherein the agent or the composition is located within pores or holes of the pump.
6388. The method of claim 6171 wherein the agent or the composition is located within a channel, lumen, or divet of the pump.
6389. The method of claim 6171 wherein the pump further comprises an echogenic material.
6390. The method of claim 6171 wherein the pump further comprises an echogenic material, wherein the echogenic material is in the form of a coating.
6391. The method of claim 6171 wherein the pump is sterile.
6392. The method of claim 6171 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump.
6393. The method of claim 6171 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is connective tissue.
6394. The method of claim 6171 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is muscle tissue.
6395. The method of claim 6171 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is nerve tissue.
6396. The method of claim 6171 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is epithelium tissue.
6397. The method of claim 6171 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from the time of deployment of the pump to about 1 year.
6398. The method of claim 6171 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from about 1 month to 6 months.
6399. The method of claim 6171 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from about 1 - 90 days.
6400. The method of claim 6171 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at a constant rate.
6401. The method of claim 6171 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at an increasing rate.
6402. The method of claim 6171 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at a decreasing rate.
6403. The method of claim 6171 wherein the agent is delivered from the pump, wherein the pump comprises about 0.01 μg to about 10 μg of the agent
6404. The method of claim 6171 wherein the agent is delivered from the pump, wherein the pump comprises about 10 μg to about 10 mg of the agent.
6405. The method of claim 6171 wherein the agent is delivered from the pump, wherein the pump comprises about 10 mg to about 250 mg of the agent.
6406. The method of claim 6171 wherein the agent is delivered from the pump, wherein the pump comprises about 250 mg to about 1000 mg of the agent.
6407. The method of claim 6171 wherein the agent is delivered from the pump, wherein the pump comprises about 1000 mg to about 2500 mg of the agent.
6408. The method of claim 6171 wherein the agent is delivered from the pump, wherein a surface of the pump comprises less than 0.01 μg of the agent per mm2 of pump surface to which the agent is applied.
6409. The method of claim 6171 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 0.01 μg to about 1 μg of the agent per mm2 of pump surface to which the agent is applied.
6410. The method of claim 6171 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 1 μg to about 10 μg of the agent per mm2 of pump surface to which the agent is applied.
6411. The method of claim 6171 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 10 μg to about 250 μg of the agent per mm2 of pump surface to which the agent is applied.
6412. The method of claim 6171 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 250 μg to about 1000 μg of the agent per mm2 of pump surface to which the agent is applied.
6413. The method of claim 6171 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 1000 μg to about 2500 μg of the agent per mm2 of pump surface to which the agent is applied.
6414. The method of claim 6171 , wherein the pump further comprises a coating, and the coating is a uniform coating.
6415. The method of claim 6171 , wherein the pump further comprises a coating, and the coating is a non-uniform coating.
6416. The method of claim 6171 , wherein the pump further comprises a coating, and the coating is a discontinuous coating.
6417. The method of claim 6171 , wherein the pump further comprises a coating, and the coating is a patterned coating.
6418. The method of claim 6171 , wherein the pump further comprises a coating, and the coating has a thickness of 100 μm or less.
6419. The method of claim 6171 , wherein the pump further comprises a coating, and the coating has a thickness of 10 μm or less.
6420. The method of claim 6171 , wherein the pump further comprises a coating, and the coating adheres to the surface of the pump upon deployment of the pump.
6421. The method of claim 6171 , wherein the pump further comprises a coating, and the coating is stable at room temperature for a period of at least 1 year.
6422. The method of claim 6171 , wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 0.0001% to about 1 % by weight.
6423. The method of claim 6171 , wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
6424. The method of claim 6171 , wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
6425. The method of claim 6171 , wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
6426. The method of claim 6171 , wherein the pump further comprises a coating, and the coating comprises a polymer.
6427. The method of claim 6171 , wherein the pump comprises a first coating having a first composition and a second coating having a second composition.
6428. The method of claim 6171 , wherein the pump comprises a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
6429. The method of claim 6171 wherein the agent or the composition is affixed to the pump.
6430. The method of claim 6171 wherein the agent or the composition is covalently attached to the pump.
6431. The method of claim 6171 wherein the agent or the composition is non-covalently attached to the pump.
6432. The method of claim 6171 wherein the pump comprises a coating that absorbs the agent or the composition.
6433. The method of claim 6171 wherein the pump is interweaved with a thread composed of, or coated with, the agent or the composition.
6434. The method of claim 6171 wherein a portion of the pump is covered with a sleeve that contains the agent or the composition.
6435. The method of claim 6171 wherein the pump is completely covered with a sleeve that contains the agent or the composition.
6436. The method of claim 6171 wherein a portion of the pump is covered with a mesh that contains the agent or the composition.
6437. The method of claim 6171 wherein the pump is completely covered with a mesh that contains the agent or the composition.
6438. The method of claim 6171 wherein the pump is linked to a sensor.
6439. The method of claim 6171 wherein the agent or the composition is applied to the pump surface prior to to the placing of the pump into the host.
6440. The method of claim 6171 wherein the agent or the composition is applied to the pump surface during the placing of the pump into the host.
6441. The method of claim 6171 wherein the agent or the composition is applied to the pump surface immediately after the placing of the pump into the host.
6442. The method of claim 6171 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump prior to to the placing of the pump into the host.
6443. The method of claim 6171 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump during the placing of the pump into the host.
6444. The method of claim 6171 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump immediately after the placing of the pump into the host.
6445. The method of claim 6171 wherein the agent or the composition is topically applied into the anatomical space where the pump is placed.
6446. The method of claim 6171 wherein the agent or the composition is percutaneously injected into the tissue in the host surrounding the pump.
6447. The method of any one of claims 6171-6446 wherein the pump is adapted for delivering pain medication directly into the cerebrospinal fluid of the intrathecal space surrounding the spinal cord.
6448. The method of any one of claims 6171-6446 wherein the pump is adapted for delivering a drug to the brain.
6449. The method of any one of claims 6171-6446 wherein the pump is adapted for intrathecal delivering baclofen.
6450. The method of any one of claims 6171 -6446 wherein the pump further comprises an intraspinal catheter.
6451. The method of any one of claims 6171-6446 further comprising placing a second intrathecal drug delivery pump into the host.
6452. The method of any one of claims 6171-6446 wherein the pump further comprises a catheter and an electrode.
6453. A method for inhibiting scarring comprising placing an implantable drug delivery pump for chemotherapy and an anti-scarring agent or a composition comprising an anti-scarring agent into an animal host, wherein the agent inhibits scarring.
6454. The method of claim 6453 wherein the agent inhibits cell regeneration.
6455. The method of claim 6453 wherein the agent inhibits angiogenesis.
6456. The method of claim 6453 wherein the agent inhibits fibroblast migration.
6457. The method of claim 6453 wherein the agent inhibits fibroblast proliferation.
6458. The method of claim 6453 wherein the agent inhibits deposition of extracellular matrix.
6459. The method of claim 6453 wherein the agent inhibits tissue remodeling.
6460. The method of claim 6453 wherein the agent is an angiogenesis inhibitor.
6461. The method of claim 6453 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
6462. The method of claim 6453 wherein the agent is a chemokine receptor antagonist.
6463. The method of claim 6453 wherein the agent is a cell cycle inhibitor.
6464. The method of claim 6453 wherein the agent is a taxane.
6465. The method of claim 6453 wherein the agent is an antimicrotubule agent.
6466. The method of claim 6453 wherein the agent is paclitaxel.
6467. The method of claim 6453 wherein the agent is not paclitaxel.
6468. The method of claim 6453 wherein the agent is an analogue or derivative of paclitaxel.
6469. The method of claim 6453 wherein the agent is a vinca alkaloid.
6470. The method of claim 6453 wherein the agent is camptothecin or an analogue or derivative thereof.
6471. The method of claim 6453 wherein the agent is a podophyllotoxin.
6472. The method of claim 6453 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
6473. The method of claim 6453 wherein the agent is an anthracycline.
6474. The method of claim 6453 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
6475. The method of claim 6453 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
6476. The method of claim 6453 wherein the agent is a platinum compound.
6477. The method of claim 6453 wherein the agent is a nitrosourea.
6478. The method of claim 6453 wherein the agent is a nitroimidazole.
6479. The method of claim 6453 wherein the agent is a folic acid antagonist.
6480. The method of claim 6453 wherein the agent is a cytidine analogue.
6481. The method of claim 6453 wherein the agent is a pyrimidine analogue.
6482. The method of claim 6453 wherein the agent is a fluoropyrimidine analogue.
6483. The method of claim 6453 wherein the agent is a purine analogue.
6484. The method of claim 6453 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
6485. The method of claim 6453 wherein the agent is a hydroxyurea.
6486. The method of claim 6453 wherein the agent is a mytomicin or an analogue or derivative thereof.
6487. The method of claim 6453 wherein the agent is an alkyl sulfonate.
6488. The method of claim 6453 wherein the agent is a benzamide or an analogue or derivative thereof.
6489. The method of claim 6453 wherein the agent is a nicotinamide or an analogue or derivative thereof.
6490. The method of claim 6453 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
6491. The method of claim 6453 wherein the agent is a DNA alkylating agent.
6492. The method of claim 6453 wherein the agent is an antimicrotubule agent
6493. The method of claim 6453 wherein the agent is a topoisomerase inhibitor.
6494. The method of claim 6453 wherein the agent is a DNA cleaving agent.
6495. The method of claim 6453 wherein the agent is an antimetabolite.
6496. The method of claim 6453 wherein the agent inhibits adenosine deaminase.
6497. The method of claim 6453 wherein the agent inhibits purine ring synthesis.
6498. The method of claim 6453 wherein the agent is a nucleotide interconversion inhibitor.
6499. The method of claim 6453 wherein the agent inhibits dihydrofolate reduction.
6500. The method of claim 6453 wherein the agent blocks thymidine monophosphate.
6501. The method of claim 6453 wherein the agent causes DNA damage.
6502. The method of claim 6453 wherein the agent is a DNA intercalation agent.
6503. The method of claim 6453 wherein the agent is a RNA synthesis inhibitor.
6504. The method of claim 6453 wherein the agent is a pyrimidine synthesis inhibitor.
6505. The method of claim 6453 wherein the agent inhibits ribonucleotide synthesis or function.
6506. The method of claim 6453 wherein the agent inhibits thymidine monophosphate synthesis or function.
6507. The method of claim 6453 wherein the agent inhibits DNA synthesis.
6508. The method of claim 6453 wherein the agent causes DNA adduct formation.
6509. The method of claim 6453 wherein the agent inhibits protein synthesis.
6510. The method of claim 6453 wherein the agent inhibits microtubule function.
6511. The method of claim 6453 wherein the agent is a cyclin dependent protein kinase inhibitor.
6512. The method of claim 6453 wherein the agent is an epidermal growth factor kinase inhibitor.
6513. The method of claim 6453 wherein the agent is an elastase inhibitor.
6514. The method of claim 6453 wherein the agent is a factor Xa inhibitor.
6515. The method of claim 6453 wherein the agent is a farnesyltransferase inhibitor.
6516. The method of claim 6453 wherein the agent is a fibrinogen antagonist.
6517. The method of claim 6453 wherein the agent is a guanylate cyclase stimulant.
6518. The method of claim 6453 wherein the agent is a heat shock protein 90 antagonist.
6519. The method of claim 6453 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
6520. The method of claim 6453 wherein the agent is a guanylate cyclase stimulant.
6521. The method of claim 6453 wherein the agent is a HMGCoA reductase inhibitor.
6522. The method of claim 6453 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
6523. The method of claim 6453 wherein the agent is a hydroorotate dehydrogenase inhibitor.
6524. The method of claim 6453 wherein the agent is an IKK2 inhibitor.
6525. The method of claim 6453 wherein the agent is an IL-1 antagonist.
6526. The method of claim 6453 wherein the agent is an ICE antagonist
6527. The method of claim 6453 wherein the agent is an IRAK antagonist.
6528. The method of claim 6453 wherein the agent is an IL-4 agonist.
6529. The method of claim 6453 wherein the agent is an immunomodulatory agent
6530. The method of claim 6453 wherein the agent is sirolimus or an analogue or derivative thereof.
6531. The method of claim 6453 wherein the agent is not sirolimus.
6532. The method of claim 6453 wherein the agent is everolimus or an analogue or derivative thereof.
6533. The method of claim 6453 wherein the agent is tacrolimus or an analogue or derivative thereof.
6534. The method of claim 6453 wherein the agent is not tacrolimus.
6535. The method of claim 6453 wherein the agent is biolmus or an analogue or derivative thereof.
6536. The method of claim 6453 wherein the agent is tresperimus or an analogue or derivative thereof.
6537. The method of claim 6453 wherein the agent is auranofin or an analogue or derivative thereof.
6538. The method of claim 6453 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
6539. The method of claim 6453 wherein the agent is gusperimus or an analogue or derivative thereof.
6540. The method of claim 6453 wherein the agent is pimecrolimus or an analogue or derivative thereof.
6541. The method of claim 6453 wherein the agent is ABT-578 or an analogue or derivative thereof.
6542. The method of claim 6453 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
6543. The method of claim 6453 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
6544. The method of claim 6453 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
6545. The method of claim 6453 wherein the agent is a leukotriene inhibitor.
6546. The method of claim 6453 wherein the agent is a MCP-1 antagonist
6547. The method of claim 6453 wherein the agent is a MMP inhibitor.
6548. The method of claim 6453 wherein the agent is an NF kappa B inhibitor.
6549. The method of claim 6453 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
6550. The method of claim 6453 wherein the agent is an NO antagonist.
6551. The method of claim 6453 wherein the agent is a p38 MAP kinase inhibitor.
6552. The method of claim 6453 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
6553. The method of claim 6453 wherein the agent is a phosphodiesterase inhibitor.
6554. The method of claim 6453 wherein the agent is a TGF beta inhibitor.
6555. The method of claim 6453 wherein the agent is a thromboxane A2 antagonist.
6556. The method of claim 6453 wherein the agent is a TNF alpha antagonist.
6557. The method of claim 6453 wherein the agent is a TACE inhibitor.
6558. The method of claim 6453 wherein the agent is a tyrosine kinase inhibitor.
6559. The method of claim 6453 wherein the agent is a vitronectin inhibitor.
6560. The method of claim 6453 wherein the agent is a fibroblast growth factor inhibitor.
6561. The method of claim 6453 wherein the agent is a protein kinase inhibitor.
6562. The method of claim 6453 wherein the agent is a PDGF receptor kinase inhibitor.
6563. The method of claim 6453 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
6564. The method of claim 6453 wherein the agent is a retinoic acid receptor antagonist.
6565. The method of claim 6453 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
6566. The method of claim 6453 wherein the agent is a fibrinogen antagonist.
6567. The method of claim 6453 wherein the agent is an antimycotic agent.
6568. The method of claim 6453 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
6569. The method of claim 6453 wherein the agent is a bisphosphonate.
6570. The method of claim 6453 wherein the agent is a phospholipase A1 inhibitor.
6571. The method of claim 6453 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
6572. The method of claim 6453 wherein the agent is a macrolide antibiotic.
6573. The method of claim 6453 wherein the agent is a GPIIb/llla receptor antagonist.
6574. The method of claim 6453 wherein the agent is an endothelin receptor antagonist.
6575. The method of claim 6453 wherein the agent is a peroxisome proliferator-activated receptor agonist.
6576. The method of claim 6453 wherein the agent is an estrogen receptor agent.
6577. The method of claim 6453 wherein the agent is a somastostatin analogue.
6578. The method of claim 6453 wherein the agent is a neurokinin 1 antagonist.
6579. The method of claim 6453 wherein the agent is a neurokinin 3 antagonist.
6580. The method of claim 6453 wherein the agent is a VLA-4 antagonist
6581. The method of claim 6453 wherein the agent is an osteoclast inhibitor.
6582. The method of claim 6453 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
6583. The method of claim 6453 wherein the agent is an angiotensin I converting enzyme inhibitor.
6584. The method of claim 6453 wherein the agent is an angiotensin II antagonist.
6585. The method of claim 6453 wherein the agent is an enkephalinase inhibitor.
6586. The method of claim 6453 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
6587. The method of claim 6453 wherein the agent is a protein kinase C inhibitor.
6588. The method of claim 6453 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
6589. The method of claim 6453 wherein the agent is a CXCR3 inhibitor.
6590. The method of claim 6453 wherein the agent is an Itk inhibitor.
6591. The method of claim 6453 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
6592. The method of claim 6453 wherein the agent is a PPAR agonist.
6593. The method of claim 6453 wherein the agent is an immunosuppressant
6594. The method of claim 6453 wherein the agent is an Erb inhibitor.
6595. The method of claim 6453 wherein the agent is an apoptosis agonist.
6596. The method of claim 6453 wherein the agent is a lipocortin agonist.
6597. The method of claim 6453 wherein the agent is a VCAM-1 antagonist
6598. The method of claim 6453 wherein the agent is a collagen antagonist
6599. The method of claim 6453 wherein the agent is an alpha 2 integrin antagonist.
6600. The method of claim 6453 wherein the agent is a TNF alpha inhibitor.
6601. The method of claim 6453 wherein the agent is a nitric oxide inhibitor
6602. The method of claim 6453 wherein the agent is a cathepsin inhibitor.
6603. The method of claim 6453 wherein the agent is not an anti- inflammatory agent.
6604. The method of claim 6453 wherein the agent is not a steroid.
6605. The method of claim 6453 wherein the agent is not a glucocorticosteroid.
6606. The method of claim 6453 wherein the agent is not dexamethasone.
6607. The method of claim 6453 wherein the agent is not beclomethasone.
6608. The method of claim 6453 wherein the agent is not dipropionate.
6609. The method of claim 6453 wherein the agent is not an anti- infective agent.
6610. The method of claim 6453 wherein the agent is not an antibiotic.
6611. The method of claim 6453 wherein the agent is not an anti- fungal agent.
6612. The method of claim 6453, wherein the composition comprises a polymer.
6613. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a copolymer.
6614. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a block copolymer.
6615. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a random copolymer.
6616. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a biodegradable polymer.
6617. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-biodegradable polymer.
6618. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophilic polymer.
6619. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophobic polymer.
6620. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophilic domains.
6621. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophobic domains.
6622. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-conductive polymer.
6623. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, an elastomer.
6624. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrogel.
6625. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a silicone polymer.
6626. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrocarbon polymer.
6627. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a styrene-derived polymer.
6628. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a butadiene-derived polymer.
6629. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a macromer.
6630. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, a poly(ethylene glycol) polymer.
6631. The method of claim 6453, wherein the composition comprises a polymer, and the polymer is, or comprises, an amorphous polymer.
6632. The method of claim 6453, wherein the composition further comprises a second pharmaceutically active agent.
6633. The method of claim 6453, wherein the composition further comprises an anti-inflammatory agent
6634. The method of claim 6453, wherein the composition further comprises an agent that inhibits infection.
6635. The method of claim 6453, wherein the composition further comprises an anthracycline.
6636. The method of claim 6453, wherein the composition further comprises doxorubicin.
6637. The method of claim 6453 wherein the composition further comprises mitoxantrone.
6638. The method of claim 6453 wherein the composition further comprises a fluoropyrimidine.
6639. The method of claim 6453, wherein the composition further comprises 5-fluorouracil (5-FU).
6640. The method of claim 6453, wherein the composition further comprises a folic acid antagonist.
6641. The method of claim 6453, wherein the composition further comprises methotrexate.
6642. The method of claim 6453, wherein the composition further comprises a podophylotoxin.
6643. The method of claim 6453, wherein the composition further comprises etoposide.
6644. The method of claim 6453, wherein the composition further comprises camptothecin.
6645. The method of claim 6453, wherein the composition further comprises a hydroxyurea.
6646. The method of claim 6453, wherein the composition further comprises a platinum complex.
6647. The method of claim 6453, wherein the composition further comprises cisplatin.
6648. The method of claim 6453 wherein the composition further comprises an anti-thrombotic agent.
6649. The method of claim 6453, wherein the composition further comprises a visualization agent.
6650. The method of claim 6453, wherein the composition further comprises a visualization agent, and the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
6651. The method of claim 6453, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, barium, tantalum, or technetium.
6652. The method of claim 6453, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, an MRI responsive material.
6653. The method of claim 6453, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a gadolinium chelate.
6654. The method of claim 6453, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron, magnesium, manganese, copper, or chromium.
6655. The method of claim 6453, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron oxide compound.
6656. The method of claim 6453, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a dye, pigment, or colorant.
6657. The method of claim 6453 wherein the agent is released in effective concentrations from the composition comprising the agent by diffusion over a period ranging from the time of administration to about 90 days.
6658. The method of claim 6453 wherein the agent is released in effective concentrations from the composition comprising the agent by erosion of the composition over a period ranging from the time of administration to about 90 days.
6659. The method of claim 6453 wherein the composition further comprises an inflammatory cytokine.
6660. The method of claim 6453 wherein the composition further comprises an agent that stimulates cell proliferation.
6661. The method of claim 6453 wherein the composition further comprises a polymeric carrier.
6662. The method of claim 6453 wherein the composition is in the form of a gel, paste, or spray.
6663. The method of claim 6453 wherein the pump is partially constructed with the agent or the composition.
6664. The method of claim 6453 wherein the pump is impregnated with the agent or the composition.
6665. The method of claim 6453, wherein the agent or the composition forms a coating, and the coating directly contacts the pump.
6666. The method of claim 6453, wherein the agent or the composition forms a coating, and the coating indirectly contacts the pump.
6667. The method of claim 6453 wherein the agent or the composition forms a coating, and the coating partially covers the pump.
6668. The method of claim 6453, wherein the agent or the composition forms a coating, and the coating completely covers the pump.
6669. The method of claim 6453 wherein the agent or the composition is located within pores or holes of the pump.
6670. The method of claim 6453 wherein the agent or the composition is located within a channel, lumen, or divet of the pump.
6671. The method of claim 6453 wherein the pump further comprises an echogenic material.
6672. The method of claim 6453 wherein the pump further comprises an echogenic material, wherein the echogenic material is in the form of a coating.
6673. The method of claim 6453 wherein the pump is sterile.
6674. The method of claim 6453 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump.
6675. The method of claim 6453 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is connective tissue.
6676. The method of claim 6453 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is muscle tissue.
6677. The method of claim 6453 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is nerve tissue.
6678. The method of claim 6453 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is epithelium tissue.
6679. The method of claim 6453 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from the time of deployment of the pump to about 1 year.
6680. The method of claim 6453 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from about 1 month to 6 months.
6681. The method of claim 6453 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from about 1 - 90 days.
6682. The method of claim 6453 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at a constant rate.
6683. The method of claim 6453 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at an increasing rate.
6684. The method of claim 6453 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at a decreasing rate.
6685. The method of claim 6453 wherein the agent is delivered from the pump, wherein the pump comprises about 0.01 μg to about 10 μg of the agent.
6686. The method of claim 6453 wherein the agent is delivered from the pump, wherein the pump comprises about 10 μg to about 10 mg of the agent.
6687. The method of claim 6453 wherein the agent is delivered from the pump, wherein the pump comprises about 10 mg to about 250 mg of the agent.
6688. The method of claim 6453 wherein the agent is delivered from the pump, wherein the pump comprises about 250 mg to about 1000 mg of the agent.
6689. The method of claim 6453 wherein the agent is delivered from the pump, wherein the pump comprises about 1000 mg to about 2500 mg of the agent.
6690. The method of claim 6453 wherein the agent is delivered from the pump, wherein a surface of the pump comprises less than 0.01 μg of the agent per mm2 of pump surface to which the agent is applied.
6691. The method of claim 6453 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 0.01 μg to about 1 μg of the agent per mm2 of pump surface to which the agent is applied.
6692. The method of claim 6453 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 1 μg to about 10 μg of the agent per mm2 of pump surface to which the agent is applied.
6693. The method of claim 6453 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 10 μg to about 250 μg of the agent per mm2 of pump surface to which the agent is applied.
6694. The method of claim 6453 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 250 μg to about 1000 μg of the agent per mm2 of pump surface to which the agent is applied.
6695. The method of claim 6453 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 1000 μg to about 2500 μg of the agent per mm2 of pump surface to which the agent is applied.
6696. The method of claim 6453, wherein the pump further comprises a coating, and the coating is a uniform coating.
6697. The method of claim 6453, wherein the pump further comprises a coating, and the coating is a non-uniform coating.
6698. The method of claim 6453, wherein the pump further comprises a coating, and the coating is a discontinuous coating.
6699. The method of claim 6453, wherein the pump further comprises a coating, and the coating is a patterned coating.
6700. The method of claim 6453, wherein the pump further comprises a coating, and the coating has a thickness of 100 μm or less.
6701. The method of claim 6453, wherein the pump further comprises a coating, and the coating has a thickness of 10 μm or less.
6702. The method of claim 6453, wherein the pump further comprises a coating, and the coating adheres to the surface of the pump upon deployment of the pump.
6703. The method of claim 6453, wherein the pump further comprises a coating, and the coating is stable at room temperature for a period of at least 1 year.
6704. The method of claim 6453, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 0.0001% to about 1 % by weight.
6705. The method of claim 6453, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
6706. The method of claim 6453, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
6707. The method of claim 6453, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 25% to about 70% by weight
6708. The method of claim 6453, wherein the pump further comprises a coating, and the coating comprises a polymer.
6709. The method of claim 6453, wherein the pump comprises a first coating having a first composition and a second coating having a second composition.
6710. The method of claim 6453, wherein the pump comprises a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
6711. The method of claim 6453 wherein the agent or the composition is affixed to the pump.
6712. The method of claim 6453 wherein the agent or the composition is covalently attached to the pump.
6713. The method of claim 6453 wherein the agent or the composition is non-covalently attached to the pump.
6714. The method of claim 6453 wherein the pump comprises a coating that absorbs the agent or the composition.
6715. The method of claim 6453 wherein the pump is interweaved with a thread composed of, or coated with, the agent or the composition.
6716. The method of claim 6453 wherein a portion of the pump is covered with a sleeve that contains the agent or the composition.
6717. The method of claim 6453 wherein the pump is completely covered with a sleeve that contains the agent or the composition.
6718. The method of claim 6453 wherein a portion of the pump is covered with a mesh that contains the agent or the composition.
6719. The method of claim 6453 wherein the pump is completely covered with a mesh that contains the agent or the composition.
6720. The method of claim 6453 wherein the pump is linked to a sensor.
6721. The method of claim 6453 wherein the agent or the composition is applied to the pump surface prior to to the placing of the pump into the host.
6722. The method of claim 6453 wherein the agent or the composition is applied to the pump surface during the placing of the pump into the host.
6723. The method of claim 6453 wherein the agent or the composition is applied to the pump surface immediately after the placing of the pump into the host.
6724. The method of claim 6453 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump prior to to the placing of the pump into the host
6725. The method of claim 6453 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump during the placing of the pump into the host.
6726. The method of claim 6453 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump immediately after the placing of the pump into the host
6727. The method of claim 6453 wherein the agent or the composition is topically applied into the anatomical space where the pump is placed.
6728. The method of claim 6453 wherein the agent or the composition is percutaneously injected into the tissue in the host surrounding the pump.
6729. The method of any one of claims 6453-6728 wherein the pump is adapted for delivering 2'-deoxy 5-fluorouridine.
6730. The method of any one of claims 6453-6728 wherein the host has a solid tumor, and the pump is adapted for infusing a chemotherapeutic agent to the solid tumor.
6731. The method of any one of claims 6453-6728 wherein the host has a tumor, and the pump is adapted for infusing a chemotherapeutic agent to the blood vessels that supply the tumor.
6732. The method of any one of claims 6453-6728 wherein the host has a hepatic tumor, and the pump is adapted for delivering a chemotherapeutic agent to the artery that provides blood supply to the liver of the host.
6733. A method for inhibiting scarring comprising placing a drug delivery pump for treating heart disease and an anti-scarring agent or a composition comprising an anti-scarring agent into an animal host, wherein the agent inhibits scarring.
6734. The method of claim 6733 wherein the agent inhibits cell regeneration.
6735. The method of claim 6733 wherein the agent inhibits angiogenesis.
6736. The method of claim 6733 wherein the agent inhibits fibroblast migration.
6737. The method of claim 6733 wherein the agent inhibits fibroblast proliferation.
6738. The method of claim 6733 wherein the agent inhibits deposition of extracellular matrix.
6739. The method of claim 6733 wherein the agent inhibits tissue remodeling.
6740. The method of claim 6733 wherein the agent is an angiogenesis inhibitor.
6741. The method of claim 6733 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
6742. The method of claim 6733 wherein the agent is a chemokine receptor antagonist
6743. The method of claim 6733 wherein the agent is a cell cycle inhibitor.
6744. The method of claim 6733 wherein the agent is a taxane.
6745. The method of claim 6733 wherein the agent is an antimicrotubule agent.
6746. The method of claim 6733 wherein the agent is paclitaxel.
6747. The method of claim 6733 wherein the agent is not paclitaxel.
6748. The method of claim 6733 wherein the agent is an analogue or derivative of paclitaxel.
6749. The method of claim 6733 wherein the agent is a vinca alkaloid.
6750. The method of claim 6733 wherein the agent is camptothecin or an analogue or derivative thereof.
6751. The method of claim 6733 wherein the agent is a podophyllotoxin.
6752. The method of claim 6733 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
6753. The method of claim 6733 wherein the agent is an anthracycline.
6754. The method of claim 6733 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
6755. The method of claim 6733 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
6756. The method of claim 6733 wherein the agent is a platinum compound.
6757. The method of claim 6733 wherein the agent is a nitrosourea.
6758. The method of claim 6733 wherein the agent is a nitroimidazole.
6759. The method of claim 6733 wherein the agent is a folic acid antagonist.
6760. The method of claim 6733 wherein the agent is a cytidine analogue.
6761. The method of claim 6733 wherein the agent is a pyrimidine analogue.
6762. The method of claim 6733 wherein the agent is a fluoropyrimidine analogue.
6763. The method of claim 6733 wherein the agent is a purine analogue.
6764. The method of claim 6733 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
6765. The method of claim 6733 wherein the agent is a hydroxyurea.
6766. The method of claim 6733 wherein the agent is a mytomicin or an analogue or derivative thereof.
6767. The method of claim 6733 wherein the agent is an alkyl sulfonate.
6768. The method of claim 6733 wherein the agent is a benzamide or an analogue or derivative thereof.
6769. The method of claim 6733 wherein the agent is a nicotinamide or an analogue or derivative thereof.
6770. The method of claim 6733 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
6771. The method of claim 6733 wherein the agent is a DNA alkylating agent.
6772. The method of claim 6733 wherein the agent is an antimicrotubule agent.
6773. The method of claim 6733 wherein the agent is a topoisomerase inhibitor.
6774. The method of claim 6733 wherein the agent is a DNA cleaving agent.
6775. The method of claim 6733 wherein the agent is an antimetabolite.
6776. The method of claim 6733 wherein the agent inhibits adenosine deaminase.
6777. The method of claim 6733 wherein the agent inhibits purine ring synthesis.
6778. The method of claim 6733 wherein the agent is a nucleotide interconversion inhibitor.
6779. The method of claim 6733 wherein the agent inhibits dihydrofolate reduction.
6780. The method of claim 6733 wherein the agent blocks thymidine monophosphate.
6781. The method of claim 6733 wherein the agent causes DNA damage.
6782. The method of claim 6733 wherein the agent is a DNA intercalation agent.
6783. The method of claim 6733 wherein the agent is a RNA synthesis inhibitor.
6784. The method of claim 6733 wherein the agent is a pyrimidine synthesis inhibitor.
6785. The method of claim 6733 wherein the agent inhibits ribonucleotide synthesis or function.
6786. The method of claim 6733 wherein the agent inhibits thymidine monophosphate synthesis or function.
6787. The method of claim 6733 wherein the agent inhibits DNA synthesis.
6788. The method of claim 6733 wherein the agent causes DNA adduct formation.
6789. The method of claim 6733 wherein the agent inhibits protein synthesis.
6790. The method of claim 6733 wherein the agent inhibits microtubule function.
6791. The method of claim 6733 wherein the agent is a cyclin dependent protein kinase inhibitor.
6792. The method of claim 6733 wherein the agent is an epidermal growth factor kinase inhibitor.
6793. The method of claim 6733 wherein the agent is an elastase inhibitor.
6794. The method of claim 6733 wherein the agent is a factor Xa inhibitor.
6795. The method of claim 6733 wherein the agent is a farnesyltransferase inhibitor.
6796. The method of claim 6733 wherein the agent is a fibrinogen antagonist.
6797. The method of claim 6733 wherein the agent is a guanylate cyclase stimulant.
6798. The method of claim 6733 wherein the agent is a heat shock protein 90 antagonist.
6799. The method of claim 6733 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
6800. The method of claim 6733 wherein the agent is a guanylate cyclase stimulant.
6801. The method of claim 6733 wherein the agent is a HMGCoA reductase inhibitor.
6802. The method of claim 6733 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
6803. The method of claim 6733 wherein the agent is a hydroorotate dehydrogenase inhibitor.
6804. The method of claim 6733 wherein the agent is an IKK2 inhibitor.
6805. The method of claim 6733 wherein the agent is an IL-1 antagonist.
6806. The method of claim 6733 wherein the agent is an ICE antagonist.
6807. The method of claim 6733 wherein the agent is an IRAK antagonist.
6808. The method of claim 6733 wherein the agent is an IL-4 agonist.
6809. The method of claim 6733 wherein the agent is an immunomodulatory agent.
6810. The method of claim 6733 wherein the agent is sirolimus or an analogue or derivative thereof.
6811. The method of claim 6733 wherein the agent is not sirolimus.
6812. The method of claim 6733 wherein the agent is everolimus or an analogue or derivative thereof.
6813. The method of claim 6733 wherein the agent is tacrolimus or an analogue or derivative thereof.
6814. The method of claim 6733 wherein the agent is not tacrolimus.
6815. The method of claim 6733 wherein the agent is biolmus or an analogue or derivative thereof.
6816. The method of claim 6733 wherein the agent is tresperimus or an analogue or derivative thereof.
6817. The method of claim 6733 wherein the agent is auranofin or an analogue or derivative thereof.
6818. The method of claim 6733 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
6819. The method of claim 6733 wherein the agent is gusperimus or an analogue or derivative thereof.
6820. The method of claim 6733 wherein the agent is pimecrolimus or an analogue or derivative thereof.
6821. The method of claim 6733 wherein the agent is ABT-578 or an analogue or derivative thereof.
6822. The method of claim 6733 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
6823. The method of claim 6733 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
6824. The method of claim 6733 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
6825. The method of claim 6733 wherein the agent is a leukotriene inhibitor.
6826. The method of claim 6733 wherein the agent is a MCP-1 antagonist.
6827. The method of claim 6733 wherein the agent is a MMP inhibitor.
6828. The method of claim 6733 wherein the agent is an NF kappa B inhibitor.
6829. The method of claim 6733 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
6830. The method of claim 6733 wherein the agent is an NO antagonist.
6831. The method of claim 6733 wherein the agent is a p38 MAP kinase inhibitor.
6832. The method of claim 6733 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
6833. The method of claim 6733 wherein the agent is a phosphodiesterase inhibitor.
6834. The method of claim 6733 wherein the agent is a TGF beta inhibitor.
6835. The method of claim 6733 wherein the agent is a thromboxane A2 antagonist.
6836. The method of claim 6733 wherein the agent is a TNF alpha antagonist.
6837. The method of claim 6733 wherein the agent is a TACE inhibitor.
6838. The method of claim 6733 wherein the agent is a tyrosine kinase inhibitor.
6839. The method of claim 6733 wherein the agent is a vitronectin inhibitor.
6840. The method of claim 6733 wherein the agent is a fibroblast growth factor inhibitor.
6841. The method of claim 6733 wherein the agent is a protein kinase inhibitor.
6842. The method of claim 6733 wherein the agent is a PDGF receptor kinase inhibitor.
6843. The method of claim 6733 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
6844. The method of claim 6733 wherein the agent is a retinoic acid receptor antagonist.
6845. The method of claim 6733 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
6846. The method of claim 6733 wherein the agent is a fibrinogen antagonist.
6847. The method of claim 6733 wherein the agent is an antimycotic agent.
6848. The method of claim 6733 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
6849. The method of claim 6733 wherein the agent is a bisphosphonate.
6850. The method of claim 6733 wherein the agent is a phospholipase A1 inhibitor.
6851. The method of claim 6733 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
6852. The method of claim 6733 wherein the agent is a macrolide antibiotic.
6853. The method of claim 6733 wherein the agent is a GPIIb/llla receptor antagonist.
6854. The method of claim 6733 wherein the agent is an endothelin receptor antagonist.
6855. The method of claim 6733 wherein the agent is a peroxisome proliferator-activated receptor agonist.
6856. The method of claim 6733 wherein the agent is an estrogen receptor agent.
6857. The method of claim 6733 wherein the agent is a somastostatin analogue.
6858. The method of claim 6733 wherein the agent is a neurokinin 1 antagonist.
6859. The method of claim 6733 wherein the agent is a neurokinin 3 antagonist.
6860. The method of claim 6733 wherein the agent is a VLA-4 antagonist
6861. The method of claim 6733 wherein the agent is an osteoclast inhibitor.
6862. The method of claim 6733 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
6863. The method of claim 6733 wherein the agent is an angiotensin I converting enzyme inhibitor.
6864. The method of claim 6733 wherein the agent is an angiotensin II antagonist.
6865. The method of claim 6733 wherein the agent is an enkephalinase inhibitor.
6866. The method of claim 6733 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
6867. The method of claim 6733 wherein the agent is a protein kinase C inhibitor.
6868. The method of claim 6733 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
6869. The method of claim 6733 wherein the agent is a CXCR3 inhibitor.
6870. The method of claim 6733 wherein the agent is an Itk inhibitor.
6871. The method of claim 6733 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
6872. The method of claim 6733 wherein the agent is a PPAR agonist.
6873. The method of claim 6733 wherein the agent is an immunosuppressant.
6874. The method of claim 6733 wherein the agent is an Erb inhibitor.
6875. The method of claim 6733 wherein the agent is an apoptosis agonist.
6876. The method of claim 6733 wherein the agent is a lipocortin agonist.
6877. The method of claim 6733 wherein the agent is a VCAM-1 antagonist.
6878. The method of claim 6733 wherein the agent is a collagen antagonist.
6879. The method of claim 6733 wherein the agent is an alpha 2 integrin antagonist.
6880. The method of claim 6733 wherein the agent is a TNF alpha inhibitor.
6881. The method of claim 6733 wherein the agent is a nitric oxide inhibitor
6882. The method of claim 6733 wherein the agent is a cathepsin inhibitor.
6883. The method of claim 6733 wherein the agent is not an anti- inflammatory agent.
6884. The method of claim 6733 wherein the agent is not a steroid.
6885. The method of claim 6733 wherein the agent is not a glucocorticosteroid .
6886. The method of claim 6733 wherein the agent is not dexamethasone.
6887. The method of claim 6733 wherein the agent is not beclomethasone.
6888. The method of claim 6733 wherein the agent is not dipropionate.
6889. The method of claim 6733 wherein the agent is not an anti- infective agent.
6890. The method of claim 6733 wherein the agent is not an antibiotic.
6891. The method of claim 6733 wherein the agent is not an anti- fungal agent.
6892. The method of claim 6733, wherein the composition comprises a polymer.
6893. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a copolymer.
6894. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a block copolymer.
6895. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a random copolymer.
6896. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a biodegradable polymer.
6897. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-biodegradable polymer.
6898. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophilic polymer.
6899. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophobic polymer.
6900. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophilic domains.
6901. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophobic domains.
6902. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-conductive polymer.
6903. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, an elastomer.
6904. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrogel.
6905. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a silicone polymer.
6906. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrocarbon polymer.
6907. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a styrene-derived polymer.
6908. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a butadiene-derived polymer.
6909. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a macromer.
6910. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, a poly( ethylene glycol) polymer.
6911. The method of claim 6733, wherein the composition comprises a polymer, and the polymer is, or comprises, an amorphous polymer.
6912. The method of claim 6733, wherein the composition further comprises a second pharmaceutically active agent.
6913. The method of claim 6733, wherein the composition further comprises an anti-inflammatory agent.
6914. The method of claim 6733, wherein the composition further comprises an agent that inhibits infection.
6915. The method of claim 6733, wherein the composition further comprises an anthracycline.
6916. The method of claim 6733, wherein the composition further comprises doxorubicin.
6917. The method of claim 6733 wherein the composition further comprises mitoxantrone.
6918. The method of claim 6733 wherein the composition further comprises a fluoropyrimidine.
6919. The method of claim 6733, wherein the composition further comprises 5-fluorouracil (5-FU).
6920. The method of claim 6733, wherein the composition further comprises a folic acid antagonist.
6921. The method of claim 6733, wherein the composition further comprises methotrexate.
6922. The method of claim 6733, wherein the composition further comprises a podophylotoxin.
6923. The method of claim 6733, wherein the composition further comprises etoposide.
6924. The method, of claim 6733, wherein the composition further comprises camptothecin.
6925. The method of claim 6733, wherein the composition further comprises a hydroxyurea.
6926. The method of claim 6733, wherein the composition further comprises a platinum complex.
6927. The method of claim 6733, wherein the composition further comprises cisplatin.
6928. The method of claim 6733 wherein the composition further comprises an anti-thrombotic agent.
6929. The method of claim 6733, wherein the composition further comprises a visualization agent.
6930. The method of claim 6733, wherein the composition further comprises a visualization agent, and the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
6931. The method of claim 6733, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, barium, tantalum, or technetium.
6932. The method of claim 6733, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, an MRI responsive material.
6933. The method of claim 6733, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a gadolinium chelate.
6934. The method of claim 6733, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron, magnesium, manganese, copper, or chromium.
6935. The method of claim 6733, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron oxide compound.
6936. The method of claim 6733, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a dye, pigment, or colorant.
6937. The method of claim 6733 wherein the agent is released in effective concentrations from the composition comprising the agent by diffusion over a period ranging from the time of administration to about 90 days.
6938. The method of claim 6733 wherein the agent is released in effective concentrations from the composition comprising the agent by erosion of the composition over a period ranging from the time of administration to about 90 days.
6939. The method of claim 6733 wherein the composition further comprises an inflammatory cytokine.
6940. The method of claim 6733 wherein the composition further comprises an agent that stimulates cell proliferation.
6941. The method of claim 6733 wherein the composition further comprises a polymeric carrier.
6942. The method of claim 6733 wherein the composition is in the form of a gel, paste, or spray.
6943. The method of claim 6733 wherein the pump is partially constructed with the agent or the composition.
6944. The method of claim 6733 wherein the pump is impregnated with the agent or the composition.
6945. The method of claim 6733, wherein the agent or the composition forms a coating, and the coating directly contacts the pump.
6946. The method of claim 6733, wherein the agent or the composition forms a coating, and the coating indirectly contacts the pump.
6947. The method of claim 6733 wherein the agent or the composition forms a coating, and the coating partially covers the pump.
6948. The method of claim 6733, wherein the agent or the composition forms a coating, and the coating completely covers the pump.
6949. The method of claim 6733 wherein the agent or the composition is located within pores or holes of the pump.
6950. The method of claim 6733 wherein the agent or the composition is located within a channel, lumen, or divet of the pump.
6951. The method of claim 6733 wherein the pump further comprises an echogenic material.
6952. The method of claim 6733 wherein the pump further comprises an echogenic material, wherein the echogenic material is in the form of a coating.
6953. The method of claim 6733 wherein the pump is sterile.
6954. The method of claim 6733 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump.
6955. The method of claim 6733 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is connective tissue.
6956. The method of claim 6733 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is muscle tissue.
6957. The method of claim 6733 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is nerve tissue.
6958. The method of claim 6733 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is epithelium tissue.
6959. The method of claim 6733 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from the time of deployment of the pump to about 1 year.
6960. The method of claim 6733 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from about 1 month to 6 months.
6961. The method of claim 6733 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from about 1 - 90 days.
6962. The method of claim 6733 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at a constant rate.
6963. The method of claim 6733 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at an increasing rate.
6964. The method of claim 6733 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at a decreasing rate.
6965. The method of claim 6733 wherein the agent is delivered from the pump, wherein the pump comprises about 0.01 μg to about 10 μg of the agent.
6966. The method of claim 6733 wherein the agent is delivered from the pump, wherein the pump comprises about 10 μg to about 10 mg of the agent.
6967. The method of claim 6733 wherein the agent is delivered from the pump, wherein the pump comprises about 10 mg to about 250 mg of the agent.
6968. The method of claim 6733 wherein the agent is delivered from the pump, wherein the pump comprises about 250 mg to about 1000 mg of the agent.
6969. The method of claim 6733 wherein the agent is delivered from the pump, wherein the pump comprises about 1000 mg to about 2500 mg of the agent.
6970. The method of claim 6733 wherein the agent is delivered from the pump, wherein a surface of the pump comprises less than 0.01 μg of the agent per mm2 of pump surface to which the agent is applied.
6971. The method of claim 6733 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 0.01 μg to about 1 μg of the agent per mm2 of pump surface to which the agent is applied.
6972. The method of claim 6733 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 1 μg to about 10 μg of the agent per mm2 of pump surface to which the agent is applied.
6973. The method of claim 6733 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 10 μg to about 250 μg of the agent per mm2 of pump surface to which the agent is applied.
6974. The method of claim 6733 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 250 μg to about 1000 μg of the agent per mm2 of pump surface to which the agent is applied.
6975. The method of claim 6733 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 1000 μg to about 2500 μg of the agent per mm2 of pump surface to which the agent is applied.
6976. The method of claim 6733, wherein the pump further comprises a coating, and the coating is a uniform coating.
6977. The method of claim 6733, wherein the pump further comprises a coating, and the coating is a non-uniform coating.
6978. The method of claim 6733, wherein the pump further comprises a coating, and the coating is a discontinuous coating.
6979. The method of claim 6733, wherein the pump further comprises a coating, and the coating is a patterned coating.
6980. The method of claim 6733, wherein the pump further comprises a coating, and the coating has a thickness of 100 μm or less.
6981. The method of claim 6733, wherein the pump further comprises a coating, and the coating has a thickness of 10 μm or less.
6982. The method of claim 6733, wherein the pump further comprises a coating, and the coating adheres to the surface of the pump upon
' deployment of the pump.
6983. The method of claim 6733, wherein the pump further comprises a coating, and the coating is stable at room temperature for a period of at least 1 year.
6984. The method of claim 6733, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 0.0001 % to about 1% by weight
6985. The method of claim 6733, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
6986. The method of claim 6733, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
6987. The method of claim 6733, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
6988. The method of claim 6733, wherein the pump further comprises a coating, and the coating comprises a polymer.
6989. The method of claim 6733, wherein the pump comprises a first coating having a first composition and a second coating having a second composition.
6990. The method of claim 6733, wherein the pump comprises a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
6991. The method of claim 6733 wherein the agent or the composition is affixed to the pump.
6992. The method of claim 6733 wherein the agent or the composition is covalently attached to the pump.
6993. The method of claim 6733 wherein the agent or the composition is non-covalently attached to the pump.
6994. The method of claim 6733 wherein the pump comprises a coating that absorbs the agent or the composition.
6995. The method of claim 6733 wherein the pump is interweaved with a thread composed of, or coated with, the agent or the composition.
6996. The method of claim 6733 wherein a portion of the pump is covered with a sleeve that contains the agent or the composition.
6997. The method of claim 6733 wherein the pump is completely covered with a sleeve that contains the agent or the composition.
6998. The method of claim 6733 wherein a portion of the pump is covered with a mesh that contains the agent or the composition.
6999. The method of claim 6733 wherein the pump is completely covered with a mesh that contains the agent or the composition.
7000. The method of claim 6733 wherein the pump is linked to a sensor.
7001. The method of claim 6733 wherein the agent or the composition is applied to the pump surface prior to to the placing of the pump into the host.
7002. The method of claim 6733 wherein the agent or the composition is applied to the pump surface during the placing of the pump into the host.
7003. The method of claim 6733 wherein the agent or the composition is applied to the pump surface immediately after the placing of the pump into the host.
7004. The method of claim 6733 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump prior to to the placing of the pump into the host.
7005. The method of claim 6733 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump during the placing of the pump into the host.
7006. The method of claim 6733 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump immediately after the placing of the pump into the host.
7007. The method of claim 6733 wherein the agent or the composition is topically applied into the anatomical space where the pump is placed.
7008. The method of claim 6733 wherein the agent or the composition is percutaneously injected into the tissue in the host surrounding the pump.
7009. The method of any one of claims 6733-7008 wherein the pump is an implantable cardiac electrode that delivers stimulation energy and dispenses drug adjacent to the stimulation site.
7010. A method for inhibiting scarring comprising placing a drug delivery implant (i.e., a pump) and an anti-scarring agent or a composition comprising an anti-scarring agent into an animal host, wherein the agent inhibits scarring.
7011. The method of claim 7010 wherein the agent inhibits cell regeneration.
7012. The method of claim 7010 wherein the agent inhibits angiogenesis.
7013. The method of claim 7010 wherein the agent inhibits fibroblast migration.
7014. The method of claim 7010 wherein the agent inhibits fibroblast proliferation.
7015. The method of claim 7010 wherein the agent inhibits deposition of extracellular matrix.
7016. The method of claim 7010 wherein the agent inhibits tissue remodeling.
7017. The method of claim 7010 wherein the agent is an angiogenesis inhibitor.
7018. The method of claim 7010 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
7019. The method of claim 7010 wherein the agent is a chemokine receptor antagonist.
7020. The method of claim 7010 wherein the agent is a cell cycle inhibitor.
7021. The method of claim 7010 wherein the agent is a taxane.
7022. The method of claim 7010 wherein the agent is an antimicrotubule agent.
7023. The method of claim 7010 wherein the agent is paclitaxel.
7024. The method of claim 7010 wherein the agent is not paclitaxel.
7025. The method of claim 7010 wherein the agent is an analogue or derivative of paclitaxel.
7026. The method of claim 7010 wherein the agent is a vinca alkaloid.
7027. The method of claim 7010 wherein the agent is camptothecin or an analogue or derivative thereof.
7028. The method of claim 7010 wherein the agent is a podophyllotoxin.
7029. The method of claim 7010 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
7030. The method of claim 7010 wherein the agent is an anthracycline.
7031. The method of claim 7010 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
7032. The method of claim 7010 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
7033. The method of claim 7010 wherein the agent is a platinum compound.
7034. The method of claim 7010 wherein the agent is a nitrosourea.
7035. The method of claim 7010 wherein the agent is a nitroimidazole.
7036. The method of claim 7010 wherein the agent is a folic acid antagonist.
7037. The method of claim 7010 wherein the agent is a cytidine analogue.
7038. The method of claim 7010 wherein the agent is a pyrimidine analogue.
7039. The method of claim 7010 wherein the agent is a fluoropyrimidine analogue.
7040. The method of claim 7010 wherein the agent is a purine analogue.
7041. The method of claim 7010 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
7042. The method of claim 7010 wherein the agent is a hydroxyurea.
7043. The method of claim 7010 wherein the agent is a mytomicin or an analogue or derivative thereof.
7044. The method of claim 7010 wherein the agent is an alkyl sulfonate.
7045. The method of claim 7010 wherein the agent is a benzamide or an analogue or derivative thereof.
7046. The method of claim 7010 wherein the agent is a nicotinamide or an analogue or derivative thereof.
7047. The method of claim 7010 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
7048. The method of claim 7010 wherein the agent is a DNA alkylating agent.
7049. The method of claim 7010 wherein the agent is an antimicrotubule agent.
7050. The method of claim 7010 wherein the agent is a topoisomerase inhibitor.
7051. The method of claim 7010 wherein the agent is a DNA cleaving agent
7052. The method of claim 7010 wherein the agent is an antimetabolite.
7053. The method of claim 7010 wherein the agent inhibits adenosine deaminase.
7054. The method of claim 7010 wherein the agent inhibits purine ring synthesis.
7055. The method of claim 7010 wherein the agent is a nucleotide interconversion inhibitor.
7056. The method of claim 7010 wherein the agent inhibits dihydrofolate reduction.
7057. The method of claim 7010 wherein the agent blocks thymidine monophosphate.
7058. The method of claim 7010 wherein the agent causes DNA damage.
7059. The method of claim 7010 wherein the agent is a DNA intercalation agent.
7060. The method of claim 7010 wherein the agent is a RNA synthesis inhibitor.
7061. The method of claim 7010 wherein the agent is a pyrimidine synthesis inhibitor.
7062. The method of claim 7010 wherein the agent inhibits ribonucleotide synthesis or function.
7063. The method of claim 7010 wherein the agent inhibits thymidine monophosphate synthesis or function.
7064. The method of claim 7010 wherein the agent inhibits DNA synthesis.
7065. The method of claim 7010 wherein the agent causes DNA adduct formation.
7066. The method of claim 7010 wherein the agent inhibits protein synthesis.
7067. The method of claim 7010 wherein the agent inhibits microtubule function.
7068. The method of claim 7010 wherein the agent is a cyclin dependent protein kinase inhibitor.
7069. The method of claim 7010 wherein the agent is an epidermal growth factor kinase inhibitor.
7070. The method of claim 7010 wherein the agent is an elastase inhibitor.
7071. The method of claim 7010 wherein the agent is a factor Xa inhibitor.
7072. The method of claim 7010 wherein the agent is a farnesyltransferase inhibitor.
7073. The method of claim 7010 wherein the agent is a fibrinogen antagonist.
7074. The method of claim 7010 wherein the agent is a guanylate cyclase stimulant.
7075. The method of claim 7010 wherein the agent is a heat shock protein 90 antagonist.
7076. The method of claim 7010 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
7077. The method of claim 7010 wherein the agent is a guanylate cyclase stimulant.
7078. The method of claim 7010 wherein the agent is a HMGCoA reductase inhibitor.
7079. The method of claim 7010 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
7080. The method of claim 7010 wherein the agent is a hydroorotate dehydrogenase inhibitor.
7081. The method of claim 7010 wherein the agent is an IKK2 inhibitor.
7082. The method of claim 7010 wherein the agent is an IL-1 antagonist
7083. The method of claim 7010 wherein the agent is an ICE antagonist.
7084. The method of claim 7010 wherein the agent is an IRAK antagonist
7085. The method of claim 7010 wherein the agent is an IL-4 agonist.
7086. The method of claim 7010 wherein the agent is an immunomodulatory agent.
7087. The method of claim 7010 wherein the agent is sirolimus or an analogue or derivative thereof.
7088. The method of claim 7010 wherein the agent is not sirolimus.
7089. The method of claim 7010 wherein the agent is everolimus or an analogue or derivative thereof.
7090. The method of claim 7010 wherein the agent is tacrolimus or an analogue or derivative thereof.
7091. The method of claim 7010 wherein the agent is not tacrolimus.
7092. The method of claim 7010 wherein the agent is biolmus or an analogue or derivative thereof.
7093. The method of claim 7010 wherein the agent is tresperimus or an analogue or derivative thereof.
7094. The method of claim 7010 wherein the agent is auranofin or an analogue or derivative thereof.
7095. The method of claim 7010 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
7096. The method of claim 7010 wherein the agent is gusperimus or an analogue or derivative thereof.
7097. The method of claim 7010 wherein the agent is pimecrolimus or an analogue or derivative thereof.
7098. The method of claim 7010 wherein the agent is ABT-578 or an analogue or derivative thereof.
7099. The method of claim 7010 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
7100. The method of claim 7010 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
7101. The method of claim 7010 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
7102. The method of claim 7010 wherein the agent is a leukotriene inhibitor.
7103. The method of claim 7010 wherein the agent is a MCP-1 antagonist.
7104. The method of claim 7010 wherein the agent is a MMP inhibitor.
7105. The method of claim 7010 wherein the agent is an NF kappa B inhibitor.
7106. The method of claim 7010 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
7107. The method of claim 7010 wherein the agent is an NO antagonist.
7108. The method of claim 7010 wherein the agent is a p38 MAP kinase inhibitor.
7109. The method of claim 7010 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
7110. The method of claim 7010 wherein the agent is a phosphodiesterase inhibitor.
7111. The method of claim 7010 wherein the agent is a TGF beta inhibitor.
7112. The method of claim 7010 wherein the agent is a thromboxane A2 antagonist.
7113. The method of claim 7010 wherein the agent is a TNF alpha antagonist.
7114. The method of claim 7010 wherein the agent is a TACE inhibitor.
7115. The method of claim 7010 wherein the agent is a tyrosine kinase inhibitor.
7116. The method of claim 7010 wherein the agent is a vitronectin inhibitor.
7117. The method of claim 7010 wherein the agent is a fibroblast growth factor inhibitor.
7118. The method of claim 7010 wherein the agent is a protein kinase inhibitor.
7119. The method of claim 7010 wherein the agent is a PDGF receptor kinase inhibitor.
7120. The method of claim 7010 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
7121. The method of claim 7010 wherein the agent is a retinoic acid receptor antagonist.
7122. The method of claim 7010 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
7123. The method of claim 7010 wherein the agent is a fibrinogen antagonist.
7124. The method of claim 7010 wherein the agent is an antimycotic agent.
7125. The method of claim 7010 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
7126. The method of claim 7010 wherein the agent is a bisphosphonate.
7127. The method of claim 7010 wherein the agent is a phospholipase A1 inhibitor.
7128. The method of claim 7010 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
7129. The method of claim 7010 wherein the agent is a macrolide antibiotic.
7130. The method of claim 7010 wherein the agent is a GPIIb/llla receptor antagonist.
7131. The method of claim 7010 wherein the agent is an endothelin receptor antagonist.
7132. The method of claim 7010 wherein the agent is a peroxisome proliferator-activated receptor agonist.
7133. The method of claim 7010 wherein the agent is an estrogen receptor agent.
7134. The method of claim 7010 wherein the agent is a somastostatin analogue.
7135. The method of claim 7010 wherein the agent is a neurokinin 1 antagonist.
7136. The method of claim 7010 wherein the agent is a neurokinin 3 antagonist.
7137. The method of claim 7010 wherein the agent is a VLA-4 antagonist.
7138. The method of claim 7010 wherein the agent is an osteoclast inhibitor.
7139. The method of claim 7010 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
7140. The method of claim 7010 wherein the agent is an angiotensin I converting enzyme inhibitor.
7141. The method of claim 7010 wherein the agent is an angiotensin II antagonist.
7142. The method of claim 7010 wherein the agent is an enkephalinase inhibitor.
7143. The method of claim 7010 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
7144. The method of claim 7010 wherein the agent is a protein kinase C inhibitor.
7145. The method of claim 7010 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
7146. The method of claim 7010 wherein the agent is a CXCR3 inhibitor.
7147. The method of claim 7010 wherein the agent is an Itk inhibitor.
7148. The method of claim 7010 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
7149. The method of claim 7010 wherein the agent is a PPAR agonist.
7150. The method of claim 7010 wherein the agent is an immunosuppressant
7151. The method of claim 7010 wherein the agent is an Erb inhibitor.
7152. The method of claim 7010 wherein the agent is an apoptosis agonist.
7153. The method of claim 7010 wherein the agent is a lipocortin agonist.
7154. The method of claim 7010 wherein the agent is a VCAM-1 antagonist.
7155. The method of claim 7010 wherein the agent is a collagen antagonist.
7156. The method of claim 7010 wherein the agent is an alpha 2 integrin antagonist.
7157. The method of claim 7010 wherein the agent is a TNF alpha inhibitor.
7158. The method of claim 7010 wherein the agent is a nitric oxide inhibitor
7159. The method of claim 7010 wherein the agent is a cathepsin inhibitor.
7160. The method of claim 7010 wherein the agent is not an anti- inflammatory agent.
7161. The method of claim 7010 wherein the agent is not a steroid.
7162. The method of claim 7010 wherein the agent is not a glucocorticosteroid.
7163. The method of claim 7010 wherein the agent is not dexamethasone.
7164. The method of claim 7010 wherein the agent is not beclomethasone.
7165. The method of claim 7010 wherein the agent is not dipropionate.
7166. The method of claim 7010 wherein the agent is not an anti- infective agent.
7167. The method of claim 7010 wherein the agent is not an antibiotic.
7168. The method of claim 7010 wherein the agent is not an anti- fungal agent.
7169. The method of claim 7010, wherein the composition comprises a polymer.
7170. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a copolymer.
7171. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a block copolymer.
7172. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a random copolymer.
7173. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a biodegradable polymer.
7174. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-biodegradable polymer.
7175. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophilic polymer.
7176. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrophobic polymer.
7177. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophilic domains.
7178. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a polymer having hydrophobic domains.
7179. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a non-conductive polymer.
7180. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, an elastomer.
7181. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrogel.
7182. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a silicone polymer.
7183. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a hydrocarbon polymer.
7184. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a styrene-derived polymer.
7185. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a butadiene-derived polymer.
7186. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a macromer.
7187. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, a poly(ethylene glycol) polymer.
7188. The method of claim 7010, wherein the composition comprises a polymer, and the polymer is, or comprises, an amorphous polymer.
7189. The method of claim 7010, wherein the composition further comprises a second pharmaceutically active agent.
7190. The method of claim 7010, wherein the composition further comprises an anti-inflammatory agent.
7191. The method of claim 7010, wherein the composition further comprises an agent that inhibits infection.
7192. The method of claim 7010, wherein the composition further comprises an anthracycline.
7193. The method of claim 7010, wherein the composition further comprises doxorubicin.
7194. The method of claim 7010 wherein the composition further comprises mitoxantrone.
7195. The method of claim 7010 wherein the composition further comprises a fluoropyrimidine.
7196. The method of claim 7010, wherein the composition further comprises 5-fluorouracil (5-FU).
7197. The method of claim 7010, wherein the composition further comprises a folic acid antagonist.
7198. The method of claim 7010, wherein the composition further comprises methotrexate.
7199. The method of claim 7010, wherein the composition further comprises a podophylotoxin.
7200. The method of claim 7010, wherein the composition further comprises etoposide.
7201. The method of claim 7010, wherein the composition further comprises camptothecin.
7202. The method of claim 7010, wherein the composition further comprises a hydroxyurea.
7203. The method of claim 7010, wherein the composition further comprises a platinum complex.
7204. The method of claim 7010, wherein the composition further comprises cisplatin.
7205. The method of claim 7010 wherein the composition further comprises an anti-thrombotic agent.
7206. The method of claim 7010, wherein the composition further comprises a visualization agent.
7207. The method of claim 7010, wherein the composition further comprises a visualization agent, and the visualization agent is a radiopaque material, wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
7208. The method of claim 7010, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, barium, tantalum, ortechnetium.
7209. The method of claim 7010, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, an MRI responsive material.
7210. The method of claim 7010, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a gadolinium chelate.
7211. The method of claim 7010, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron, magnesium, manganese, copper, or chromium.
7212. The method of claim 7010, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, iron oxide compound.
7213. The method of claim 7010, wherein the composition further comprises a visualization agent, and the visualization agent is, or comprises, a dye, pigment, or colorant.
7214. The method of claim 7010 wherein the agent is released in effective concentrations from the composition comprising the agent by diffusion over a period ranging from the time of administration to about 90 days.
7215. The method of claim 7010 wherein the agent is released in effective concentrations from the composition comprising the agent by erosion of the composition over a period ranging from the time of administration to about 90 days.
7216. The method of claim 7010 wherein the composition further comprises an inflammatory cytokine.
7217. The method of claim 7010 wherein the composition further comprises an agent that stimulates cell proliferation.
7218. The method of claim 7010 wherein the composition further comprises a polymeric carrier.
7219. The method of claim 7010 wherein the composition is in the form of a gel, paste, or spray.
7220. The method of claim 7010 wherein the pump is partially constructed with the agent or the composition.
7221. The method of claim 7010 wherein the pump is impregnated with the agent or the composition.
7222. The method of claim 7010, wherein the agent or the composition forms a coating, and the coating directly contacts the pump.
7223. The method of claim 7010, wherein the agent or the composition forms a coating, and the coating indirectly contacts the pump.
7224. The method of claim 7010 wherein the agent or the composition forms a coating, and the coating partially covers the pump.
7225. The method of claim 7010, wherein the agent or the composition forms a coating, and the coating completely covers the pump.
7226. The method of claim 7010 wherein the agent or the composition is located within pores or holes of the pump.
7227. The method of claim 7010 wherein the agent or the composition is located within a channel, lumen, or divet of the pump.
7228. The method of claim 7010 wherein the pump further comprises an echogenic material.
7229. The method of claim 7010 wherein the pump further comprises an echogenic material, wherein the echogenic material is in the form of a coating.
7230. The method of claim 7010 wherein the pump is sterile.
7231. The method of claim 7010 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump.
7232. The method of claim 7010 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is connective tissue.
7233. The method of claim 7010 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is muscle tissue.
7234. The method of claim 7010 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is nerve tissue.
7235. The method of claim 7010 wherein the agent is delivered from the pump, wherein the agent is released into tissue in the vicinity of the pump after deployment of the pump, wherein the tissue is epithelium tissue.
7236. The method of claim 7010 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from the time of deployment of the pump to about 1 year.
7237. The method of claim 7010 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from about 1 month to 6 months.
7238. The method of claim 7010 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump over a period ranging from about 1 - 90 days.
7239. The method of claim 7010 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at a constant rate.
7240. The method of claim 7010 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at an increasing rate.
7241. The method of claim 7010 wherein the agent is delivered from the pump, wherein the agent is released in effective concentrations from the pump at a decreasing rate.
7242. The method of claim 7010 wherein the agent is delivered from the pump, wherein the pump comprises about 0.01 μg to about 10 μg of the agent.
7243. The method of claim 7010 wherein the agent is delivered from the pump, wherein the pump comprises about 10 μg to about 10 mg of the agent.
7244. The method of claim 7010 wherein the agent is delivered from the pump, wherein the pump comprises about 10 mg to about 250 mg of the agent.
7245. The method of claim 7010 wherein the agent is delivered from the pump, wherein the pump comprises about 250 mg to about 1000 mg of the agent.
7246. The method of claim 7010 wherein the agent is delivered from the pump, wherein the pump comprises about 1000 mg to about 2500 mg of the agent.
7247. The method of claim 7010 wherein the agent is delivered from the pump, wherein a surface of the pump comprises less than 0.01 μg of the agent per mm2 of pump surface to which the agent is applied.
7248. The method of claim 7010 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 0.01 μg to about 1 μg of the agent per mm2 of pump surface to which the agent is applied.
7249. The method of claim 7010 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 1 μg to about 10 μg of the agent per mm2 of pump surface to which the agent is applied.
7250. The method of claim 7010 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 10 μg to about 250 μg of the agent per mm2 of pump surface to which the agent is applied.
7251. The method of claim 7010 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 250 μg to about 1000 μg of the agent per mm2 of pump surface to which the agent is applied.
7252. The method of claim 7010 wherein the agent is delivered from the pump, wherein a surface of the pump comprises about 1000 μg to about 2500 μg of the agent per mm2 of pump surface to which the agent is applied.
7253. The method of claim 7010, wherein the pump further comprises a coating, and the coating is a uniform coating.
7254. The method of claim 7010, wherein the pump further comprises a coating, and the coating is a non-uniform coating.
7255. The method of claim 7010, wherein the pump further comprises a coating, and the coating is a discontinuous coating.
7256. The method of claim 7010, wherein the pump further comprises a coating, and the coating is a patterned coating.
7257. The method of claim 7010, wherein the pump further comprises a coating, and the coating has a thickness of 100 μm or less.
7258. The method of claim 7010, wherein the pump further comprises a coating, and the coating has a thickness of 10 μm or less.
7259. The method of claim 7010, wherein the pump further comprises a coating, and the coating adheres to the surface of the pump upon deployment of the pump.
7260. The method of claim 7010, wherein the pump further comprises a coating, and the coating is stable at room temperature for a period of at least 1 year.
7261. The method of claim 7010, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 0.0001 % to about 1 % by weight.
7262. The method of claim 7010, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
7263. The method of claim 7010, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
7264. The method of claim 7010, wherein the pump further comprises a coating, and the agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
7265. The method of claim 7010, wherein the pump further comprises a coating, and the coating comprises a polymer.
7266. The method of claim 7010, wherein the pump comprises a first coating having a first composition and a second coating having a second composition.
7267. The method of claim 7010, wherein the pump comprises a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different
7268. The method of claim 7010 wherein the agent or the composition is affixed to the pump.
7269. The method of claim 7010 wherein the agent or the composition is covalently attached to the pump.
7270. The method of claim 7010 wherein the agent or the composition is non-covalently attached to the pump.
7271. The method of claim 7010 wherein the pump comprises a coating that absorbs the agent or the composition.
7272. The method of claim 7010 wherein the pump is interweaved with a thread composed of, or coated with, the agent or the composition.
7273. The method of claim 7010 wherein a portion of the pump is covered with a sleeve that contains the agent or the composition.
7274. The method of claim 7010 wherein the pump is completely covered with a sleeve that contains the agent or the composition.
7275. The method of claim 7010 wherein a portion of the pump is covered with a mesh that contains the agent or the composition.
7276. The method of claim 7010 wherein the pump is completely covered with a mesh that contains the agent or the composition.
7277. The method of claim 7010 wherein the pump is linked to a sensor.
7278. The method of claim 7010 wherein the agent or the composition is applied to the pump surface prior to to the placing of the pump into the host.
7279. The method of claim 7010 wherein the agent or the composition is applied to the pump surface during the placing of the pump into the host.
7280. The method of claim 7010 wherein the agent or the composition is applied to the pump surface immediately after the placing of the pump into the host.
7281. The method of claim 7010 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump prior to to the placing of the pump into the host.
7282. The method of claim 7010 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump during the placing of the pump into the host.
7283. The method of claim 7010 wherein the agent or the composition is applied to the surface of the tissue in the host surrounding the pump immediately after the placing of the pump into the host.
7284. The method of claim 7010 wherein the agent or the composition is topically applied into the anatomical space where the pump is placed.
7285. The method of claim 7010 wherein the agent or the composition is percutaneously injected into the tissue in the host surrounding the pump.
7286. A method for making a device comprising: combining a sensor and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
7287. The method of claim 7286 wherein the agent inhibits cell regeneration.
7288. The method of claim 7286 wherein the agent inhibits angiogenesis.
7289. The method of claim 7286 wherein the agent inhibits fibroblast migration.
7290. The method of claim 7286 wherein the agent inhibits fibroblast proliferation.
7291. The method of claim 7286 wherein the agent inhibits deposition of extracellular matrix.
7292. The method of claim 7286 wherein the agent inhibits tissue remodeling.
7293. The method of claim 7286 wherein the agent is an angiogenesis inhibitor.
7294. The method of claim 7286 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
7295. The method of claim 7286 wherein the agent is a chemokine receptor antagonist.
7296. The method of claim 7286 wherein the agent is a cell cycle inhibitor.
7297. The method of claim 7286 wherein the agent is a taxane.
7298. The method of claim 7286 wherein the agent is an antimicrotubule agent.
7299. The method of claim 7286 wherein the agent is paclitaxel.
7300. The method of claim 7286 wherein the agent is not paclitaxel.
7301. The method of claim 7286 wherein the agent is an analogue or derivative of paclitaxel.
7302. The method of claim 7286 wherein the agent is a vinca alkaloid.
7303. The method of claim 7286 wherein the agent is camptothecin or an analogue or derivative thereof.
7304. The method of claim 7286 wherein the agent is a podophyllotoxin.
7305. The method of claim 7286 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
7306. The method of claim 7286 wherein the agent is an anthracycline.
7307. The method of claim 7286 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
7308. The method of claim 7286 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
7309. The method of claim 7286 wherein the agent is a platinum compound.
7310. The method of claim 7286 wherein the agent is a nitrosourea.
7311. The method of claim 7286 wherein the agent is a nitroimidazole.
7312. The method of claim 7286 wherein the agent is a folic acid antagonist.
7313. The method of claim 7286 wherein the agent is a cytidine analogue.
7314. The method of claim 7286 wherein the agent is a pyrimidine analogue.
7315. The method of claim 7286 wherein the agent is a fluoropyrimidine analogue.
7316. The method of claim 7286 wherein the agent is a purine analogue.
7317. The method of claim 7286 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
7318. The method of claim 7286 wherein the agent is a hydroxyurea.
7319. The method of claim 7286 wherein the agent is a mytomicin or an analogue or derivative thereof.
7320. The method of claim 7286 wherein the agent is an alkyl sulfonate.
7321. The method of claim 7286 wherein the agent is a benzamide or an analogue or derivative thereof.
7322. The method of claim 7286 wherein the agent is a nicotinamide or an analogue or derivative thereof.
7323. The method of claim 7286 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
7324. The method of claim 7286 wherein the agent is a DNA alkylating agent.
7325. The method of claim 7286 wherein the agent is an antimicrotubule agent.
7326. The method of claim 7286 wherein the agent is a topoisomerase inhibitor.
7327. The method of claim 7286 wherein the agent is a DNA cleaving agent.
7328. The method of claim 7286 wherein the agent is an antimetabolite.
7329. The method of claim 7286 wherein the agent inhibits adenosine deaminase.
7330. The method of claim 7286 wherein the agent inhibits purine ring synthesis.
7331. The method of claim 7286 wherein the agent is a nucleotide interconversion inhibitor.
7332. The method of claim 7286 wherein the agent inhibits dihydrofolate reduction.
7333. The method of claim 7286 wherein the agent blocks thymidine monophosphate.
7334. The method of claim 7286 wherein the agent causes DNA damage.
7335. The method of claim 7286 wherein the agent is a DNA intercalation agent.
7336. The method of claim 7286 wherein the agent is a RNA synthesis inhibitor.
7337. The method of claim 7286 wherein the agent is a pyrimidine synthesis inhibitor.
7338. The method of claim 7286 wherein the agent inhibits ribonucleotide synthesis or function.
7339. The method of claim 7286 wherein the agent inhibits thymidine monophosphate synthesis or function.
7340. The method of claim 7286 wherein the agent inhibits DNA synthesis.
7341. The method of claim 7286 wherein the agent causes DNA adduct formation.
7342. The method of claim 7286 wherein the agent inhibits protein synthesis.
7343. The method of claim 7286 wherein the agent inhibits microtubule function.
7344. The method of claim 7286 wherein the agent is a cyclin dependent protein kinase inhibitor.
7345. The method of claim 7286 wherein the agent is an epidermal growth factor kinase inhibitor.
7346. The method of claim 7286 wherein the agent is an elastase inhibitor.
7347. The method of claim 7286 wherein the agent is a factor Xa inhibitor.
7348. The method of claim 7286 wherein the agent is a farnesyltransferase inhibitor.
7349. The method of claim 7286 wherein the agent is a fibrinogen antagonist.
7350. The method of claim 7286 wherein the agent is a guanylate cyclase stimulant.
7351. The method of claim 7286 wherein the agent is a heat shock protein 90 antagonist.
7352. The method of claim 7286 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
7353. The method of claim 7286 wherein the agent is a guanylate cyclase stimulant.
7354. The method of claim 7286 wherein the agent is a HMGCoA reductase inhibitor.
7355. The method of claim 7286 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
7356. The method of claim 7286 wherein the agent is a hydroorotate dehydrogenase inhibitor.
7357. The method of claim 7286 wherein the agent is an IKK2 inhibitor.
7358. The method of claim 7286 wherein the agent is an IL-1 antagonist.
7359. The method of claim 7286 wherein the agent is an ICE antagonist.
7360. The method of claim 7286 wherein the agent is an IRAK antagonist.
7361. The method of claim 7286 wherein the agent is an IL-4 agonist.
7362. The method of claim 7286 wherein the agent is an immunomodulatory agent.
7363. The method of claim 7286 wherein the agent is sirolimus or an analogue or derivative thereof.
7364. The method of claim 7286 wherein the agent is not sirolimus.
7365. The method of claim 7286 wherein the agent is everolimus or an analogue or derivative thereof.
7366. The method of claim 7286 wherein the agent is tacrolimus or an analogue or derivative thereof.
7367. The method of claim 7286 wherein the agent is not tacrolimus.
7368. The method of claim 7286 wherein the agent is biolmus or an analogue or derivative thereof.
7369. The method of claim 7286 wherein the agent is tresperimus or an analogue or derivative thereof.
7370. The method of claim 7286 wherein the agent is auranofin or an analogue or derivative thereof.
7371. The method of claim 7286 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
7372. The method of claim 7286 wherein the agent is gusperimus or an analogue or derivative thereof.
7373. The method of claim 7286 wherein the agent is pimecrolimus or an analogue or derivative thereof.
7374. The method of claim 7286 wherein the agent is ABT-578 or an analogue or derivative thereof.
7375. The method of claim 7286 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
7376. The method of claim 7286 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
7377. The method of claim 7286 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
7378. The method of claim 7286 wherein the agent is a leukotriene inhibitor.
7379. The method of claim 7286 wherein the agent is a MCP-1 antagonist.
7380. The method of claim 7286 wherein the agent is a MMP inhibitor.
7381. The method of claim 7286 wherein the agent is an NF kappa B inhibitor.
7382. The method of claim 7286 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
7383. The method of claim 7286 wherein the agent is an NO antagonist.
7384. The method of claim 7286 wherein the agent is a p38 MAP kinase inhibitor.
7385. The method of claim 7286 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
7386. The method of claim 7286 wherein the agent is a phosphodiesterase inhibitor.
7387. The method of claim 7286 wherein the agent is a TGF beta inhibitor.
7388. The method of claim 7286 wherein the agent is a thromboxane A2 antagonist.
7389. The method of claim 7286 wherein the agent is a TNF alpha antagonist.
7390. The method of claim 7286 wherein the agent is a TACE inhibitor.
7391. The method of claim 7286 wherein the agent is a tyrosine kinase inhibitor.
7392. The method of claim 7286 wherein the agent is a vitronectin inhibitor.
7393. The method of claim 7286 wherein the agent is a fibroblast growth factor inhibitor.
7394. The method of claim 7286 wherein the agent is a protein kinase inhibitor.
7395. The method of claim 7286 wherein the agent is a PDGF receptor kinase inhibitor.
7396. The method of claim 7286 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
7397. The method of claim 7286 wherein the agent is a retinoic acid receptor antagonist.
7398. The method of claim 7286 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
7399. The method of claim 7286 wherein the agent is a fibrinogen antagonist.
7400. The method of claim 7286 wherein the agent is an antimycotic agent.
7401. The method of claim 7286 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
7402. The method of claim 7286 wherein the agent is a bisphosphonate.
7403. The method of claim 7286 wherein the agent is a phospholipase A1 inhibitor.
7404. The method of claim 7286 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
7405. The method of claim 7286 wherein the agent is a macrolide antibiotic.
7406. The method of claim 7286 wherein the agent is a GPIIb/llla receptor antagonist.
7407. The method of claim 7286 wherein the agent is an endothelin receptor antagonist.
7408. The method of claim 7286 wherein the agent is a peroxisome proliferator-activated receptor agonist.
7409. The method of claim 7286 wherein the agent is an estrogen receptor agent.
7410. The method of claim 7286 wherein the agent is a somastostatin analogue.
7411. The method of claim 7286 wherein the agent is a neurokinin 1 antagonist.
7412. The method of claim 7286 wherein the agent is a neurokinin 3 antagonist.
7413. The method of claim 7286 wherein the agent is a VLA-4 antagonist.
7414. The method of claim 7286 wherein the agent is an osteoclast inhibitor.
7415. The method of claim 7286 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
7416. The method of claim 7286 wherein the agent is an angiotensin I converting enzyme inhibitor.
7417. The method of claim 7286 wherein the agent is an angiotensin II antagonist.
7418. The method of claim 7286 wherein the agent is an enkephalinase inhibitor.
7419. The method of claim 7286 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
7420. The method of claim 7286 wherein the agent is a protein kinase C inhibitor.
7421. The method of claim 7286 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
7422. The method of claim 7286 wherein the agent is a CXCR3 inhibitor.
7423. The method of claim 7286 wherein the agent is an Itk inhibitor.
7424. The method of claim 7286 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
7425. The method of claim 7286 wherein the agent is a PPAR agonist.
7426. The method of claim 7286 wherein the agent is an immunosuppressant.
7427. The method of claim 7286 wherein the agent is an Erb inhibitor.
7428. The method of claim 7286 wherein the agent is an apoptosis agonist.
7429. The method of claim 7286 wherein the agent is a lipocortin agonist.
7430. The method of claim 7286 wherein the agent is a VCAM-1 antagonist.
7431. The method of claim 7286 wherein the agent is a collagen antagonist.
7432. The method of claim 7286 wherein the agent is an alpha 2 integrin antagonist.
7433. The method of claim 7286 wherein the agent is a TNF alpha inhibitor.
7434. The method of claim 7286 wherein the agent is a nitric oxide inhibitor
7435. The method of claim 7286 wherein the agent is a cathepsin inhibitor.
7436. The method of claim 7286 wherein the agent is not an anti- inflammatory agent.
7437. The method of claim 7286 wherein the agent is not a steroid.
7438. The method of claim 7286 wherein the agent is not a glucocorticosteroid.
7439. The method of claim 7286 wherein the agent is not dexamethasone.
7440. The method of claim 7286 wherein the agent is not beclomethasone.
7441. The method of claim 7286 wherein the agent is not dipropionate.
7442. The method of claim 7286 wherein the agent is not an anti- infective agent.
7443. The method of claim 7286 wherein the agent is not an antibiotic.
7444. The method of claim 7286 wherein the agent is not an anti- fungal agent.
7445. The method of claim 7286, wherein the composition comprises a polymer.
7446. The method of claim 7286, wherein the composition comprises a polymeric carrier.
7447. The method of claim 7286 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
7448. The method of claim 7286 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
7449. The method of claim 7286 wherein the device has a coating that comprises the anti-scarring agent.
7450. The method of claim 7286, wherein the device has a coating that comprises the agent and is disposed on a surface of the sensor.
7451. The method of claim 7286, wherein the device has a coating that comprises the agent and directly contacts the sensor.
7452. The method of claim 7286, wherein the device has a coating that comprises the agent and indirectly contacts the sensor.
7453. The method of claim 7286, wherein the device has a coating that comprises the agent and partially covers the sensor.
7454. The method of claim 7286, wherein the device has a coating that comprises the agent and completely covers the sensor.
7455. The method of claim 7286, wherein the device has a uniform coating.
7456. The method of claim 7286, wherein the device has a non- uniform coating.
7457. The method of claim 7286, wherein the device has a discontinuous coating.
7458. The method of claim 7286, wherein the device has a patterned coating.
7459. The method of claim 7286, wherein the device has a coating with a thickness of 100 μm or less.
7460. The method of claim 7286, wherein the device has a coating with a thickness of 10 μm or less.
7461. The method of claim 7286, wherein the device has a coating, and the coating adheres to the surface of the sensor upon deployment of the sensor.
7462. The method of claim 7286, wherein the device has a coating, and wherein the coating is stable at room temperature for a period of 1 year.
7463. The method of claim 7286, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1 % by weight.
7464. The method of claim 7286, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
7465. The method of claim 7286, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
7466. The method of claim 7286, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
7467. The method of claim 7286, wherein the device has a coating, and wherein the coating further comprises a polymer.
7468. The method of claim 7286, wherein the device has a first coating having a first composition and a second coating having a second composition.
7469. The method of claim 7286, wherein the device has a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
7470. The method of claim 7286, wherein the composition comprises a polymer.
7471. The method of claim 7286, wherein the composition comprises a polymeric carrier.
7472. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a copolymer.
7473. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a block copolymer.
7474. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a random copolymer.
7475. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a biodegradable polymer.
7476. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-biodegradable polymer.
7477. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophilic polymer.
7478. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophobic polymer.
7479. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophilic domains.
7480. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophobic domains.
7481. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-conductive polymer.
7482. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an elastomer.
7483. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrogel.
7484. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a silicone polymer.
7485. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrocarbon polymer.
7486. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a styrene-derived polymer.
7487. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a butadiene polymer.
7488. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a macromer.
7489. The method of claim 7286, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
7490. The method of claim 7286 wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an amorphous polymer.
7491. The method of claim 7286, wherein the device comprises a lubricious coating.
7492. The method of claim 7286 wherein the anti-scarring agent is located within pores or holes of the device.
7493. The method of claim 7286 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
7494. The method of claim 7286, wherein the device comprises a second pharmaceutically active agent.
7495. The method of claim 7286 wherein the device comprises an anti-inflammatory agent.
7496. The method of claim 7286 wherein the device comprises an agent that inhibits infection.
7497. The method of claim 7286 wherein the device comprises an agent that inhibits infection, and wherein the agent is an anthracycline.
7498. The method of claim 7286 wherein the device comprises an agent that inhibits infection, and wherein the agent is doxorubicin.
7499. The method of claim 7286 wherein the device comprises an agent that inhibits infection, and wherein the agent is mitoxantrone.
7500. The method of claim 7286 wherein the device comprises an agent that inhibits infection, and wherein the agent is a fluoropyrimidine.
7501. The method of claim 7286 wherein the device comprises an agent that inhibits infection, and wherein the agent is 5-fluorouracil (5-FU).
7502. The method of claim 7286 wherein the device comprises an agent that inhibits infection, and wherein the agent is a folic acid antagonist.
7503. The method of claim 7286 wherein the device comprises an agent that inhibits infection, and wherein the agent is methotrexate.
7504. The method of claim 7286 wherein the device comprises an agent that inhibits infection, and wherein the agent is a podophylotoxin.
7505. The method of claim 7286 wherein the device comprises an agent that inhibits infection, and wherein the agent is etoposide.
7506. The method of claim 7286 wherein the device comprises an agent that inhibits infection, and wherein the agent is a camptothecin.
7507. The method of claim 7286 wherein the device comprises an agent that inhibits infection, and wherein the agent is a hydroxyurea.
7508. The method of claim 7286 wherein the device comprises an agent that inhibits infection, and wherein the agent is a platinum complex.
7509. The method of claim 7286 wherein the device comprises an agent that inhibits infection, and wherein the agent is cisplatin.
7510. The method of claim 7286, further comprising an anti- thrombotic agent.
7511. The method of claim 7286 wherein the device comprises a visualization agent.
7512. The method of claim 7286 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
7513. The method of claim 7286 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises barium, tantalum, or technetium.
7514. The method of claim 7286 wherein the device comprises a visualization agent, and wherein the visualization agent is a MRI responsive material.
7515. The method of claim 7286 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a gadolinium chelate.
7516. The method of claim 7286 wherein the device comprises a visualization agent, and wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
7517. The method of claim 7286 wherein the device comprises a visualization agent, and wherein the visualization agent comprises an iron oxide compound.
7518. The method of claim 7286 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a dye, pigment, or colorant.
7519. The method of claim 7286 wherein the device comprises an echogenic material.
7520. The method of claim 7286 wherein the device comprises an echogenic material, and wherein the echogenic material is in the form of a coating.
7521. The method of claim 7286 wherein the device is sterile.
7522. The method of claim 7286 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
7523. The method of claim 7286 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is connective tissue.
7524. The method of claim 7286 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is muscle tissue.
7525. The method of claim 7286 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is nerve tissue.
7526. The method of claim 7286 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is epithelium tissue.
7527. The method of claim 7286 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
7528. The method of claim 7286 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
7529. The method of claim 7286 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
7530. The method of claim 7286 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
7531. The method of claim 7286 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
7532. The method of claim 7286 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
7533. The method of claim 7286 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
7534. The method of claim 7286 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
7535. The method of claim 7286 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
7536. The method of claim 7286 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
7537. The method of claim 7286 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
7538. The method of claim 7286 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
7539. The method of claim 7286 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
7540. The method of claim 7286 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
7541. The method of claim 7286 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
7542. The method of claim 7286 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
7543. The method of claim 7286 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
7544. The method of claim 7286 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
7545. The method of claim 7286 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
7546. The method of claim 7286 wherein the combining is performed by direct affixing the agent or the composition to the sensor.
7547. The method of claim 7286 wherein the combining is performed by spraying the agent or the component onto the sensor.
7548. The method of claim 7286 wherein the combining is performed by electrospraying the agent or the composition onto the sensor.
7549. The method of claim 7286 wherein the combining is performed by dipping the sensor into a solution comprising the agent or the composition.
7550. The method of claim 7286 wherein the combining is performed by covalently attaching the agent or the composition to the sensor.
7551. The method of claim 7286 wherein the combining is performed by non-covalently attaching the agent or the composition to the sensor.
7552. The method of claim 7286 wherein the combining is performed by coating the sensor with a substance that contains the agent or the composition.
7553. The method of claim 7286 wherein the combining is performed by coating the sensor with a substance that absorbs the agent.
7554. The method of claim 7286 wherein the combining is performed by interweaving a thread composed of, or coated with, the agent or the composition.
7555. The method of claim 7286 wherein the combining is performed by completely covering the sensor with a sleeve that contains the agent or the composition.
7556. The method of claim 7286 wherein the combining is performed by covering a portion of the sensor with a sleeve that contains the agent or the composition.
7557. The method of claim 7286 wherein the combining is performed by completely covering the sensor with a cover that contains the agent or the composition.
7558. The method of claim 7286 wherein the combining is performed by covering a portion of the sensor with a cover that contains the agent or the composition.
7559. The method of claim 7286 wherein the combining is performed by completely covering the sensor with an electrospun fabric that contains the agent or the composition.
7560. The method of claim 7286 wherein the combining is performed by covering a portion of the sensor with an electrospun fabric that contains the agent or the composition.
7561. The method of claim 7286 wherein the combining is performed by completely covering the sensor with a mesh that contains the agent or the composition.
7562. The method of claim 7286 wherein the combining is performed by covering a portion of the sensor with a mesh that contains the agent or the composition.
7563. The method of claim 7286 wherein the combining is performed by constructing a portion of the sensor with the agent or the composition.
7564. The method of claim 7286 wherein the combining is performed by impregnating the sensor with the agent or the composition.
7565. The method of claim 7286 wherein the combining is performed by constructing a portion of the sensor from a degradable polymer that releases the agent.
7566. The method of claim 7286 wherein the combining is performed by dipping the sensor into a solution that comprise the agent and an inert solvent for the sensor.
7567. The method of claim 7286 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
7568. The method of claim 7286 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
7569. The method of claim 7286 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
7570. The method of claim 7286 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
7571. The method of claim 7286 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
7572. The method of claim 7286 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and an inert solvent for the sensor.
7573. The method of claim 7286 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
7574. The method of claim 7286 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
7575. The method of claim 7286 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
7576. The method of claim 7286 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
7577. The method of claim 7286 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
7578. The method of any one of claims 7286-7577 wherein the sensor is a blood or tissue glucose monitor.
7579. The method of any one of claims 7286-7577 wherein the sensor is an electrolyte sensor.
7580. The method of any one of claims 7286-7577 wherein the sensor is a blood constituent sensor.
7581. The method of any one of claims 7286-7577 wherein the sensor is a temperature sensor.
7582. The method of any one of claims 7286-7577 wherein the sensor is a pH sensor.
7583. The method of any one of claims 7286-7577 wherein the sensor is an optical sensor.
7584. The method of any one of claims 7286-7577 wherein the sensor is an amperometric sensor.
7585. The method of any one of claims 7286-7577 wherein the sensor is a pressure sensor.
7586. The method of any one of claims 7286-7577 wherein the sensor is a biosensor.
7587. The method of any one of claims 7286-7577 wherein the sensor is a sensing transponder.
7588. The method of any one of claims 7286-7577 wherein the sensor is a strain sensor.
7589. The method of any one of claims 7286-7577 wherein the sensor is a magnetoresistive sensor.
7590. The method of any one of claims 7286-7577 wherein the sensor is a cardiac sensor.
7591. The method of any one of claims 7286-7577 wherein the sensor is a respiratory sensor.
7592. The method of any one of claims 7286-7577 wherein the sensor is an auditory sensor.
7593. The method of any one of claims 7286-7577 wherein the sensor is a metabolite sensor.
7594. The method of any one of claims 7286-7577 wherein the sensor detects mechanical changes.
7595. The method of any one of claims 7286-7577 wherein the sensor detects physical changes.
7596. The method of any one of claims 7286-7577 wherein the sensor detects electrochemical changes.
7597. The method of any one of claims 7286-7577 wherein the sensor detects magnetic changes.
7598. The method of any one of claims 7286-7577 wherein the sensor detects acceleration changes.
7599. The method of any one of claims 7286-7577 wherein the sensor detects ionizing radiation changes.
7600. The method of any one of claims 7286-7577 wherein the sensor detects acoustic wave changes.
7601. The method of any one of claims 7286-7577 wherein the sensor detects chemical changes.
7602. The method of any one of claims 7286-7577 wherein the sensor detects drug concentration changes.
7603. The method of any one of claims 7286-7577 wherein the sensor detects hormone changes.
7604. The method of any one of claims 7286-7577 wherein the sensor detects barometric changes.
7605. A method for making a device comprising: combining a blood or tissue glucose monitor (i.e., a sensor) and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
7606. The method of claim 7605 wherein the agent inhibits cell regeneration.
7607. The method of claim 7605 wherein the agent inhibits angiogenesis.
7608. The method of claim 7605 wherein the agent inhibits fibroblast migration.
7609. The method of claim 7605 wherein the agent inhibits fibroblast proliferation.
7610. The method of claim 7605 wherein the agent inhibits deposition of extracellular matrix.
7611. The method of claim 7605 wherein the agent inhibits tissue remodeling.
7612. The method of claim 7605 wherein the agent is an angiogenesis inhibitor.
7613. The method of claim 7605 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
7614. The method of claim 7605 wherein the agent is a chemokine receptor antagonist.
7615. The method of claim 7605 wherein the agent is a cell cycle inhibitor.
7616. The method of claim 7605 wherein the agent is a taxane.
7617. The method of claim 7605 wherein the agent is an antimicrotubule agent.
7618. The method of claim 7605 wherein the agent is paclitaxel.
7619. The method of claim 7605 wherein the agent is not paclitaxel.
7620. The method of claim 7605 wherein the agent is an analogue or derivative of paclitaxel.
7621. The method of claim 7605 wherein the agent is a vinca alkaloid.
7622. The method of claim 7605 wherein the agent is camptothecin or an analogue or derivative thereof.
7623. The method of claim 7605 wherein the agent is a podophyllotoxin.
7624. The method of claim 7605 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
7625. The method of claim 7605 wherein the agent is an anthracycline.
7626. The method of claim 7605 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
7627. The method of claim 7605 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
7628. The method of claim 7605 wherein the agent is a platinum compound.
7629. The method of claim 7605 wherein the agent is a nitrosourea.
7630. The method of claim 7605 wherein the agent is a nitroimidazole.
7631. The method of claim 7605 wherein the agent is a folic acid antagonist
7632. The method of claim 7605 wherein the agent is a cytidine analogue.
7633. The method of claim 7605 wherein the agent is a pyrimidine analogue.
7634. The method of claim 7605 wherein the agent is a fluoropyrimidine analogue.
7635. The method of claim 7605 wherein the agent is a purine analogue.
7636. The method of claim 7605 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
7637. The method of claim 7605 wherein the agent is a hydroxyurea.
7638. The method of claim 7605 wherein the agent is a mytomicin or an analogue or derivative thereof.
7639. The method of claim 7605 wherein the agent is an alkyl sulfonate.
7640. The method of claim 7605 wherein the agent is a benzamide or an analogue or derivative thereof.
7641. The method of claim 7605 wherein the agent is a nicotinamide or an analogue or derivative thereof.
7642. The method of claim 7605 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
7643. The method of claim 7605 wherein the agent is a DNA alkylating agent
7644. The method of claim 7605 wherein the agent is an antimicrotubule agent
7645. The method of claim 7605 wherein the agent is a topoisomerase inhibitor.
7646. The method of claim 7605 wherein the agent is a DNA cleaving agent.
7647. The method of claim 7605 wherein the agent is an antimetabolite.
7648. The method of claim 7605 wherein the agent inhibits adenosine deaminase.
7649. The method of claim 7605 wherein the agent inhibits purine ring synthesis.
7650. The method of claim 7605 wherein the agent is a nucleotide interconversion inhibitor.
7651. The method of claim 7605 wherein the agent inhibits dihydrofolate reduction.
7652. The method of claim 7605 wherein the agent blocks thymidine monophosphate.
7653. The method of claim 7605 wherein the agent causes DNA damage.
7654. The method of claim 7605 wherein the agent is a DNA intercalation agent.
7655. The method of claim 7605 wherein the agent is a RNA synthesis inhibitor.
7656. The method of claim 7605 wherein the agent is a pyrimidine synthesis inhibitor.
7657. The method of claim 7605 wherein the agent inhibits ribonucleotide synthesis or function.
7658. The method of claim 7605 wherein the agent inhibits thymidine monophosphate synthesis or function.
7659. The method of claim 7605 wherein the agent inhibits DNA synthesis.
7660. The method of claim 7605 wherein the agent causes DNA adduct formation.
7661. The method of claim 7605 wherein the agent inhibits protein synthesis.
7662. The method of claim 7605 wherein the agent inhibits microtubule function.
7663. The method of claim 7605 wherein the agent is a cyclin dependent protein kinase inhibitor.
7664. The method of claim 7605 wherein the agent is an epidermal growth factor kinase inhibitor.
7665. The method of claim 7605 wherein the agent is an elastase inhibitor.
7666. The method of claim 7605 wherein the agent is a factor Xa inhibitor.
7667. The method of claim 7605 wherein the agent is a farnesyltransferase inhibitor.
7668. The method of claim 7605 wherein the agent is a fibrinogen antagonist.
7669. The method of claim 7605 wherein the agent is a guanylate cyclase stimulant.
7670. The method of claim 7605 wherein the agent is a heat shock protein 90 antagonist.
7671. The method of claim 7605 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
7672. The method of claim 7605 wherein the agent is a guanylate cyclase stimulant.
7673. The method of claim 7605 wherein the agent is a HMGCoA reductase inhibitor.
7674. The method of claim 7605 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
7675. The method of claim 7605 wherein the agent is a hydroorotate dehydrogenase inhibitor.
7676. The method of claim 7605 wherein the agent is an IKK2 inhibitor.
7677. The method of claim 7605 wherein the agent is an IL-1 antagonist.
7678. The method of claim 7605 wherein the agent is an ICE antagonist
7679. The method of claim 7605 wherein the agent is an IRAK antagonist.
7680. The method of claim 7605 wherein the agent is an IL-4 agonist.
7681. The method of claim 7605 wherein the agent is an immunomodulatory agent.
7682. The method of claim 7605 wherein the agent is sirolimus or an analogue or derivative thereof.
7683. The method of claim 7605 wherein the agent is not sirolimus.
7684. The method of claim 7605 wherein the agent is everolimus or an analogue or derivative thereof.
7685. The method of claim 7605 wherein the agent is tacrolimus or an analogue or derivative thereof.
7686. The method of claim 7605 wherein the agent is not tacrolimus.
7687. The method of claim 7605 wherein the agent is biolmus or an analogue or derivative thereof.
7688. The method of claim 7605 wherein the agent is tresperimus or an analogue or derivative thereof.
7689. The method of claim 7605 wherein the agent is auranofin or an analogue or derivative thereof.
7690. The method of claim 7605 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
7691. The method of claim 7605 wherein the agent is gusperimus or an analogue or derivative thereof.
7692. The method of claim 7605 wherein the agent is pimecrolimus or an analogue or derivative thereof.
7693. The method of claim 7605 wherein the agent is ABT-578 or an analogue or derivative thereof.
7694. The method of claim 7605 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
7695. The method of claim 7605 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
7696. The method of claim 7605 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
7697. The method of claim 7605 wherein the agent is a leukotriene inhibitor.
7698. The method of claim 7605 wherein the agent is a MCP-1 antagonist.
7699. The method of claim 7605 wherein the agent is a MMP inhibitor.
7700. The method of claim 7605 wherein the agent is an NF kappa B inhibitor.
7701. The method of claim 7605 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
7702. The method of claim 7605 wherein the agent is an NO antagonist.
7703. The method of claim 7605 wherein the agent is a p38 MAP kinase inhibitor.
7704. The method of claim 7605 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
7705. The method of claim 7605 wherein the agent is a phosphodiesterase inhibitor.
7706. The method of claim 7605 wherein the agent is a TGF beta inhibitor.
7707. The method of claim 7605 wherein the agent is a thromboxane A2 antagonist.
7708. The method of claim 7605 wherein the agent is a TNF alpha antagonist.
7709. The method of claim 7605 wherein the agent is a TACE inhibitor.
7710. The method of claim 7605 wherein the agent is a tyrosine kinase inhibitor.
7711. The method of claim 7605 wherein the agent is a vitronectin inhibitor.
7712. The method of claim 7605 wherein the agent is a fibroblast growth factor inhibitor.
7713. The method of claim 7605 wherein the agent is a protein kinase inhibitor.
7714. The method of claim 7605 wherein the agent is a PDGF receptor kinase inhibitor.
7715. The method of claim 7605 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
7716. The method of claim 7605 wherein the agent is a retinoic acid receptor antagonist.
7717. The method of claim 7605 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
7718. The method of claim 7605 wherein the agent is a fibrinogen antagonist.
7719. The method of claim 7605 wherein the agent is an antimycotic agent.
7720. The method of claim 7605 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
7721. The method of claim 7605 wherein the agent is a bisphosphonate.
7722. The method of claim 7605 wherein the agent is a phospholipase A1 inhibitor.
7723. The method of claim 7605 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
7724. The method of claim 7605 wherein the agent is a macrolide antibiotic.
7725. The method of claim 7605 wherein the agent is a GPIIb/llla receptor antagonist.
7726. The method of claim 7605 wherein the agent is an endothelin receptor antagonist.
7727. The method of claim 7605 wherein the agent is a peroxisome proliferator-activated receptor agonist.
7728. The method of claim 7605 wherein the agent is an estrogen receptor agent.
7729. The method of claim 7605 wherein the agent is a somastostatin analogue.
7730. The method of claim 7605 wherein the agent is a neurokinin 1 antagonist.
7731. The method of claim 7605 wherein the agent is a neurokinin 3 antagonist.
7732. The method of claim 7605 wherein the agent is a VLA-4 antagonist.
7733. The method of claim 7605 wherein the agent is an osteoclast inhibitor.
7734. The method of claim 7605 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
7735. The method of claim 7605 wherein the agent is an angiotensin I converting enzyme inhibitor.
7736. The method of claim 7605 wherein the agent is an angiotensin II antagonist.
7737. The method of claim 7605 wherein the agent is an enkephalinase inhibitor.
7738. The method of claim 7605 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
7739. The method of claim 7605 wherein the agent is a protein kinase C inhibitor.
7740. The method of claim 7605 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
7741. The method of claim 7605 wherein the agent is a CXCR3 inhibitor.
7742. The method of claim 7605 wherein the agent is an Itk inhibitor.
7743. The method of claim 7605 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
7744. The method of claim 7605 wherein the agent is a PPAR agonist.
7745. The method of claim 7605 wherein the agent is an immunosuppressant.
7746. The method of claim 7605 wherein the agent is an Erb inhibitor.
7747. The method of claim 7605 wherein the agent is an apoptosis agonist.
7748. The method of claim 7605 wherein the agent is a lipocortin agonist.
7749. The method of claim 7605 wherein the agent is a VCAM-1 antagonist.
7750. The method of claim 7605 wherein the agent is a collagen antagonist.
7751. The method of claim 7605 wherein the agent is an alpha 2 integrin antagonist.
7752. The method of claim 7605 wherein the agent is a TNF alpha inhibitor.
7753. The method of claim 7605 wherein the agent is a nitric oxide inhibitor
7754. The method of claim 7605 wherein the agent is a cathepsin inhibitor.
7755. The method of claim 7605 wherein the agent is not an anti- inflammatory agent.
7756. The method of claim 7605 wherein the agent is not a steroid.
7757. The method of claim 7605 wherein the agent is not a glucocorticosteroid.
7758. The method of claim 7605 wherein the agent is not dexamethasone.
7759. The method of claim 7605 wherein the agent is not beclomethasone.
7760. The method of claim 7605 wherein the agent is not dipropionate.
7761. The method of claim 7605 wherein the agent is not an anti- infective agent.
7762. The method of claim 7605 wherein the agent is not an antibiotic.
7763. The method of claim 7605 wherein the agent is not an anti- fungal agent.
7764. The method of claim 7605, wherein the composition comprises a polymer.
7765. The method of claim 7605, wherein the composition comprises a polymeric carrier.
7766. The method of claim 7605 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
7767. The method of claim 7605 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
7768. The method of claim 7605 wherein the device has a coating that comprises the anti-scarring agent.
7769. The method of claim 7605, wherein the device has a coating that comprises the agent and is disposed on a surface of the sensor.
7770. The method of claim 7605, wherein the device has a coating that comprises the agent and directly contacts the sensor.
7771. The method of claim 7605, wherein the device has a coating that comprises the agent and indirectly contacts the sensor.
7772. The method of claim 7605, wherein the device has a coating that comprises the agent and partially covers the sensor.
7773. The method of claim 7605, wherein the device has a coating that comprises the agent and completely covers the sensor.
7774. The method of claim 7605, wherein the device has a uniform coating.
7775. The method of claim 7605, wherein the device has a non- uniform coating.
7776. The method of claim 7605, wherein the device has a discontinuous coating.
7777. The method of claim 7605, wherein the device has a patterned coating.
7778. The method of claim 7605, wherein the device has a coating with a thickness of 100 μm or less.
7779. The method of claim 7605, wherein the device has a coating with a thickness of 10 μm or less.
7780. The method of claim 7605, wherein the device has a coating, and the coating adheres to the surface of the sensor upon deployment of the sensor.
7781. The method of claim 7605, wherein the device has a coating, and wherein the coating is stable at room temperature for a period of 1 year.
7782. The method of claim 7605, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1% by weight
7783. The method of claim 7605, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
7784. The method of claim 7605, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
7785. The method of claim 7605, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
7786. The method of claim 7605, wherein the device has a coating, and wherein the coating further comprises a polymer.
7787. The method of claim 7605, wherein the device has a first coating having a first composition and a second coating having a second composition.
7788. The method of claim 7605, wherein the device has a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
7789. The method of claim 7605, wherein the composition comprises a polymer.
7790. The method of claim 7605, wherein the composition comprises a polymeric carrier.
7791. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a copolymer.
7792. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a block copolymer.
7793. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a random copolymer.
7794. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a biodegradable polymer.
7795. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-biodegradable polymer.
7796. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophilic polymer.
7797. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophobic polymer.
7798. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophilic domains.
7799. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophobic domains.
7800. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-conductive polymer.
7801. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an elastomer.
7802. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrogel.
7803. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a silicone polymer.
7804. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrocarbon polymer.
7805. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a styrene-derived polymer.
7806. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a butadiene polymer.
7807. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a macromer.
7808. The method of claim 7605, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a poly( ethylene glycol) polymer.
7809. The method of claim 7605 wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an amorphous polymer.
7810. The method of claim 7605, wherein the device comprises a lubricious coating.
7811. The method of claim 7605 wherein the anti-scarring agent is located within pores or holes of the device.
7812. The method of claim 7605 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
7813. The method of claim 7605, wherein the device comprises a second pharmaceutically active agent
7814. The method of claim 7605 wherein the device comprises an anti-inflammatory agent.
7815. The method of claim 7605 wherein the device comprises an agent that inhibits infection.
7816. The method of claim 7605 wherein the device comprises an agent that inhibits infection, and wherein the agent is an anthracycline.
7817. The method of claim 7605 wherein the device comprises an agent that inhibits infection, and wherein the agent is doxorubicin.
7818. The method of claim 7605 wherein the device comprises an agent that inhibits infection, and wherein the agent is mitoxantrone.
7819. The method of claim 7605 wherein the device comprises an agent that inhibits infection, and wherein the agent is a fluoropyrimidine.
7820. The method of claim 7605 wherein the device comprises an agent that inhibits infection, and wherein the agent is 5-fluorouracil (5-FU).
7821. The method of claim 7605 wherein the device comprises an agent that inhibits infection, and wherein the agent is a folic acid antagonist.
7822. The method of claim 7605 wherein the device comprises an agent that inhibits infection, and wherein the agent is methotrexate.
7823. The method of claim 7605 wherein the device comprises an agent that inhibits infection, and wherein the agent is a podophylotoxin.
7824. The method of claim 7605 wherein the device comprises an agent that inhibits infection, and wherein the agent is etoposide.
7825. The method of claim 7605 wherein the device comprises an agent that inhibits infection, and wherein the agent is a camptothecin.
7826. The method of claim 7605 wherein the device comprises an agent that inhibits infection, and wherein the agent is a hydroxyurea.
7827. The method of claim 7605 wherein the device comprises an agent that inhibits infection, and wherein the agent is a platinum complex.
7828. The method of claim 7605 wherein the device comprises an agent that inhibits infection, and wherein the agent is cisplatin.
7829. The method of claim 7605, further comprising an anti- thrombotic agent.
7830. The method of claim 7605 wherein the device comprises a visualization agent.
7831. The method of claim 7605 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
7832. The method of claim 7605 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises barium, tantalum, or technetium.
7833. The method of claim 7605 wherein the device comprises a visualization agent, and wherein the visualization agent is a MRI responsive material.
7834. The method of claim 7605 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a gadolinium chelate.
7835. The method of claim 7605 wherein the device comprises a visualization agent, and wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
7836. The method of claim 7605 wherein the device comprises a visualization agent, and wherein the visualization agent comprises an iron oxide compound.
7837. The method of claim 7605 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a dye, pigment, or colorant.
7838. The method of claim 7605 wherein the device comprises an echogenic material.
7839. The method of claim 7605 wherein the device comprises an echogenic material, and wherein the echogenic material is in the form of a coating.
7840. The method of claim 7605 wherein the device is sterile.
7841. The method of claim 7605 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
7842. The method of claim 7605 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is connective tissue.
7843. The method of claim 7605 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is muscle tissue.
7844. The method of claim 7605 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is nerve tissue.
7845. The method of claim 7605 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is epithelium tissue.
7846. The method of claim 7605 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
7847. The method of claim 7605 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
7848. The method of claim 7605 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
7849. The method of claim 7605 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
7850. The method of claim 7605 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
7851. The method of claim 7605 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
7852. The method of claim 7605 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
7853. The method of claim 7605 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
7854. The method of claim 7605 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
7855. The method of claim 7605 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
7856. The method of claim 7605 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
7857. The method of claim 7605 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
7858. The method of claim 7605 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
7859. The method of claim 7605 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
7860. The method of claim 7605 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
7861. The method of claim 7605 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
7862. The method of claim 7605 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
7863. The method of claim 7605 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
7864. The method of claim 7605 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
7865. The method of claim 7605 wherein the combining is performed by direct affixing the agent or the composition to the sensor.
7866. The method of claim 7605 wherein the combining is performed by spraying the agent or the component onto the sensor.
7867. The method of claim 7605 wherein the combining is performed by electrospraying the agent or the composition onto the sensor.
7868. The method of claim 7605 wherein the combining is performed by dipping the sensor into a solution comprising the agent or the composition.
7869. The method of claim 7605 wherein the combining is performed by covalently attaching the agent or the composition to the sensor.
7870. The method of claim 7605 wherein the combining is performed by non-covalently attaching the agent or the composition to the sensor.
7871. The method of claim 7605 wherein the combining is performed by coating the sensor with a substance that contains the agent or the composition.
7872. The method of claim 7605 wherein the combining is performed by coating the sensor with a substance that absorbs the agent.
7873. The method of claim 7605 wherein the combining is performed by interweaving a thread composed of, or coated with, the agent or the composition.
7874. The method of claim 7605 wherein the combining is performed by completely covering the sensor with a sleeve that contains the agent or the composition.
7875. The method of claim 7605 wherein the combining is performed by covering a portion of the sensor with a sleeve that contains the agent or the composition.
7876. The method of claim 7605 wherein the combining is performed by completely covering the sensor with a cover that contains the agent or the composition.
7877. The method of claim 7605 wherein the combining is performed by covering a portion of the sensor with a cover that contains the agent or the composition.
7878. The method of claim 7605 wherein the combining is performed by completely covering the sensor with an electrospun fabric that contains the agent or the composition.
7879. The method of claim 7605 wherein the combining is performed by covering a portion of the sensor with an electrospun fabric that contains the agent or the composition.
7880. The method of claim 7605 wherein the combining is performed by completely covering the sensor with a mesh that contains the agent or the composition.
7881. The method of claim 7605 wherein the combining is performed by covering a portion of the sensor with a mesh that contains the agent or the composition.
7882. The method of claim 7605 wherein the combining is performed by constructing a portion of the sensor with the agent or the composition.
7883. The method of claim 7605 wherein the combining is performed by impregnating the sensor with the agent or the composition.
7884. The method of claim 7605 wherein the combining is performed by constructing a portion of the sensor from a degradable polymer that releases the agent.
7885. The method of claim 7605 wherein the combining is performed by dipping the sensor into a solution that comprise the agent and an inert solvent for the sensor.
7886. The method of claim 7605 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
7887. The method of claim 7605 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
7888. The method of claim 7605 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
7889. The method of claim 7605 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
7890. The method of claim 7605 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
7891. The method of claim 7605 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and an inert solvent for the sensor.
7892. The method of claim 7605 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
7893. The method of claim 7605 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
7894. The method of claim 7605 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
7895. The method of claim 7605 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
7896. The method of claim 7605 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
7897. The method of any one of claims 7605-7896 wherein the device is deliverable to the vascular system transluminally using a catheter on a stent platform.
7898. The method of any one of claims 7605-7896 wherein the device is composed of glucose sensitive living cells that monitor blood glucose levels and produce a detectable electrical or optical signal in response to changes in glucose concentrations.
7899. The method of any one of claims 7605-7896 wherein the device is an electrode composed of an analyte responsive enzyme.
7900. The method of any one of claims 7605-7896 wherein the device is a closed loop insulin delivery system that comprises a sensing means that detects the host's blood glucose level and stimulates an insulin pump to supply insulin.
7901. The method of any one of claims 7605-7896 wherein the device is a closed loop insulin delivery system that comprises a sensing means that detects the host's blood glucose level and stimulates the pancreas to supply insulin.
7902. A method for making a device comprising: combining a pressure or stress sensor and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
7903. The method of claim 7902 wherein the agent inhibits cell regeneration.
7904. The method of claim 7902 wherein the agent inhibits angiogenesis.
7905. The method of claim 7902 wherein the agent inhibits fibroblast migration.
7906. The method of claim 7902 wherein the agent inhibits fibroblast proliferation.
7907. The method of claim 7902 wherein the agent inhibits deposition of extracellular matrix.
7908. The method of claim 7902 wherein the agent inhibits tissue remodeling.
7909. The method of claim 7902 wherein the agent is an angiogenesis inhibitor.
7910. The method of claim 7902 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
7911. The method of claim 7902 wherein the agent is a chemokine receptor antagonist.
7912. The method of claim 7902 wherein the agent is a cell cycle inhibitor.
7913. The method of claim 7902 wherein the agent is a taxane.
7914. The method of claim 7902 wherein the agent is an antimicrotubule agent.
7915. The method of claim 7902 wherein the agent is paclitaxel.
7916. The method of claim 7902 wherein the agent is not paclitaxel.
7917. The method of claim 7902 wherein the agent is an analogue or derivative of paclitaxel.
7918. The method of claim 7902 wherein the agent is a vinca alkaloid.
7919. The method of claim 7902 wherein the agent is camptothecin or an analogue or derivative thereof.
7920. The method of claim 7902 wherein the agent is a podophyllotoxin.
7921. The method of claim 7902 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
7922. The method of claim 7902 wherein the agent is an anthracycline.
7923. The method of claim 7902 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
7924. The method of claim 7902 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
7925. The method of claim 7902 wherein the agent is a platinum compound.
7926. The method of claim 7902 wherein the agent is a nitrosourea.
7927. The method of claim 7902 wherein the agent is a nitroimidazole.
7928. The method of claim 7902 wherein the agent is a folic acid antagonist.
7929. The method of claim 7902 wherein the agent is a cytidine analogue.
7930. The method of claim 7902 wherein the agent is a pyrimidine analogue.
7931. The method of claim 7902 wherein the agent is a fluoropyrimidine analogue.
7932. The method of claim 7902 wherein the agent is a purine analogue.
7933. The method of claim 7902 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
7934. The method of claim 7902 wherein the agent is a hydroxyurea.
7935. The method of claim 7902 wherein the agent is a mytomicin or an analogue or derivative thereof.
7936. The method of claim 7902 wherein the agent is an alkyl sulfonate.
7937. The method of claim 7902 wherein the agent is a benzamide or an analogue or derivative thereof.
7938. The method of claim 7902 wherein the agent is a nicotinamide or an analogue or derivative thereof.
7939. The method of claim 7902 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
7940. The method of claim 7902 wherein the agent is a DNA alkylating agent
7941. The method of claim 7902 wherein the agent is an antimicrotubule agent.
7942. The method of claim 7902 wherein the agent is a topoisomerase inhibitor.
7943. The method of claim 7902 wherein the agent is a DNA cleaving agent
7944. The method of claim 7902 wherein the agent is an antimetabolite.
7945. The method of claim 7902 wherein the agent inhibits adenosine deaminase.
7946. The method of claim 7902 wherein the agent inhibits purine ring synthesis.
7947. The method of claim 7902 wherein the agent is a nucleotide interconversion inhibitor.
7948. The method of claim 7902 wherein the agent inhibits dihydrofolate reduction.
7949. The method of claim 7902 wherein the agent blocks thymidine monophosphate.
7950. The method of claim 7902 wherein the agent causes DNA damage.
7951. The method of claim 7902 wherein the agent is a DNA intercalation agent.
7952. The method of claim 7902 wherein the agent is a RNA synthesis inhibitor.
7953. The method of claim 7902 wherein the agent is a pyrimidine synthesis inhibitor.
7954. The method of claim 7902 wherein the agent inhibits ribonucleotide synthesis or function.
7955. The method of claim 7902 wherein the agent inhibits thymidine monophosphate synthesis or function.
7956. The method of claim 7902 wherein the agent inhibits DNA synthesis.
7957. The method of claim 7902 wherein the agent causes DNA adduct formation.
7958. The method of claim 7902 wherein the agent inhibits protein synthesis.
7959. The method of claim 7902 wherein the agent inhibits microtubule function.
7960. The method of claim 7902 wherein the agent is a cyclin dependent protein kinase inhibitor.
7961. The method of claim 7902 wherein the agent is an epidermal growth factor kinase inhibitor.
7962. The method of claim 7902 wherein the agent is an elastase inhibitor.
7963. The method of claim 7902 wherein the agent is a factor Xa inhibitor.
7964. The method of claim 7902 wherein the agent is a farnesyltransferase inhibitor.
7965. The method of claim 7902 wherein the agent is a fibrinogen antagonist.
7966. The method of claim 7902 wherein the agent is a guanylate cyclase stimulant
7967. The method of claim 7902 wherein the agent is a heat shock protein 90 antagonist.
7968. The method of claim 7902 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
7969. The method of claim 7902 wherein the agent is a guanylate cyclase stimulant
7970. The method of claim 7902 wherein the agent is a HMGCoA reductase inhibitor.
7971. The method of claim 7902 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
7972. The method of claim 7902 wherein the agent is a hydroorotate dehydrogenase inhibitor.
7973. The method of claim 7902 wherein the agent is an IKK2 inhibitor.
7974. The method of claim 7902 wherein the agent is an IL-1 antagonist.
7975. The method of claim 7902 wherein the agent is an ICE antagonist.
7976. The method of claim 7902 wherein the agent is an IRAK antagonist.
7977. The method of claim 7902 wherein the agent is an IL-4 agonist.
7978. The method of claim 7902 wherein the agent is an immunomodulatory agent.
7979. The method of claim 7902 wherein the agent is sirolimus or an analogue or derivative thereof.
7980. The method of claim 7902 wherein the agent is not sirolimus.
7981. The method of claim 7902 wherein the agent is everolimus or an analogue or derivative thereof.
7982. The method of claim 7902 wherein the agent is tacrolimus or an analogue or derivative thereof.
7983. The method of claim 7902 wherein the agent is not tacrolimus.
7984. The method of claim 7902 wherein the agent is biolmus or an analogue or derivative thereof.
7985. The method of claim 7902 wherein the agent is tresperimus or an analogue or derivative thereof.
7986. The method of claim 7902 wherein the agent is auranofin or an analogue or derivative thereof.
7987. The method of claim 7902 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
7988. The method of claim 7902 wherein the agent is gusperimus or an analogue or derivative thereof.
7989. The method of claim 7902 wherein the agent is pimecrolimus or an analogue or derivative thereof.
7990. The method of claim 7902 wherein the agent is ABT-578 or an analogue or derivative thereof.
7991. The method of claim 7902 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
7992. The method of claim 7902 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
7993. The method of claim 7902 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
7994. The method of claim 7902 wherein the agent is a leukotriene inhibitor.
7995. The method of claim 7902 wherein the agent is a MCP-1 antagonist
7996. The method of claim 7902 wherein the agent is a MMP inhibitor.
7997. The method of claim 7902 wherein the agent is an NF kappa B inhibitor.
7998. The method of claim 7902 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
7999. The method of claim 7902 wherein the agent is an NO antagonist
8000. The method of claim 7902 wherein the agent is a p38 MAP kinase inhibitor.
8001. The method of claim 7902 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
8002. The method of claim 7902 wherein the agent is a phosphodiesterase inhibitor.
8003. The method of claim 7902 wherein the agent is a TGF beta inhibitor.
8004. The method of claim 7902 wherein the agent is a thromboxane A2 antagonist.
8005. The method of claim 7902 wherein the agent is a TNF alpha antagonist.
8006. The method of claim 7902 wherein the agent is a TACE inhibitor.
8007. The method of claim 7902 wherein the agent is a tyrosine kinase inhibitor.
8008. The method of claim 7902 wherein the agent is a vitronectin inhibitor.
8009. The method of claim 7902 wherein the agent is a fibroblast growth factor inhibitor.
8010. The method of claim 7902 wherein the agent is a protein kinase inhibitor.
8011. The method of claim 7902 wherein the agent is a PDGF receptor kinase inhibitor.
8012. The method of claim 7902 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
8013. The method of claim 7902 wherein the agent is a retinoic acid receptor antagonist.
8014. The method of claim 7902 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
8015. The method of claim 7902 wherein the agent is a fibrinogen antagonist.
8016. The method of claim 7902 wherein the agent is an antimycotic agent. I
8017. The method of claim 7902 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
8018. The method of claim 7902 wherein the agent is a bisphosphonate.
8019. The method of claim 7902 wherein the agent is a phospholipase A1 inhibitor.
8020. The method of claim 7902 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
8021. The method of claim 7902 wherein the agent is a macrolide antibiotic.
8022. The method of claim 7902 wherein the agent is a GPIIb/llla receptor antagonist.
8023. The method of claim 7902 wherein the agent is an endothelin receptor antagonist.
8024. The method of claim 7902 wherein the agent is a peroxisome proliferator-activated receptor agonist.
8025. The method of claim 7902 wherein the agent is an estrogen receptor agent.
8026. The method of claim 7902 wherein the agent is a somastostatin analogue.
8027. The method of claim 7902 wherein the agent is a neurokinin 1 antagonist.
8028. The method of claim 7902 wherein the agent is a neurokinin 3 antagonist.
8029. The method of claim 7902 wherein the agent is a VLA-4 antagonist.
8030. The method of claim 7902 wherein the agent is an osteoclast inhibitor.
8031. The method of claim 7902 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
8032. The method of claim 7902 wherein the agent is an angiotensin I converting enzyme inhibitor.
8033. The method of claim 7902 wherein the agent is an angiotensin II antagonist.
8034. The method of claim 7902 wherein the agent is an enkephalinase inhibitor.
8035. The method of claim 7902 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
8036. The method of claim 7902 wherein the agent is a protein kinase C inhibitor.
8037. The method of claim 7902 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
8038. The method of claim 7902 wherein the agent is a CXCR3 inhibitor.
8039. The method of claim 7902 wherein the agent is an Itk inhibitor.
8040. The method of claim 7902 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
8041. The method of claim 7902 wherein the agent is a PPAR agonist.
8042. The method of claim 7902 wherein the agent is an immunosuppressant.
8043. The method of claim 7902 wherein the agent is an Erb inhibitor.
8044. The method of claim 7902 wherein the agent is an apoptosis agonist.
8045. The method of claim 7902 wherein the agent is a lipocortin agonist.
8046. The method of claim 7902 wherein the agent is a VCAM-1 antagonist.
8047. The method of claim 7902 wherein the agent is a collagen antagonist.
8048. The method of claim 7902 wherein the agent is an alpha 2 integrin antagonist.
8049. The method of claim 7902 wherein the agent is a TNF alpha inhibitor.
8050. The method of claim 7902 wherein the agent is a nitric oxide inhibitor
8051. The method of claim 7902 wherein the agent is a cathepsin inhibitor.
8052. The method of claim 7902 wherein the agent is not an anti- inflammatory agent.
8053. The method of claim 7902 wherein the agent is not a steroid.
8054. The method of claim 7902 wherein the agent is not a glucocorticosteroid.
8055. The method of claim 7902 wherein the agent is not dexamethasone.
8056. The method of claim 7902 wherein the agent is not beclomethasone.
8057. The method of claim 7902 wherein the agent is not dipropionate.
8058. The method of claim 7902 wherein the agent is not an anti- infective agent.
8059. The method of claim 7902 wherein the agent is not an antibiotic.
8060. The method of claim 7902 wherein the agent is not an anti- fungal agent.
8061. The method of claim 7902, wherein the composition comprises a polymer.
8062. The method of claim 7902, wherein the composition comprises a polymeric carrier.
8063. The method of claim 7902 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
8064. The method of claim 7902 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
8065. The method of claim 7902 wherein the device has a coating that comprises the anti-scarring agent.
8066. The method of claim 7902, wherein the device has a coating that comprises the agent and is disposed on a surface of the sensor.
8067. The method of claim 7902, wherein the device has a coating that comprises the agent and directly contacts the sensor.
8068. The method of claim 7902, wherein the device has a coating that comprises the agent and indirectly contacts the sensor.
8069. The method of claim 7902, wherein the device has a coating that comprises the agent and partially covers the sensor.
8070. The method of claim 7902, wherein the device has a coating that comprises the agent and completely covers the sensor.
8071. The method of claim 7902, wherein the device has a uniform coating.
8072. The method of claim 7902, wherein the device has a non- uniform coating.
8073. The method of claim 7902, wherein the device has a discontinuous coating.
8074. The method of claim 7902, wherein the device has a patterned coating.
8075. The method of claim 7902, wherein the device has a coating with a thickness of 100 μm or less.
8076. The method of claim 7902, wherein the device has a coating with a thickness of 10 μm or less.
8077. The method of claim 7902, wherein the device has a coating, and the coating adheres to the surface of the sensor upon deployment of the sensor.
8078. The method of claim 7902, wherein the device has a coating, and wherein the coating is stable at room temperature for a period of 1 year.
8079. The method of claim 7902, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001 % to about 1 % by weight.
8080. The method of claim 7902, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
8081. The method of claim 7902, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
8082. The method of claim 7902, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
8083. The method of claim 7902, wherein the device has a coating, and wherein the coating further comprises a polymer.
8084. The method of claim 7902, wherein the device has a first coating having a first composition and a second coating having a second composition.
8085. The method of claim 7902, wherein the device has a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
8086. The method of claim 7902, wherein the composition comprises a polymer.
8087. The method of claim 7902, wherein the composition comprises a polymeric carrier.
8088. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a copolymer.
8089. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a block copolymer.
8090. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a random copolymer.
8091. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a biodegradable polymer.
8092. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-biodegradable polymer.
8093. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophilic polymer.
8094. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophobic polymer.
8095. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophilic domains.
8096. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophobic domains.
8097. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-conductive polymer.
8098. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an elastomer.
8099. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrogel.
8100. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a silicone polymer.
8101. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrocarbon polymer.
8102. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a styrene-derived polymer.
8103. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a butadiene polymer.
8104. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a macromer.
8105. The method of claim 7902, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
8106. The method of claim 7902 wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an amorphous polymer.
8107. The method of claim 7902, wherein the device comprises a lubricious coating.
8108. The method of claim 7902 wherein the anti-scarring agent is located within pores or holes of the device.
8109. The method of claim 7902 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
8110. The method of claim 7902, wherein the device comprises a second pharmaceutically active agent.
8111. The method of claim 7902 wherein the device comprises an anti-inflammatory agent.
8112. The method of claim 7902 wherein the device comprises an agent that inhibits infection.
8113. The method of claim 7902 wherein the device comprises an agent that inhibits infection, and wherein the agent is an anthracycline.
8114. The method of claim 7902 wherein the device comprises an agent that inhibits infection, and wherein the agent is doxorubicin.
8115. The method of claim 7902 wherein the device comprises an agent that inhibits infection, and wherein the agent is mitoxantrone.
8116. The method of claim 7902 wherein the device comprises an agent that inhibits infection, and wherein the agent is a fluoropyrimidine.
8117. The method of claim 7902 wherein the device comprises an agent that inhibits infection, and wherein the agent is 5-fluorouracil (5-FU).
8118. The method of claim 7902 wherein the device comprises an agent that inhibits infection, and wherein the agent is a folic acid antagonist.
8119. The method of claim 7902 wherein the device comprises an agent that inhibits infection, and wherein the agent is methotrexate.
8120. The method of claim 7902 wherein the device comprises an agent that inhibits infection, and wherein the agent is a podophylotoxin.
8121. The method of claim 7902 wherein the device comprises an agent that inhibits infection, and wherein the agent is etoposide.
8122. The method of claim 7902 wherein the device comprises an agent that inhibits infection, and wherein the agent is a camptothecin.
8123. The method of claim 7902 wherein the device comprises an agent that inhibits infection, and wherein the agent is a hydroxyurea.
8124. The method of claim 7902 wherein the device comprises an agent that inhibits infection, and wherein the agent is a platinum complex.
8125. The method of claim 7902 wherein the device comprises an agent that inhibits infection, and wherein the agent is cisplatin.
8126. The method of claim 7902, further comprising an anti- thrombotic agent
8127. The method of claim 7902 wherein the device comprises a visualization agent.
8128. The method of claim 7902 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
8129. The method of claim 7902 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises barium, tantalum, or technetium.
8130. The method of claim 7902 wherein the device comprises a visualization agent, and wherein the visualization agent is a MRI responsive material.
8131. The method of claim 7902 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a gadolinium chelate.
8132. The method of claim 7902 wherein the device comprises a visualization agent, and wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
8133. The method of claim 7902 wherein the device comprises a visualization agent, and wherein the visualization agent comprises an iron oxide compound.
8134. The method of claim 7902 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a dye, pigment, or colorant.
8135. The method of claim 7902 wherein the device comprises an echogenic material.
8136. The method of claim 7902 wherein the device comprises an echogenic material, and wherein the echogenic material is in the form of a coating.
8137. The method of claim 7902 wherein the device is sterile.
8138. The method of claim 7902 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
8139. The method of claim 7902 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is connective tissue.
8140. The method of claim 7902 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is muscle tissue.
8141. The method of claim 7902 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is nerve tissue.
8142. The method of claim 7902 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is epithelium tissue.
8143. The method of claim 7902 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
8144. The method of claim 7902 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
8145. The method of claim 7902 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
8146. The method of claim 7902 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
8147. The method of claim 7902 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
8148. The method of claim 7902 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
8149. The method of claim 7902 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
8150. The method of claim 7902 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
8151. The method of claim 7902 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
8152. The method of claim 7902 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
8153. The method of claim 7902 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
8154. The method of claim 7902 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
8155. The method of claim 7902 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
8156. The method of claim 7902 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8157. The method of claim 7902 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8158. The method of claim 7902 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8159. The method of claim 7902 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8160. The method of claim 7902 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8161. The method of claim 7902 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8162. The method of claim 7902 wherein the combining is performed by direct affixing the agent or the composition to the sensor.
8163. The method of claim 7902 wherein the combining is performed by spraying the agent or the component onto the sensor.
8164. The method of claim 7902 wherein the combining is performed by electrospraying the agent or the composition onto the sensor.
8165. The method of claim 7902 wherein the combining is performed by dipping the sensor into a solution comprising the agent or the composition.
8166. The method of claim 7902 wherein the combining is performed by covalently attaching the agent or the composition to the sensor.
8167. The method of claim 7902 wherein the combining is performed by non-covalently attaching the agent or the composition to the sensor.
8168. The method of claim 7902 wherein the combining is performed by coating the sensor with a substance that contains the agent or the composition.
8169. The method of claim 7902 wherein the combining is performed by coating the sensor with a substance that absorbs the agent.
8170. The method of claim 7902 wherein the combining is performed by interweaving a thread composed of, or coated with, the agent or the composition.
8171. The method of claim 7902 wherein the combining is performed by completely covering the sensor with a sleeve that contains the agent or the composition.
8172. The method of claim 7902 wherein the combining is performed by covering a portion of the sensor with a sleeve that contains the agent or the composition.
8173. The method of claim 7902 wherein the combining is performed by completely covering the sensor with a cover that contains the agent or the composition.
8174. The method of claim 7902 wherein the combining is performed by covering a portion of the sensor with a cover that contains the agent or the composition.
8175. The method of claim 7902 wherein the combining is performed by completely covering the sensor with an electrospun fabric that contains the agent or the composition.
8176. The method of claim 7902 wherein the combining is performed by covering a portion of the sensor with an electrospun fabric that contains the agent or the composition.
8177. The method of claim 7902 wherein the combining is performed by completely covering the sensor with a mesh that contains the agent or the composition.
8178. The method of claim 7902 wherein the combining is performed by covering a portion of the sensor with a mesh that contains the agent or the composition.
8179. The method of claim 7902 wherein the combining is performed by constructing a portion of the sensor with the agent or the composition. ',
8180. The method of claim 7902 wherein the combining is performed by impregnating the sensor with the agent or the composition.
8181. The method of claim 7902 wherein the combining is performed by constructing a portion of the sensor from a degradable polymer that releases the agent.
8182. The method of claim 7902 wherein the combining is performed by dipping the sensor into a solution that comprise the agent and an inert solvent for the sensor.
8183. The method of claim 7902 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
8184. The method of claim 7902 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
8185. The method of claim 7902 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
8186. The method of claim 7902 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
8187. The method of claim 7902 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
8188. The method of claim 7902 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and an inert solvent for the sensor.
8189. The method of claim 7902 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
8190. The method of claim 7902 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
8191. The method of claim 7902 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
8192. The method of claim 7902 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
8193. The method of claim 7902 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
8194. The method of any one of claims 7902-8193 wherein the device monitors blood pressure.
8195. The method of any one of claims 7902-8193 wherein the device monitors fluid flow.
8196. The method of any one of claims 7902-8193 wherein the device monitors pressure within an aneurysm sac.
8197. The method of any one of claims 7902-8193 wherein the device monitors intracranial pressure.
8198. The method of any one of claims 7902-8193 wherein the device monitors mechanical pressure associated with a bone fracture.
8199. The method of any one of claims 7902-8193 wherein the device monitors barometric pressure.
8200. The method of any one of claims 7902-8193 wherein the device monitors eye tremors.
8201. The method of any one of claims 7902-8193 wherein the device monitors the depth of a corneal implant.
8202. The method of any one of claims 7902-8193 wherein the device monitors intraocular pressure.
8203. The method of any one of claims 7902-8193 wherein the device is a passive sensor with an inductor-capacitor circuit
8204. The method of any one of claims 7902-8193 wherein the device is a self-powered strain sensing system.
8205. The method of any one of claims 7902-8193 wherein the sensor comprises a lead, a sensor module, a sensor circuit and means for providing voltage.
8206. A method for making a device comprising: combining a cardiac sensor and an anti-scarring agent or a composition comprising an anti- scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
8207. The method of claim 8206 wherein the agent inhibits cell regeneration.
8208. The method of claim 8206 wherein the agent inhibits angiogenesis.
8209. The method of claim 8206 wherein the agent inhibits fibroblast migration.
8210. The method of claim 8206 wherein the agent inhibits fibroblast proliferation.
8211. The method of claim 8206 wherein the agent inhibits deposition of extracellular matrix.
8212. The method of claim 8206 wherein the agent inhibits tissue remodeling.
8213. The method of claim 8206 wherein the agent is an angiogenesis inhibitor.
8214. The method of claim 8206 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
8215. The method of claim 8206 wherein the agent is a chemokine receptor antagonist.
8216. The method of claim 8206 wherein the agent is a cell cycle inhibitor.
8217. The method of claim 8206 wherein the agent is a taxane.
8218. The method of claim 8206 wherein the agent is an antimicrotubule agent.
8219. The method of claim 8206 wherein the agent is paclitaxel.
8220. The method of claim 8206 wherein the agent is not paclitaxel.
8221. The method of claim 8206 wherein the agent is an analogue or derivative of paclitaxel.
8222. The method of claim 8206 wherein the agent is a vinca alkaloid.
8223. The method of claim 8206 wherein the agent is camptothecin or an analogue or derivative thereof.
8224. The method of claim 8206 wherein the agent is a podophyllotoxin.
8225. The method of claim 8206 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
8226. The method of claim 8206 wherein the agent is an anthracycline.
8227. The method of claim 8206 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
8228. The method of claim 8206 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
8229. The method of claim 8206 wherein the agent is a platinum compound.
8230. The method of claim 8206 wherein the agent is a nitrosourea.
8231. The method of claim 8206 wherein the agent is a nitroimidazole.
8232. The method of claim 8206 wherein the agent is a folic acid antagonist.
8233. The method of claim 8206 wherein the agent is a cytidine analogue.
8234. The method of claim 8206 wherein the agent is a pyrimidine analogue.
8235. The method of claim 8206 wherein the agent is a fluoropyrimidine analogue.
8236. The method of claim 8206 wherein the agent is a purine analogue.
8237. The method of claim 8206 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
8238. The method of claim 8206 wherein the agent is a hydroxyurea.
8239. The method of claim 8206 wherein the agent is a mytomicin or an analogue or derivative thereof.
8240. The method of claim 8206 wherein the agent is an alkyl sulfonate.
8241. The method of claim 8206 wherein the agent is a benzamide or an analogue or derivative thereof.
8242. The method of claim 8206 wherein the agent is a nicotinamide or an analogue or derivative thereof.
8243. The method of claim 8206 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
8244. The method of claim 8206 wherein the agent is a DNA alkylating agent.
8245. The method of claim 8206 wherein the agent is an antimicrotubule agent.
8246. The method of claim 8206 wherein the agent is a topoisomerase inhibitor.
8247. The method of claim 8206 wherein the agent is a DNA cleaving agent.
8248. The method of claim 8206 wherein the agent is an antimetabolite.
8249. The method of claim 8206 wherein the agent inhibits adenosine deaminase.
8250. The method of claim 8206 wherein the agent inhibits purine ring synthesis.
8251. The method of claim 8206 wherein the agent is a nucleotide interconversion inhibitor.
8252. The method of claim 8206 wherein the agent inhibits dihydrofolate reduction.
8253. The method of claim 8206 wherein the agent blocks thymidine monophosphate.
8254. The method of claim 8206 wherein the agent causes DNA damage.
8255. The method of claim 8206 wherein the agent is a DNA intercalation agent.
8256. The method of claim 8206 wherein the agent is a RNA synthesis inhibitor.
8257. The method of claim 8206 wherein the agent is a pyrimidine synthesis inhibitor.
8258. The method of claim 8206 wherein the agent inhibits ribonucleotide synthesis or function.
8259. The method of claim 8206 wherein the agent inhibits thymidine monophosphate synthesis or function.
8260. The method of claim 8206 wherein the agent inhibits DNA synthesis.
8261. The method of claim 8206 wherein the agent causes DNA adduct formation.
8262. The method of claim 8206 wherein the agent inhibits protein synthesis.
8263. The method of claim 8206 wherein the agent inhibits microtubule function.
8264. The method of claim 8206 wherein the agent is a cyclin dependent protein kinase inhibitor.
8265. The method of claim 8206 wherein the agent is an epidermal growth factor kinase inhibitor.
8266. The method of claim 8206 wherein the agent is an elastase inhibitor.
8267. The method of claim 8206 wherein the agent is a factor Xa inhibitor.
8268. The method of claim 8206 wherein the agent is a farnesyltransferase inhibitor.
8269. The method of claim 8206 wherein the agent is a fibrinogen antagonist.
8270. The method of claim 8206 wherein the agent is a guanylate cyclase stimulant.
8271. The method of claim 8206 wherein the agent is a heat shock protein 90 antagonist.
8272. The method of claim 8206 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
8273. The method of claim 8206 wherein the agent is a guanylate cyclase stimulant
8274. The method of claim 8206 wherein the agent is a HMGCoA reductase inhibitor.
8275. The method of claim 8206 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
8276. The method of claim 8206 wherein the agent is a hydroorotate dehydrogenase inhibitor.
8277. The method of claim 8206 wherein the agent is an IKK2 inhibitor.
8278. The method of claim 8206 wherein the agent is an IL-1 antagonist.
8279. The method of claim 8206 wherein the agent is an ICE antagonist.
8280. The method of claim 8206 wherein the agent is an IRAK antagonist.
8281. The method of claim 8206 wherein the agent is an IL-4 agonist.
8282. The method of claim 8206 wherein the agent is an immunomodulatory agent.
8283. The method of claim 8206 wherein the agent is sirolimus or an analogue or derivative thereof.
8284. The method of claim 8206 wherein the agent is not sirolimus.
8285. The method of claim 8206 wherein the agent is everolimus or an analogue or derivative thereof.
8286. The method of claim 8206 wherein the agent is tacrolimus or an analogue or derivativethereof.
8287. The method of claim 8206 wherein the agent is not tacrolimus.
8288. The method of claim 8206 wherein the agent is biolmus or an analogue or derivative thereof.
8289. The method of claim 8206 wherein the agent is tresperimus or an analogue or derivative thereof.
8290. The method of claim 8206 wherein the agent is auranofin or an analogue or derivative thereof.
8291. The method of claim 8206 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
8292. The method of claim 8206 wherein the agent is gusperimus or an analogue or derivative thereof.
8293. The method of claim 8206 wherein the agent is pimecrolimus or an analogue or derivative thereof.
8294. The method of claim 8206 wherein the agent is ABT-578 or an analogue or derivative thereof.
8295. The method of claim 8206 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
8296. The method of claim 8206 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
8297. The method of claim 8206 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
8298. The method of claim 8206 wherein the agent is a leukotriene inhibitor.
8299. The method of claim 8206 wherein the agent is a MCP-1 antagonist.
8300. The method of claim 8206 wherein the agent is a MMP inhibitor.
8301. The method of claim 8206 wherein the agent is an NF kappa B inhibitor.
8302. The method of claim 8206 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
8303. The method of claim 8206 wherein the agent is an NO antagonist.
8304. The method of claim 8206 wherein the agent is a p38 MAP kinase inhibitor.
8305. The method of claim 8206 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
8306. The method of claim 8206 wherein the agent is a phosphodiesterase inhibitor.
8307. The method of claim 8206 wherein the agent is a TGF beta inhibitor.
8308. The method of claim 8206 wherein the agent is a thromboxane A2 antagonist.
8309. The method of claim 8206 wherein the agent is a TNF alpha antagonist.
8310. The method of claim 8206 wherein the agent is a TACE inhibitor.
8311. The method of claim 8206 wherein the agent is a tyrosine kinase inhibitor.
8312. The method of claim 8206 wherein the agent is a vitronectin inhibitor.
8313. The method of claim 8206 wherein the agent is a fibroblast growth factor inhibitor.
8314. The method of claim 8206 wherein the agent is a protein kinase inhibitor.
8315. The method of claim 8206 wherein the agent is a PDGF receptor kinase inhibitor.
8316. The method of claim 8206 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
8317. The method of claim 8206 wherein the agent is a retinoic acid receptor antagonist.
8318. The method of claim 8206 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
8319. The method of claim 8206 wherein the agent is a fibrinogen antagonist.
8320. The method of claim 8206 wherein the agent is an antimycotic agent.
8321. The method of claim 8206 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
8322. The method of claim 8206 wherein the agent is a bisphosphonate.
8323. The method of claim 8206 wherein the agent is a phospholipase A1 inhibitor.
8324. The method of claim 8206 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
8325. The method of claim 8206 wherein the agent is a macrolide antibiotic.
8326. The method of claim 8206 wherein the agent is a GPIIb/llla receptor antagonist.
8327. The method of claim 8206 wherein the agent is an endothelin receptor antagonist
8328. The method of claim 8206 wherein the agent is a peroxisome proliferator-activated receptor agonist.
8329. The method of claim 8206 wherein the agent is an estrogen receptor agent.
8330. The method of claim 8206 wherein the agent is a somastostatin analogue.
8331. The method of claim 8206 wherein the agent is a neurokinin 1 antagonist.
8332. The method of claim 8206 wherein the agent is a neurokinin 3 antagonist.
8333. The method of claim 8206 wherein the agent is a VLA-4 antagonist.
8334. The method of claim 8206 wherein the agent is an osteoclast inhibitor.
8335. The method of claim 8206 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
8336. The method of claim 8206 wherein the agent is an angiotensin I converting enzyme inhibitor.
8337. The method of claim 8206 wherein the agent is an angiotensin II antagonist.
8338. The method of claim 8206 wherein the agent is an enkephalinase inhibitor.
8339. The method of claim 8206 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
8340. The method of claim 8206 wherein the agent is a protein kinase C inhibitor.
8341. The method of claim 8206 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
8342. The method of claim 8206 wherein the agent is a CXCR3 inhibitor.
8343. The method of claim 8206 wherein the agent is an Itk inhibitor.
8344. The method of claim 8206 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
8345. The method of claim 8206 wherein the agent is a PPAR agonist.
8346. The method of claim 8206 wherein the agent is an immunosuppressant
8347. The method of claim 8206 wherein the agent is an Erb inhibitor.
8348. The method of claim 8206 wherein the agent is an apoptosis agonist.
8349. The method of claim 8206 wherein the agent is a lipocortin agonist.
8350. The method of claim 8206 wherein the agent is a VCAM-1 antagonist.
8351. The method of claim 8206 wherein the agent is a collagen antagonist.
8352. The method of claim 8206 wherein the agent is an alpha 2 integrin antagonist
8353. The method of claim 8206 wherein the agent is a TNF alpha inhibitor.
8354. The method of claim 8206 wherein the agent is a nitric oxide inhibitor
8355. The method of claim 8206 wherein the agent is a cathepsin inhibitor.
8356. The method of claim 8206 wherein the agent is not an anti- inflammatory agent.
8357. The method of claim 8206 wherein the agent is not a steroid.
8358. The method of claim 8206 wherein the agent is not a glucocorticosteroid.
8359. The method of claim 8206 wherein the agent is not dexamethasone.
8360. The method of claim 8206 wherein the agent is not beclomethasone.
8361. The method of claim 8206 wherein the agent is not dipropionate.
8362. The method of claim 8206 wherein the agent is not an anti- infective agent.
8363. The method of claim 8206 wherein the agent is not an antibiotic.
8364. The method of claim 8206 wherein the agent is not an anti- fungal agent.
8365. The method of claim 8206, wherein the composition comprises a polymer.
8366. The method of claim 8206, wherein the composition comprises a polymeric carrier.
8367. The method of claim 8206 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
8368. The method of claim 8206 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
8369. The method of claim 8206 wherein the device has a coating that comprises the anti-scarring agent.
8370. The method of claim 8206, wherein the device has a coating that comprises the agent and is disposed on a surface of the sensor.
8371. The method of claim 8206, wherein the device has a coating that comprises the agent and directly contacts the sensor.
8372. The method of claim 8206, wherein the device has a coating that comprises the agent and indirectly contacts the sensor.
8373. The method of claim 8206, wherein the device has a coating that comprises the agent and partially covers the sensor.
,
8374. The method of claim 8206, wherein the device has a coating that comprises the agent and completely covers the sensor.
8375. The method of claim 8206, wherein the device has a uniform coating.
8376. The method of claim 8206, wherein the device has a non- uniform coating.
8377. The method of claim 8206, wherein the device has a discontinuous coating.
8378. The method of claim 8206, wherein the device has a patterned coating.
8379. The method of claim 8206, wherein the device has a coating with a thickness of 100 μm or less.
8380. The method of claim 8206, wherein the device has a coating with a thickness of 10 μm or less.
8381. The method of claim 8206, wherein the device has a coating, and the coating adheres to the surface of the sensor upon deployment of the sensor.
8382. The method of claim 8206, wherein the device has a coating, and wherein the coating is stable at room temperature for a period of 1 year.
8383. The method of claim 8206, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001 % to about 1 % by weight.
8384. The method of claim 8206, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
8385. The method of claim 8206, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
8386. The method of claim 8206, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
8387. The method of claim 8206, wherein the device has a coating, and wherein the coating further comprises a polymer.
8388. The method of claim 8206, wherein the device has a first coating having a first composition and a second coating having a second composition.
8389. The method of claim 8206, wherein the device has a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
8390. The method of claim 8206, wherein the composition comprises a polymer. j
8391. The method of claim 8206, wherein the composition comprises a polymeric carrier.
8392. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a copolymer.
8393. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a block copolymer.
8394. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a random copolymer.
8395. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a biodegradable polymer.
8396. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-biodegradable polymer.
8397. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophilic polymer.
8398. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophobic polymer.
8399. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophilic domains.
8400. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophobic domains.
8401. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-conductive polymer.
8402. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an elastomer.
8403. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrogel.
8404. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a silicone polymer.
8405. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrocarbon polymer.
8406. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a styrene-derived polymer.
8407. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a butadiene polymer.
8408. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a macromer.
8409. The method of claim 8206, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a poly(ethyiene glycol) polymer.
8410. The method of claim 8206 wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an amorphous polymer.
8411. The method of claim 8206, wherein the device comprises a lubricious coating.
8412. The method of claim 8206 wherein the anti-scarring agent is located within pores or holes of the device.
8413. The method of claim 8206 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
8414. The method of claim 8206, wherein the device comprises a second pharmaceutically active agent.
8415. The method of claim 8206 wherein the device comprises an anti-inflammatory agent.
8416. The method of claim 8206 wherein the device comprises an agent that inhibits infection.
8417. The method of claim 8206 wherein the device comprises an agent that inhibits infection, and wherein the agent is an anthracycline.
8418. The method of claim 8206 wherein the device comprises an agent that inhibits infection, and wherein the agent is doxorubicin.
8419. The method of claim 8206 wherein the device comprises an agent that inhibits infection, and wherein the agent is mitoxantrone.
8420. The method of claim 8206 wherein the device comprises an agent that inhibits infection, and wherein the agent is a fluoropyrimidine.
8421. The method of claim 8206 wherein the device comprises an agent that inhibits infection, and wherein the agent is 5-fluorouracil (5-FU).
8422. The method of claim 8206 wherein the device comprises an agent that inhibits infection, and wherein the agent is a folic acid antagonist.
8423. The method of claim 8206 wherein the device comprises an agent that inhibits infection, and wherein the agent is methotrexate.
8424. The method of claim 8206 wherein the device comprises an agent that inhibits infection, and wherein the agent is a podophylotoxin.
8425. The method of claim 8206 wherein the device comprises an agent that inhibits infection, and wherein the agent is etoposide.
8426. The method of claim 8206 wherein the device comprises an agent that inhibits infection, and wherein the agent is a camptothecin.
8427. The method of claim 8206 wherein the device comprises an agent that inhibits infection, and wherein the agent is a hydroxyurea.
8428. The method of claim 8206 wherein the device comprises an agent that inhibits infection, and wherein the agent is a platinum complex.
8429. The method of claim 8206 wherein the device comprises an agent that inhibits infection, and wherein the agent is cisplatin.
8430. The method of claim 8206, further comprising an anti- thrombotic agent.
8431. The method of claim 8206 wherein the device comprises a visualization agent.
8432. The method of claim 8206 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
8433. The method of claim 8206 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises barium, tantalum, or technetium.
8434. The method of claim 8206 wherein the device comprises a visualization agent, and wherein the visualization agent is a MRI responsive material.
8435. The method of claim 8206 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a gadolinium chelate.
8436. The method of claim 8206 wherein the device comprises a visualization agent, and wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
8437. The method of claim 8206 wherein the device comprises a visualization agent, and wherein the visualization agent comprises an iron oxide compound.
8438. The method of claim 8206 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a dye, pigment, or colorant.
8439. The method of claim 8206 wherein the device comprises an echogenic material.
8440. The method of claim 8206 wherein the device comprises an echogenic material, and wherein the echogenic material is in the form of a coating.
8441. The method of claim 8206 wherein the device is sterile.
8442. The method of claim 8206 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
8443. The method of claim 8206 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is connective tissue.
8444. The method of claim 8206 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is muscle tissue.
8445. The method of claim 8206 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is nerve tissue.
8446. The method of claim 8206 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is epithelium tissue.
8447. The method of claim 8206 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
8448. The method of claim 8206 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
8449. The method of claim 8206 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
8450. The method of claim 8206 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
8451. The method of claim 8206 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
8452. The method of claim 8206 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
8453. The method of claim 8206 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
8454. The method of claim 8206 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
8455. The method of claim 8206 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
8456. The method of claim 8206 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
8457. The method of claim 8206 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
8458. The method of claim 8206 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
8459. The method of claim 8206 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
8460. The method of claim 8206 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8461. The method of claim 8206 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8462. The method of claim 8206 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8463. The method of claim 8206 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8464. The method of claim 8206 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8465. The method of claim 8206 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8466. The method of claim 8206 wherein the combining is performed by direct affixing the agent or the composition to the sensor.
8467. The method of claim 8206 wherein the combining is performed by spraying the agent or the component onto the sensor.
8468. The method of claim 8206 wherein the combining is performed by electrospraying the agent or the composition onto the sensor.
8469. The method of claim 8206 wherein the combining is performed by dipping the sensor into a solution comprising the agent or the composition.
8470. The method of claim 8206 wherein the combining is performed by covalently attaching the agent or the composition to the sensor.
8471. The method of claim 8206 wherein the combining is performed by non-covalently attaching the agent or the composition to the sensor.
8472. The method of claim 8206 wherein the combining is performed by coating the sensor with a substance that contains the agent or the composition.
8473. The method of claim 8206 wherein the combining is performed by coating the sensor with a substance that absorbs the agent.
8474. The method of claim 8206 wherein the combining is performed by interweaving a thread composed of, or coated with, the agent or the composition.
8475. The method of claim 8206 wherein the combining is performed by completely covering the sensor with a sleeve that contains the agent or the composition.
8476. The method of claim 8206 wherein the combining is performed by covering a portion of the sensor with a sleeve that contains the agent or the composition.
8477. The method of claim 8206 wherein the combining is performed by completely covering the sensor with a cover that contains the agent or the composition.
8478. The method of claim 8206 wherein the combining is performed by covering a portion of the sensor with a cover that contains the agent or the composition.
8479. The method of claim 8206 wherein the combining is performed by completely covering the sensor with an electrospun fabric that contains the agent or the composition.
8480. The method of claim 8206 wherein the combining is performed by covering a portion of the sensor with an electrospun fabric that contains the agent or the composition.
8481. The method of claim 8206 wherein the combining is performed by completely covering the sensor with a mesh that contains the agent or the composition.
8482. The method of claim 8206 wherein the combining is performed by covering a portion of the sensor with a mesh that contains the agent or the composition.
8483. The method of claim 8206 wherein the combining is performed by constructing a portion of the sensor with the agent or the composition.
8484. The method of claim 8206 wherein the combining is performed by impregnating the sensor with the agent or the composition.
8485. The method of claim 8206 wherein the combining is performed by constructing a portion of the sensor from a degradable polymer that releases the agent.
8486. The method of claim 8206 wherein the combining is performed by dipping the sensor into a solution that comprise the agent and an inert solvent for the sensor.
8487. The method of claim 8206 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
8488. The method of claim 8206 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
8489. The method of claim 8206 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
8490. The method of claim 8206 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
8491. The method of claim 8206 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
8492. The method of claim 8206 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and an inert solvent for the sensor.
8493. The method of claim 8206 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
8494. The method of claim 8206 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
8495. The method of claim 8206 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
8496. The method of claim 8206 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
8497. The method of claim 8206 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
8498. The method of any one of claims 8206-8497 wherein the device monitors cardiac output.
8499. The method of any one of claims 8206-8497 wherein the device monitors ejection fraction.
8500. The method of any one of claims 8206-8497 wherein the device monitors blood pressure in a heart chamber.
8501. The method of any one of claims 8206-8497 wherein the device monitors ventricular wall motions.
8502. The method of any one of claims 8206-8497 wherein the device monitors blood flow to a transplanted organ.
8503. The method of any one of claims 8206-8497 wherein the device monitors heart rate.
8504. A method for making a device comprising: combining a respiratory sensor and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
8505. The method of claim 8504 wherein the agent inhibits cell regeneration.
8506. The method of claim 8504 wherein the agent inhibits angiogenesis.
8507. The method of claim 8504 wherein the agent inhibits fibroblast migration.
8508. The method of claim 8504 wherein the agent inhibits fibroblast proliferation.
8509. The method of claim 8504 wherein the agent inhibits deposition of extracellular matrix.
8510. The method of claim 8504 wherein the agent inhibits tissue remodeling.
8511. The method of claim 8504 wherein the agent is an angiogenesis inhibitor.
8512. The method of claim 8504 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
8513. The method of claim 8504 wherein the agent is a chemokine receptor antagonist.
8514. The method of claim 8504 wherein the agent is a cell cycle inhibitor.
8515. The method of claim 8504 wherein the agent is a taxane.
8516. The method of claim 8504 wherein the agent is an antimicrotubule agent.
8517. The method of claim 8504 wherein the agent is paclitaxel.
8518. The method of claim 8504 wherein the agent is not paclitaxel.
8519. The method of claim 8504 wherein the agent is an analogue or derivative of paclitaxel.
8520. The method of claim 8504 wherein the agent is a vinca alkaloid.
8521. The method of claim 8504 wherein the agent is camptothecin or an analogue or derivative thereof.
8522. The method of claim 8504 wherein the agent is a podophyllotoxin.
8523. The method of claim 8504 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
8524. The method of claim 8504 wherein the agent is an anthracycline.
8525. The method of claim 8504 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
8526. The method of claim 8504 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
8527. The method of claim 8504 wherein the agent is a platinum compound.
8528. The method of claim 8504 wherein the agent is a nitrosourea.
8529. The method of claim 8504 wherein the agent is a nitroimidazole.
8530. The method of claim 8504 wherein the agent is a folic acid antagonist.
8531. The method of claim 8504 wherein the agent is a cytidine analogue.
8532. The method of claim 8504 wherein the agent is a pyrimidine analogue.
8533. The method of claim 8504 wherein the agent is a fluoropyrimidine analogue.
8534. The method of claim 8504 wherein the agent is a purine analogue.
8535. The method of claim 8504 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
8536. The method of claim 8504 wherein the agent is a hydroxyurea.
8537. The method of claim 8504 wherein the agent is a mytomicin or an analogue or derivative thereof.
8538. The method of claim 8504 wherein the agent is an alkyl sulfonate.
8539. The method of claim 8504 wherein the agent is a benzamide or an analogue or derivative thereof.
8540. The method of claim 8504 wherein the agent is a nicotinamide or an analogue or derivative thereof.
8541. The method of claim 8504 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
8542. The method of claim 8504 wherein the agent is a DNA alkylating agent.
8543. The method of claim 8504 wherein the agent is an antimicrotubule agent.
8544. The method of claim 8504 wherein the agent is a topoisomerase inhibitor.
8545. The method of claim 8504 wherein the agent is a DNA cleaving agent.
8546. The method of claim 8504 wherein the agent is an antimetabolite.
8547. The method of claim 8504 wherein the agent inhibits adenosine deaminase.
8548. The method of claim 8504 wherein the agent inhibits purine ring synthesis.
8549. The method of claim 8504 wherein the agent is a nucleotide interconversion inhibitor.
8550. The method of claim 8504 wherein the agent inhibits dihydrofolate reduction.
8551. The method of claim 8504 wherein the agent blocks thymidine monophosphate.
8552. The method of claim 8504 wherein the agent causes DNA damage.
8553. The method of claim 8504 wherein the agent is a DNA intercalation agent.
8554. The method of claim 8504 wherein the agent is a RNA synthesis inhibitor.
8555. The method of claim 8504 wherein the agent is a pyrimidine synthesis inhibitor.
8556. The method of claim 8504 wherein the agent inhibits ribonucleotide synthesis or function.
8557. The method of claim 8504 wherein the agent inhibits thymidine monophosphate synthesis or function.
8558. The method of claim 8504 wherein the agent inhibits DNA synthesis.
8559. The method of claim 8504 wherein the agent causes DNA adduct formation.
8560. The method of claim 8504 wherein the agent inhibits protein synthesis.
8561. The method of claim 8504 wherein the agent inhibits microtubule function.
8562. The method of claim 8504 wherein the agent is a cyclin dependent protein kinase inhibitor.
8563. The method of claim 8504 wherein the agent is an epidermal growth factor kinase inhibitor.
8564. The method of claim 8504 wherein the agent is an elastase inhibitor.
8565. The method of claim 8504 wherein the agent is a factor Xa inhibitor.
8566. The method of claim 8504 wherein the agent is a farnesyltransferase inhibitor.
8567. The method of claim 8504 wherein the agent is a fibrinogen antagonist.
8568. The method of claim 8504 wherein the agent is a guanylate cyclase stimulant.
8569. The method of claim 8504 wherein the agent is a heat shock protein 90 antagonist.
8570. The method of claim 8504 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
8571. The method of claim 8504 wherein the agent is a guanylate cyclase stimulant.
8572. The method of claim 8504 wherein the agent is a HMGCoA reductase inhibitor.
8573. The method of claim 8504 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
8574. The method of claim 8504 wherein the agent is a hydroorotate dehydrogenase inhibitor.
8575. The method of claim 8504 wherein the agent is an IKK2 inhibitor.
8576. The method of claim 8504 wherein the agent is an IL-1 antagonist.
8577. The method of claim 8504 wherein the agent is an ICE antagonist
8578. The method of claim 8504 wherein the agent is an IRAK antagonist.
8579. The method of claim 8504 wherein the agent is an IL-4 agonist.
8580. The method of claim 8504 wherein the agent is an immunomodulatory agent.
8581. The method of claim 8504 wherein the agent is sirolimus or an analogue or derivative thereof.
8582. The method of claim 8504 wherein the agent is not sirolimus.
8583. The method of claim 8504 wherein the agent is everolimus or an analogue or derivative thereof.
8584. The method of claim 8504 wherein the agent is tacrolimus or an analogue or derivative thereof.
8585. The method of claim 8504 wherein the agent is not tacrolimus.
8586. The method of claim 8504 wherein the agent is biolmus or an analogue or derivative thereof.
8587. The method of claim 8504 wherein the agent is tresperimus or an analogue or derivative thereof.
8588. The method of claim 8504 wherein the agent is auranofin or an analogue or derivative thereof.
8589. The method of claim 8504 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
8590. The method of claim 8504 wherein the agent is gusperimus or an analogue or derivative thereof.
8591. The method of claim 8504 wherein the agent is pimecrolimus or an analogue or derivative thereof.
8592. The method of claim 8504 wherein the agent is ABT-578 or an analogue or derivative thereof.
8593. The method of claim 8504 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
8594. The method of claim 8504 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
8595. The method of claim 8504 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
8596. The method of claim 8504 wherein the agent is a leukotriene inhibitor.
8597. The method of claim 8504 wherein the agent is a MCP-1 antagonist.
8598. The method of claim 8504 wherein the agent is a MMP inhibitor.
8599. The method of claim 8504 wherein the agent is an NF kappa B inhibitor.
8600. The method of claim 8504 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
8601. The method of claim 8504 wherein the agent is an NO antagonist.
8602. The method of claim 8504 wherein the agent is a p38 MAP kinase inhibitor.
8603. The method of claim 8504 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
8604. The method of claim 8504 wherein the agent is a phosphodiesterase inhibitor.
8605. The method of claim 8504 wherein the agent is a TGF beta inhibitor.
8606. The method of claim 8504 wherein the agent is a thromboxane A2 antagonist.
8607. The method of claim 8504 wherein the agent is a TNF alpha antagonist.
8608. The method of claim 8504 wherein the agent is a TACE inhibitor.
8609. The method of claim 8504 wherein the agent is a tyrosine kinase inhibitor.
8610. The method of claim 8504 wherein the agent is a vitronectin inhibitor.
8611. The method of claim 8504 wherein the agent is a fibroblast growth factor inhibitor.
8612. The method of claim 8504 wherein the agent is a protein kinase inhibitor.
8613. The method of claim 8504 wherein the agent is a PDGF receptor kinase inhibitor.
8614. The method of claim 8504 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
8615. The method of claim 8504 wherein the agent is a retinoic acid receptor antagonist.
8616. The method of claim 8504 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
8617. The method of claim 8504 wherein the agent is a fibrinogen antagonist
8618. The method of claim 8504 wherein the agent is an antimycotic agent.
8619. The method of claim 8504 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
8620. The method of claim 8504 wherein the agent is a bisphosphonate.
8621. The method of claim 8504 wherein the agent is a phospholipase A1 inhibitor.
8622. The method of claim 8504 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
8623. The method of claim 8504 wherein the agent is a macrolide antibiotic.
8624. The method of claim 8504 wherein the agent is a GPIIb/llla receptor antagonist.
8625. The method of claim 8504 wherein the agent is an endothelin receptor antagonist
8626. The method of claim 8504 wherein the agent is a peroxisome proliferator-activated receptor agonist.
8627. The method of claim 8504 wherein the agent is an estrogen receptor agent.
8628. The method of claim 8504 wherein the agent is a somastostatin analogue.
8629. The method of claim 8504 wherein the agent is a neurokinin 1 antagonist.
8630. The method of claim 8504 wherein the agent is a neurokinin 3 antagonist.
8631. The method of claim 8504 wherein the agent is a VLA-4 antagonist.
8632. The method of claim 8504 wherein the agent is an osteoclast inhibitor.
8633. The method of claim 8504 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
8634. The method of claim 8504 wherein the agent is an angiotensin I converting enzyme inhibitor.
8635. The method of claim 8504 wherein the agent is an angiotensin II antagonist.
8636. The method of claim 8504 wherein the agent is an enkephalinase inhibitor.
8637. The method of claim 8504 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
8638. The method of claim 8504 wherein the agent is a protein kinase C inhibitor.
8639. The method of claim 8504 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
8640. The method of claim 8504 wherein the agent is a CXCR3 inhibitor.
8641. The method of claim 8504 wherein the agent is an Itk inhibitor.
8642. The method of claim 8504 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
8643. The method of claim 8504 wherein the agent is a PPAR agonist.
8644. The method of claim 8504 wherein the agent is an immunosuppressant
8645. The method of claim 8504 wherein the agent is an Erb inhibitor.
8646. The method of claim 8504 wherein the agent is an apoptosis agonist.
8647. The method of claim 8504 wherein the agent is a lipocortin agonist.
8648. The method of claim 8504 wherein the agent is a VCAM-1 antagonist.
8649. The method of claim 8504 wherein the agent is a collagen antagonist.
8650. The method of claim 8504 wherein the agent is an alpha 2 integrin antagonist.
8651. The method of claim 8504 wherein the agent is a TNF alpha inhibitor.
8652. The method of claim 8504 wherein the agent is a nitric oxide inhibitor
8653. The method of claim 8504 wherein the agent is a cathepsin inhibitor.
8654. The method of claim 8504 wherein the agent is not an anti- inflammatory agent.
8655. The method of claim 8504 wherein the agent is not a steroid.
8656. The method of claim 8504 wherein the agent is not a glucocorticosteroid.
8657. The method of claim 8504 wherein the agent is not dexamethasone.
8658. The method of claim 8504 wherein the agent is not beclomethasone.
8659. The method of claim 8504 wherein the agent is not dipropionate.
8660. The method of claim 8504 wherein the agent is not an anti- infective agent.
8661. The method of claim 8504 wherein the agent is not an antibiotic.
8662. The method of claim 8504 wherein the agent is not an anti- fungal agent
8663. The method of claim 8504, wherein the composition comprises a polymer.
8664. The method of claim 8504, wherein the composition comprises a polymeric carrier.
8665. The method of claim 8504 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
8666. The method of claim 8504 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
8667. The method of claim 8504 wherein the device has a coating that comprises the anti-scarring agent.
8668. The method of claim 8504, wherein the device has a coating that comprises the agent and is disposed on a surface of the sensor.
8669. The method of claim 8504, wherein the device has a coating that comprises the agent and directly contacts the sensor.
8670. The method of claim 8504, wherein the device has a coating that comprises the agent and indirectly contacts the sensor.
8671. The method of claim 8504, wherein the device has a coating that comprises the agent and partially covers the sensor.
8672. The method of claim 8504, wherein the device has a coating that comprises the agent and completely covers the sensor.
8673. The method of claim 8504, wherein the device has a uniform coating.
8674. The method of claim 8504, wherein the device has a non- uniform coating.
8675. The method of claim 8504, wherein the device has a discontinuous coating.
8676. The method of claim 8504, wherein the device has a patterned coating.
8677. The method of claim 8504, wherein the device has a coating with a thickness of 100 μm or less.
8678. The method of claim 8504, wherein the device has a coating with a thickness of 10 μm or less.
8679. The method of claim 8504, wherein the device has a coating, and the coating adheres to the surface of the sensor upon deployment of the sensor.
8680. The method of claim 8504, wherein the device has a coating, and wherein the coating is stable at room temperature for a period of 1 year.
8681. The method of claim 8504, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001 % to about 1 % by weight
8682. The method of claim 8504, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
8683. The method of claim 8504, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
8684. The method of claim 8504, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
8685. The method of claim 8504, wherein the device has a coating, and wherein the coating further comprises a polymer.
8686. The method of claim 8504, wherein the device has a first coating having a first composition and a second coating having a second composition.
8687. The method of claim 8504, wherein the device has a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
8688. The method of claim 8504, wherein the composition comprises a polymer.
8689. The method of claim 8504, wherein the composition comprises a polymeric carrier.
8690. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a copolymer.
8691. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a block copolymer.
8692. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a random copolymer.
8693. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a biodegradable polymer.
8694. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-biodegradable polymer.
8695. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophilic polymer.
8696. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophobic polymer.
8697. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophilic domains.
8698. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophobic domains.
8699. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-conductive polymer.
8700. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an elastomer.
8701. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrogel.
8702. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a silicone polymer.
8703. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrocarbon polymer.
8704. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a styrene-derived polymer.
8705. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a butadiene polymer.
8706. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a macromer.
8707. The method of claim 8504, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
8708. The method of claim 8504 wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an amorphous polymer.
8709. The method of claim 8504, wherein the device comprises a lubricious coating.
8710. The method of claim 8504 wherein the anti-scarring agent is located within pores or holes of the device.
8711. The method of claim 8504 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
8712. The method of claim 8504, wherein the device comprises a second pharmaceutically active agent
8713. The method of claim 8504 wherein the device comprises an anti-inflammatory agent
8714. The method of claim 8504 wherein the device comprises an agent that inhibits infection.
8715. The method of claim 8504 wherein the device comprises an agent that inhibits infection, and wherein the agent is an anthracycline.
8716. The method of claim 8504 wherein the device comprises an agent that inhibits infection, and wherein the agent is doxorubicin.
8717. The method of claim 8504 wherein the device comprises an agent that inhibits infection, and wherein the agent is mitoxantrone.
8718. The method of claim 8504 wherein the device comprises an agent that inhibits infection, and wherein the agent is a fluoropyrimidine.
8719. The method of claim 8504 wherein the device comprises an agent that inhibits infection, and wherein the agent is 5-fluorouracil (5-FU).
8720. The method of claim 8504 wherein the device comprises an agent that inhibits infection, and wherein the agent is a folic acid antagonist.
8721. The method of claim 8504 wherein the device comprises an agent that inhibits infection, and wherein the agent is methotrexate.
8722. The method of claim 8504 wherein the device comprises an agent that inhibits infection, and wherein the agent is a podophylotoxin.
8723. The method of claim 8504 wherein the device comprises an agent that inhibits infection, and wherein the agent is etoposide.
8724. The method of claim 8504 wherein the device comprises an agent that inhibits infection, and wherein the agent is a camptothecin.
8725. The method of claim 8504 wherein the device comprises an agent that inhibits infection, and wherein the agent is a hydroxyurea.
8726. The method of claim 8504 wherein the device comprises an agent that inhibits infection, and wherein the agent is a platinum complex.
8727. The method of claim 8504 wherein the device comprises an agent that inhibits infection, and wherein the agent is cisplatin.
8728. The method of claim 8504, further comprising an anti- thrombotic agent.
8729. The method of claim 8504 wherein the device comprises a visualization agent.
8730. The method of claim 8504 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
8731. The method of claim 8504 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises barium, tantalum, or technetium.
8732. The method of claim 8504 wherein the device comprises a visualization agent, and wherein the visualization agent is a MRI responsive material.
8733. The method of claim 8504 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a gadolinium chelate.
8734. The method of claim 8504 wherein the device comprises a visualization agent, and wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
8735. The method of claim 8504 wherein the device comprises a visualization agent, and wherein the visualization agent comprises an iron oxide compound.
8736. The method of claim 8504 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a dye, pigment, or colorant.
8737. The method of claim 8504 wherein the device comprises an echogenic material.
8738. The method of claim 8504 wherein the device comprises . an echogenic material, and wherein the echogenic material is in the form of a coating.
8739. The method of claim 8504 wherein the device is sterile. *
8740. The method of claim 8504 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
8741. The method of claim 8504 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is connective tissue.
8742. The method of claim 8504 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is muscle tissue.
8743. The method of claim 8504 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is nerve tissue.
8744. The method of claim 8504 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is epithelium tissue.
8745. The method of claim 8504 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
8746. The method of claim 8504 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
8747. The method of claim 8504 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
8748. The method of claim 8504 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
8749. The method of claim 8504 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
8750. The method of claim 8504 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
8751. The method of claim 8504 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
8752. The method of claim 8504 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
8753. The method of claim 8504 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
8754. The method of claim 8504 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
8755. The method of claim 8504 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
8756. The method of claim 8504 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
8757. The method of claim 8504 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
8758. The method of claim 8504 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8759. The method of claim 8504 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8760. The method of claim 8504 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8761. The method of claim 8504 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8762. The method of claim 8504 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8763. The method of claim 8504 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
8764. The method of claim 8504 wherein the combining is performed by direct affixing the agent or the composition to the sensor.
8765. The method of claim 8504 wherein the combining is performed by spraying the agent or the component onto the sensor.
8766. The method of claim 8504 wherein the combining is performed by electrospraying the agent or the composition onto the sensor.
8767. The method of claim 8504 wherein the combining is performed by dipping the sensor into a solution comprising the agent or the composition.
8768. The method of claim 8504 wherein the combining is performed by covalently attaching the agent or the composition to the sensor.
8769. The method of claim 8504 wherein the combining is performed by non-covalently attaching the agent or the composition to the sensor.
8770. The method of claim 8504 wherein the combining is performed by coating the sensor with a substance that contains the agent or the composition.
8771. The method of claim 8504 wherein the combining is performed by coating the sensor with a substance that absorbs the agent.
8772. The method of claim 8504 wherein the combining is performed by interweaving a thread composed of, or coated with, the agent or the composition.
8773. The method of claim 8504 wherein the combining is performed by completely covering the sensor with a sleeve that contains the agent or the composition.
8774. The method of claim 8504 wherein the combining is performed by covering a portion of the sensor with a sleeve that contains the agent or the composition.
8775. The method of claim 8504 wherein the combining is performed by completely covering the sensor with a cover that contains the agent or the composition.
8776. The method of claim 8504 wherein the combining is performed by covering a portion of the sensor with a cover that contains the agent or the composition.
8777. The method of claim 8504 wherein the combining is performed by completely covering the sensor with an electrospun fabric that contains the agent or the composition.
8778. The method of claim 8504 wherein the combining is performed by covering a portion of the sensor with an electrospun fabric that contains the agent or the composition.
8779. The method of claim 8504 wherein the combining is performed by completely covering the sensor with a mesh that contains the agent or the composition.
8780. The method of claim 8504 wherein the combining is performed by covering a portion of the sensor with a mesh that contains the agent or the composition.
8781. The method of claim 8504 wherein the combining is performed by constructing a portion of the sensor with the agent or the composition.
8782. The method of claim 8504 wherein the combining is performed by impregnating the sensor with the agent or the composition.
8783. The method of claim 8504 wherein the combining is performed by constructing a portion of the sensor from a degradable polymer that releases the agent.
8784. The method of claim 8504 wherein the combining is performed by dipping the sensor into a solution that comprise the agent and an inert solvent for the sensor.
8785. The method of claim 8504 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
8786. The method of claim 8504 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
8787. The method of claim 8504 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
8788. The method of claim 8504 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
8789. The method of claim 8504 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
8790. The method of claim 8504 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and an inert solvent for the sensor.
8791. The method of claim 8504 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
8792. The method of claim 8504 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
8793. The method of claim 8504 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
8794. The method of claim 8504 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
8795. The method of claim 8504 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
8796. The method of claims 8504-8795 wherein the device monitors pulmonary functions.
8797. A method for making a device comprising: combining an auditory sensor and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
8798. The method of claim 8797 wherein the agent inhibits cell regeneration.
8799. The method of claim 8797 wherein the agent inhibits angiogenesis.
8800. The method of claim 8797 wherein the agent inhibits fibroblast migration.
8801. The method of claim 8797 wherein the agent inhibits fibroblast proliferation.
8802. The method of claim 8797 wherein the agent inhibits deposition of extracellular matrix.
8803. The method of claim 8797 wherein the agent inhibits tissue remodeling.
8804. The method of claim 8797 wherein the agent is an angiogenesis inhibitor.
8805. The method of claim 8797 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
8806. The method of claim 8797 wherein the agent is a chemokine receptor antagonist.
8807. The method of claim 8797 wherein the agent is a cell cycle inhibitor.
8808. The method of claim 8797 wherein the agent is a taxane.
8809. The method of claim 8797 wherein the agent is an antimicrotubule agent.
8810. The method of claim 8797 wherein the agent is paclitaxel.
8811. The method of claim 8797 wherein the agent is not paclitaxel.
8812. The method of claim 8797 wherein the agent is an analogue or derivative of paclitaxel.
8813. The method of claim 8797 wherein the agent is a vinca alkaloid.
8814. The method of claim 8797 wherein the agent is camptothecin or an analogue or derivative thereof.
8815. The method of claim 8797 wherein the agent is a podophyllotoxin.
8816. The method of claim 8797 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
8817. The method of claim 8797 wherein the agent is an anthracycline.
8818. The method of claim 8797 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
8819. The method of claim 8797 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
8820. The method of claim 8797 wherein the agent is a platinum compound.
8821. The method of claim 8797 wherein the agent is a nitrosourea.
8822. The method of claim 8797 wherein the agent is a nitroimidazole.
8823. The method of claim 8797 wherein the agent is a folic acid antagonist.
8824. The method of claim 8797 wherein the agent is a cytidine analogue.
8825. The method of claim 8797 wherein the agent is a pyrimidine analogue.
8826. The method of claim 8797 wherein the agent is a fluoropyrimidine analogue.
8827. The method of claim 8797 wherein the agent is a purine analogue.
8828. The method of claim 8797 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
8829. The method of claim 8797 wherein the agent is a hydroxyurea.
8830. The method of claim 8797 wherein the agent is a mytomicin or an analogue or derivative thereof.
8831. The method of claim 8797 wherein the agent is an alkyl sulfonate.
8832. The method of claim 8797 wherein the agent is a benzamide or an analogue or derivative thereof.
8833. The method of claim 8797 wherein the agent is a nicotinamide or an analogue or derivative thereof.
8834. The method of claim 8797 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
8835. The method of claim 8797 wherein the agent is a DNA alkylating agent.
8836. The method of claim 8797 wherein the agent is an antimicrotubule agent.
8837. The method of claim 8797 wherein the agent is a topoisomerase inhibitor.
8838. The method of claim 8797 wherein the agent is a DNA cleaving agent.
8839. The method of claim 8797 wherein the agent is an antimetabolite.
8840. The method of claim 8797 wherein the agent inhibits adenosine deaminase.
8841. The method of claim 8797 wherein the agent inhibits purine ring synthesis.
8842. The method of claim 8797 wherein the agent is a nucleotide interconversion inhibitor.
8843. The method of claim 8797 wherein the agent inhibits dihydrofolate reduction.
8844. The method of claim 8797 wherein the agent blocks thymidine monophosphate.
8845. The method of claim 8797 wherein the agent causes DNA damage.
8846. The method of claim 8797 wherein the agent is a DNA intercalation agent.
8847. The method of claim 8797 wherein the agent is a RNA synthesis inhibitor.
8848. The method of claim 8797 wherein the agent is a pyrimidine synthesis inhibitor.
8849. The method of claim 8797 wherein the agent inhibits ribonucleotide synthesis or function.
8850. The method of claim 8797 wherein the agent inhibits thymidine monophosphate synthesis or function.
8851. The method of claim 8797 wherein the agent inhibits DNA synthesis.
8852. The method of claim 8797 wherein the agent causes DNA adduct formation.
8853. The method of claim 8797 wherein the agent inhibits protein synthesis.
8854. The method of claim 8797 wherein the agent inhibits microtubule function.
8855. The method of claim 8797 wherein the agent is a cyclin dependent protein kinase inhibitor.
8856. The method of claim 8797 wherein the agent is an epidermal growth factor kinase inhibitor.
8857. The method of claim 8797 wherein the agent is an elastase inhibitor.
8858. The method of claim 8797 wherein the agent is a factor Xa inhibitor.
8859. The method of claim 8797 wherein the agent is a farnesyltransferase inhibitor.
8860. The method of claim 8797 wherein the agent is a fibrinogen antagonist.
8861. The method of claim 8797 wherein the agent is a guanylate cyclase stimulant.
8862. The method of claim 8797 wherein the agent is a heat shock protein 90 antagonist.
8863. The method of claim 8797 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
8864. The method of claim 8797 wherein the agent is a guanylate cyclase stimulant.
8865. The method of claim 8797 wherein the agent is a HMGCoA reductase inhibitor.
8866. The method of claim 8797 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
8867. The method of claim 8797 wherein the agent is a hydroorotate dehydrogenase inhibitor.
8868. The method of claim 8797 wherein the agent is an IKK2 inhibitor.
8869. The method of claim 8797 wherein the agent is an IL-1 antagonist.
8870. The method of claim 8797 wherein the agent is an ICE antagonist
8871. The method of claim 8797 wherein the agent is an IRAK antagonist.
8872. The method of claim 8797 wherein the agent is an IL-4 agonist.
8873. The method of claim 8797 wherein the agent is an immunomodulatory agent.
8874. The method of claim 8797 wherein the agent is sirolimus or an analogue or derivative thereof.
8875. The method of claim 8797 wherein the agent is not sirolimus.
8876. The method of claim 8797 wherein the agent is everolimus or an analogue or derivative thereof.
8877. The method of claim 8797 wherein the agent is tacrolimus or an analogue or derivative thereof.
8878. The method of claim 8797 wherein the agent is not tacrolimus.
8879. The method of claim 8797 wherein the agent is biolmus or an analogue or derivative thereof.
8880. The method of claim 8797 wherein the agent is tresperimus or an analogue or derivative thereof.
8881. The method of claim 8797 wherein the agent is auranofin or an analogue or derivative thereof.
8882. The method of claim 8797 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
8883. The method of claim 8797 wherein the agent is gusperimus or an analogue or derivative thereof.
8884. The method of claim 8797 wherein the agent is pimecrolimus or an analogue or derivative thereof.
8885. The method of claim 8797 wherein the agent is ABT-578 or an analogue or derivative thereof.
8886. The method of claim 8797 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
8887. The method of claim 8797 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
8888. The method of claim 8797 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D or an analogue or derivative thereof.
8889. The method of claim 8797 wherein the agent is a leukotriene inhibitor.
8890. The method of claim 8797 wherein the agent is a MCP-1 antagonist.
8891. The method of claim 8797 wherein the agent is a MMP inhibitor.
8892. The method of claim 8797 wherein the agent is an NF kappa B inhibitor.
8893. The method of claim 8797 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
8894. The method of claim 8797 wherein the agent is an NO antagonist.
8895. The method of claim 8797 wherein the agent is a p38 MAP kinase inhibitor.
8896. The method of claim 8797 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
8897. The method of claim 8797 wherein the agent is a phosphodiesterase inhibitor.
8898. The method of claim 8797 wherein the agent is a TGF beta inhibitor.
8899. The method of claim 8797 wherein the agent is a thromboxane A2 antagonist
8900. The method of claim 8797 wherein the agent is a TNF alpha antagonist.
8901. The method of claim 8797 wherein the agent is a TACE inhibitor.
8902. The method of claim 8797 wherein the agent is a tyrosine kinase inhibitor.
8903. The method of claim 8797 wherein the agent is a vitronectin inhibitor.
8904. The method of claim 8797 wherein the agent is a fibroblast growth factor inhibitor.
8905. The method of claim 8797 wherein the agent is a protein kinase inhibitor.
8906. The method of claim 8797 wherein the agent is a PDGF receptor kinase inhibitor.
8907. The method of claim 8797 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
8908. The method of claim 8797 wherein the agent is a retinoic acid receptor antagonist.
8909. The method of claim 8797 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
8910. The method of claim 8797 wherein the agent is a fibrinogen antagonist.
8911. The method of claim 8797 wherein the agent is an antimycotic agent.
8912. The method of claim 8797 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
8913. The method of claim 8797 wherein the agent is a bisphosphonate.
8914. The method of claim 8797 wherein the agent is a phospholipase A1 inhibitor.
8915. The method of claim 8797 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
8916. The method of claim 8797 wherein the agent is a macrolide antibiotic.
8917. The method of claim 8797 wherein the agent is a GPIIb/llla receptor antagonist.
8918. The method of claim 8797 wherein the agent is an endothelin receptor antagonist.
8919. The method of claim 8797 wherein the agent is a peroxisome proliferator-activated receptor agonist.
8920. The method of claim 8797 wherein the agent is an estrogen receptor agent.
8921. The method of claim 8797 wherein the agent is a somastostatin analogue.
8922. The method of claim 8797 wherein the agent is a neurokinin 1 antagonist.
8923. The method of claim 8797 wherein the agent is a neurokinin 3 antagonist.
8924. The method of claim 8797 wherein the agent is a VLA-4 antagonist.
8925. The method of claim 8797 wherein the agent is an osteoclast inhibitor.
8926. The method of claim 8797 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
8927. The method of claim 8797 wherein the agent is an angiotensin I converting enzyme inhibitor.
8928. The method of claim 8797 wherein the agent is an angiotensin II antagonist.
8929. The method of claim 8797 wherein the agent is an enkephalinase inhibitor.
8930. The method of claim 8797 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
8931. The method of claim 8797 wherein the agent is a protein kinase C inhibitor.
8932. The method of claim 8797 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
8933. The method of claim 8797 wherein the agent is a CXCR3 inhibitor.
8934. The method of claim 8797 wherein the agent is an Itk inhibitor.
8935. The method of claim 8797 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
8936. The method of claim 8797 wherein the agent is a PPAR agonist
8937. The method of claim 8797 wherein the agent is an immunosuppressant.
8938. The method of claim 8797 wherein the agent is an Erb inhibitor.
8939. The method of claim 8797 wherein the agent is an apoptosis agonist.
8940. The method of claim 8797 wherein the agent is a lipocortin agonist.
8941. The method of claim 8797 wherein the agent is a VCAM-1 antagonist.
8942. The method of claim 8797 wherein the agent is a collagen antagonist.
8943. The method of claim 8797 wherein the agent is an alpha 2 integrin antagonist.
8944. The method of claim 8797 wherein the agent is a TNF alpha inhibitor.
8945. The method of claim 8797 wherein the agent is a nitric oxide inhibitor
8946. The method of claim 8797 wherein the agent is a cathepsin inhibitor.
8947. The method of claim 8797 wherein the agent is not an anti- inflammatory agent.
8948. The method of claim 8797 wherein the agent is not a steroid.
8949. The method of claim 8797 wherein the agent is not a glucocorticosteroid.
8950. The method of claim 8797 wherein the agent is not dexamethasone.
8951. The method of claim 8797 wherein the agent is not beclomethasone.
8952. The method of claim 8797 wherein the agent is not dipropionate.
8953. The method of claim 8797 wherein the agent is not an anti- infective agent.
8954. The method of claim 8797 wherein the agent is not an antibiotic.
8955. The method of claim 8797 wherein the agent is not an anti- fungal agent.
8956. The method of claim 8797, wherein the composition comprises a polymer.
8957. The method of claim 8797, wherein the composition comprises a polymeric carrier.
8958. The method of claim 8797 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
8959. The method of claim 8797 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
8960. The method of claim 8797 wherein the device has a coating that comprises the anti-scarring agent.
8961. The method of claim 8797, wherein the device has a coating that comprises the agent and is disposed on a surface of the sensor.
8962. The method of claim 8797, wherein the device has a coating that comprises the agent and directly contacts the sensor.
8963. The method of claim 8797, wherein the device has a coating that comprises the agent and indirectly contacts the sensor.
8964. The method of claim 8797, wherein the device has a coating that comprises the agent and partially covers the sensor.
8965. The method of claim 8797, wherein the device has a coating that comprises the agent and completely covers the sensor.
8966. The method of claim 8797, wherein the device has a uniform coating.
8967. The method of claim 8797, wherein the device has a non- uniform coating.
8968. The method of claim 8797, wherein the device has a discontinuous coating.
8969. The method of claim 8797, wherein the device has a patterned coating.
8970. The method of claim 8797, wherein the device has a coating with a thickness of 100 μm or less.
8971. The method of claim 8797, wherein the device has a coating with a thickness of 10 μm or less.
8972. The method of claim 8797, wherein the device has a coating, and the coating adheres to the surface of the sensor upon deployment of the sensor.
8973. The method of claim 8797, wherein the device has a coating, and wherein the coating is stable at room temperature for a period of 1 year.
8974. The method of claim 8797, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1 % by weight.
8975. The method of claim 8797, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
8976. The method of claim 8797, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
8977. The method of claim 8797, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
8978. The method of claim 8797, wherein the device has a coating, and wherein the coating further comprises a polymer.
8979. The method of claim 8797, wherein the device has a first coating having a first composition and a second coating having a second composition.
8980. The method of claim 8797, wherein the device has a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
8981. The method of claim 8797, wherein the composition comprises a polymer.
8982. The method of claim 8797, wherein the composition comprises a polymeric carrier.
8983. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a copolymer.
8984. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a block copolymer.
8985. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a random copolymer.
8986. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a biodegradable polymer.
8987. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-biodegradable polymer.
8988. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophilic polymer.
8989. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophobic polymer.
8990. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophilic domains.
8991. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophobic domains.
8992. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-conductive polymer.
8993. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an elastomer.
8994. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrogel.
8995. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a silicone polymer.
8996. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrocarbon polymer.
8997. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a styrene-derived polymer.
8998. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a butadiene polymer.
8999. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a macromer.
9000. The method of claim 8797, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
9001. The method of claim 8797 wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an amorphous polymer.
9002. The method of claim 8797, wherein the device comprises a lubricious coating.
9003. The method of claim 8797 wherein the anti-scarring agent is located within pores or holes of the device.
9004. The method of claim 8797 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
9005. The method of claim 8797, wherein the device comprises a second pharmaceutically active agent.
9006. The method of claim 8797 wherein the device comprises an anti-inflammatory agent.
9007. The method of claim 8797 wherein the device comprises an agent that inhibits infection.
9008. The method of claim 8797 wherein the device comprises an agent that inhibits infection, and wherein the agent is an anthracycline.
9009. The method of claim 8797 wherein the device comprises an agent that inhibits infection, and wherein the agent is doxorubicin.
9010. The method of claim 8797 wherein the device comprises an agent that inhibits infection, and wherein the agent is mitoxantrone.
9011. The method of claim 8797 wherein the device comprises an agent that inhibits infection, and wherein the agent is a fluoropyrimidine.
9012. The method of claim 8797 wherein the device comprises an agent that inhibits infection, and wherein the agent is 5-fluorouracil (5-FU).
9013. The method of claim 8797 wherein the device comprises an agent that inhibits infection, and wherein the agent is a folic acid antagonist.
9014. The method of claim 8797 wherein the device comprises an agent that inhibits infection, and wherein the agent is methotrexate.
9015. The method of claim 8797 wherein the device comprises an agent that inhibits infection, and wherein the agent is a podophylotoxin.
9016. The method of claim 8797 wherein the device comprises an agent that inhibits infection, and wherein the agent is etoposide.
9017. The method of claim 8797 wherein the device comprises an agent that inhibits infection, and wherein the agent is a camptothecin.
9018. The method of claim 8797 wherein the device comprises an agent that inhibits infection, and wherein the agent is a hydroxyurea.
9019. The method of claim 8797 wherein the device comprises an agent that inhibits infection, and wherein the agent is a platinum complex.
9020. The method of claim 8797 wherein the device comprises an agent that inhibits infection, and wherein the agent is cisplatin.
9021. The method of claim 8797, further comprising an anti- thrombotic agent.
9022. The method of claim 8797 wherein the device comprises a visualization agent.
9023. The method of claim 8797 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
9024. The method of claim 8797 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises barium, tantalum, or technetium.
9025. The method of claim 8797 wherein the device comprises a visualization agent, and wherein the visualization agent is a MRI responsive material.
9026. The method of claim 8797 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a gadolinium chelate.
9027. The method of claim 8797 wherein the device comprises a visualization agent, and wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
9028. The method of claim 8797 wherein the device comprises a visualization agent, and wherein the visualization agent comprises an iron oxide compound.
9029. The method of claim 8797 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a dye, pigment, or colorant.
9030. The method of claim 8797 wherein the device comprises an echogenic material.
9031. The method of claim 8797 wherein the device comprises an echogenic material, and wherein the echogenic material is in the form of a coating.
9032. The method of claim 8797 wherein the device is sterile.
9033. The method of claim 8797 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
9034. The method of claim 8797 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is connective tissue.
9035. The method of claim 8797 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is muscle tissue.
9036. The method of claim 8797 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is nerve tissue.
9037. The method of claim 8797 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is epithelium tissue.
9038. The method of claim 8797 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
9039. The method of claim 8797 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
9040. The method of claim 8797 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
9041. The method of claim 8797 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
9042. The method of claim 8797 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
9043. The method of claim 8797 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
9044. The method of claim 8797 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
9045. The method of claim 8797 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
9046. The method of claim 8797 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
9047. The method of claim 8797 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
9048. The method of claim 8797 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
9049. The method of claim 8797 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
9050. The method of claim 8797 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
9051. The method of claim 8797 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9052. The method of claim 8797 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9053. The method of claim 8797 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9054. The method of claim 8797 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9055. The method of claim 8797 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9056. The method of claim 8797 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9057. The method of claim 8797 wherein the combining is performed by direct affixing the agent or the composition to the sensor.
9058. The method of claim 8797 wherein the combining is performed by spraying the agent or the component onto the sensor.
9059. The method of claim 8797 wherein the combining is performed by electrospraying the agent or the composition onto the sensor.
9060. The method of claim 8797 wherein the combining is performed by dipping the sensor into a solution comprising the agent or the composition.
9061. The method of claim 8797 wherein the combining is performed by covalently attaching the agent or the composition to the sensor.
9062. The method of claim 8797 wherein the combining is performed by non-covalently attaching the agent or the composition to the sensor.
9063. The method of claim 8797 wherein the combining is performed by coating the sensor with a substance that contains the agent or the composition.
9064. The method of claim 8797 wherein the combining is performed by coating the sensor with a substance that absorbs the agent.
9065. The method of claim 8797 wherein the combining is performed by interweaving a thread composed of, or coated with, the agent or the composition.
9066. The method of claim 8797 wherein the combining is performed by completely covering the sensor with a sleeve that contains the agent or the composition.
9067. The method of claim 8797 wherein the combining is performed by covering a portion of the sensor with a sleeve that contains the agent or the composition.
9068. The method of claim 8797 wherein the combining is performed by completely covering the sensor with a cover that contains the agent or the composition.
9069. The method of claim 8797 wherein the combining is performed by covering a portion of the sensor with a cover that contains the agent or the composition.
9070. The method of claim 8797 wherein the combining is performed by completely covering the sensor with an electrospun fabric that contains the agent or the composition.
9071. The method of claim 8797 wherein the combining is performed by covering a portion of the sensor with an electrospun fabric that contains the agent or the composition.
9072. The method of claim 8797 wherein the combining is performed by completely covering the sensor with a mesh that contains the agent or the composition.
9073. The method of claim 8797 wherein the combining is performed by covering a portion of the sensor with a mesh that contains the agent or the composition.
9074. The method of claim 8797 wherein the combining is performed by constructing a portion of the sensor with the agent or the composition.
9075. The method of claim 8797 wherein the combining is performed by impregnating the sensor with the agent or the composition.
9076. The method of claim 8797 wherein the combining is performed by constructing a portion of the sensor from a degradable polymer that releases the agent.
9077. The method of claim 8797 wherein the combining is performed by dipping the sensor into a solution that comprise the agent and an inert solvent for the sensor.
9078. The method of claim 8797 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
9079. The method of claim 8797 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
9080. The method of claim 8797 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
9081. The method of claim 8797 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
9082. The method of claim 8797 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
9083. The method of claim 8797 wherein the combining is performed by spraying the sensor into4a solution that comprises the agent and an inert solvent for the sensor.
9084. The method of claim 8797 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
9085. The method of claim 8797 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
9086. The method of claim 8797 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
9087. The method of claim 8797 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
9088. The method of claim 8797 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
9089. The method of any one of claims 8797-9088 wherein the device is adapted for delivering an electrical signal to an implantable electromechanical transducer that acts on the middle or inner ear.
9090. The method of any one of claims 8797-9088 wherein the device generates an electrical audio signal.
9091. The method of any one of claims 8797-9088 wherein the device is a capacitive sensor that is coupled to a vibrating auditory element.
9092. The method of any one of claims 8797-9088 wherein the device is an electromagnetic sensor.
9093. A method for making a device comprising: combining an electrolyte or metabolite sensor and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
9094. The method of claim 9093 wherein the agent inhibits cell regeneration.
9095. The method of claim 9093 wherein the agent inhibits angiogenesis.
9096. The method of claim 9093 wherein the agent inhibits fibroblast migration.
9097. The method of claim 9093 wherein the agent inhibits fibroblast proliferation.
9098. The method of claim 9093 wherein the agent inhibits deposition of extracellular matrix.
9099. The method of claim 9093 wherein the agent inhibits tissue remodeling.
9100. The method of claim 9093 wherein the agent is an angiogenesis inhibitor.
9101. The method of claim 9093 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
9102. The method of claim 9093 wherein the agent is a chemokine receptor antagonist.
9103. The method of claim 9093 wherein the agent is a cell cycle inhibitor.
9104. The method of claim 9093 wherein the agent is a taxane.
9105. The method of claim 9093 wherein the agent is an antimicrotubule agent.
9106. The method of claim 9093 wherein the agent is paclitaxel.
9107. The method of claim 9093 wherein the agent is not paclitaxel.
9108. The method of claim 9093 wherein the agent is an analogue or derivative of paclitaxel.
9109. The method of claim 9093 wherein the agent is a vinca alkaloid.
9110. The method of claim 9093 wherein the agent is camptothecin or an analogue or derivative thereof.
9111. The method of claim 9093 wherein the agent is a podophyllotoxin.
9112. The method of claim 9093 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
9113. The method of claim 9093 wherein the agent is an anthracycline.
9114. The method of claim 9093 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
9115. The method of claim 9093 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
9116. The method of claim 9093 wherein the agent is a platinum compound.
9117. The method of claim 9093 wherein the agent is a nitrosourea.
9118. The method of claim 9093 wherein the agent is a nitroimidazole.
9119. The method of claim 9093 wherein the agent is a folic acid antagonist.
9120. The method of claim 9093 wherein the agent is a cytidine analogue.
9121. The method of claim 9093 wherein the agent is a pyrimidine analogue.
9122. The method of claim 9093 wherein the agent is a fluoropyrimidine analogue.
9123. The method of claim 9093 wherein the agent is a purine analogue.
9124. The method of claim 9093 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
9125. The method of claim 9093 wherein the agent is a hydroxyurea.
9126. The method of claim 9093 wherein the agent is a mytomicin or an analogue or derivative thereof.
9127. The method of claim 9093 wherein the agent is an alkyl sulfonate.
9128. The method of claim 9093 wherein the agent is a benzamide or an analogue or derivative thereof.
9129. The method of claim 9093 wherein the agent is a nicotinamide or an analogue or derivative thereof.
9130. The method of claim 9093 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
9131. The method of claim 9093 wherein the agent is a DNA alkylating agent.
9132. The method of claim 9093 wherein the agent is an antimicrotubule agent.
9133. The method of claim 9093 wherein the agent is a topoisomerase inhibitor.
9134. The method of claim 9093 wherein the agent is a DNA cleaving agent.
9135. The method of claim 9093 wherein the agent is an antimetabolite.
9136. The method of claim 9093 wherein the agent inhibits adenosine deaminase.
9137. The method of claim 9093 wherein the agent inhibits purine ring synthesis.
9138. The method of claim 9093 wherein the agent is a nucleotide interconversion inhibitor.
9139. The method of claim 9093 wherein the agent inhibits dihydrofolate reduction.
9140. The method of claim 9093 wherein the agent blocks thymidine monophosphate.
9141. The method of claim 9093 wherein the agent causes DNA damage.
9142. The method of claim 9093 wherein the agent is a DNA intercalation agent.
9143. The method of claim 9093 wherein the agent is a RNA synthesis inhibitor.
9144. The method of claim 9093 wherein the agent is a pyrimidine synthesis inhibitor.
9145. The method of claim 9093 wherein the agent inhibits ribonucleotide synthesis or function.
9146. The method of claim 9093 wherein the agent inhibits thymidine monophosphate synthesis or function.
9147. The method of claim 9093 wherein the agent inhibits DNA synthesis.
9148. The method of claim 9093 wherein the agent causes DNA adduct formation.
9149. The method of claim 9093 wherein the agent inhibits protein synthesis.
9150. The method of claim 9093 wherein the agent inhibits microtubule function.
9151. The method of claim 9093 wherein the agent is a cyclin dependent protein kinase inhibitor.
9152. The method of claim 9093 wherein the agent is an epidermal growth factor kinase inhibitor.
9153. The method of claim 9093 wherein the agent is an elastase inhibitor.
9154. The method of claim 9093 wherein the agent is a factor Xa inhibitor.
9155. The method of claim 9093 wherein the agent is a farnesyltransferase inhibitor.
9156. The method of claim 9093 wherein the agent is a fibrinogen antagonist.
9157. The method of claim 9093 wherein the agent is a guanylate cyclase stimulant.
9158. The method of claim 9093 wherein the agent is a heat shock protein 90 antagonist.
9159. The method of claim 9093 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
9160. The method of claim 9093 wherein the agent is a guanylate cyclase stimulant.
9161. The method of claim 9093 wherein the agent is a HMGCoA reductase inhibitor.
9162. The method of claim 9093 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
9163. The method of claim 9093 wherein the agent is a hydroorotate dehydrogenase inhibitor.
9164. The method of claim 9093 wherein the agent is an IKK2 inhibitor.
9165. The method of claim 9093 wherein the agent is an IL-1 antagonist.
9166. The method of claim 9093 wherein the agent is an ICE antagonist.
9167. The method of claim 9093 wherein the agent is an IRAK antagonist.
9168. The method of claim 9093 wherein the agent is an IL-4 agonist.
9169. The method of claim 9093 wherein the agent is an immunomodulatory agent.
9170. The method of claim 9093 wherein the agent is sirolimus or an analogue or derivative thereof.
9171. The method of claim 9093 wherein the agent is not sirolimus.
9172. The method of claim 9093 wherein the agent is everolimus or an analogue or derivative thereof.
9173. The method of claim 9093 wherein the agent is tacrolimus or an analogue or derivative thereof.
9174. The method of claim 9093 wherein the agent is not tacrolimus.
9175. The method of claim 9093 wherein the agent is biolmus or an analogue or derivative thereof.
9176. The method of claim 9093 wherein the agent is tresperimus or an analogue or derivative thereof.
9177. The method of claim 9093 wherein the agent is auranofin or an analogue or derivative thereof.
9178. The method of claim 9093 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
9179. The method of claim 9093 wherein the agent is gusperimus or an analogue or derivative thereof.
9180. The method of claim 9093 wherein the agent is pimecrolimus or an analogue or derivative thereof.
9181. The method of claim 9093 wherein the agent is ABT-578 or an analogue or derivative thereof.
9182. The method of claim 9093 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
9183. The method of claim 9093 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
9184. The method of claim 9093 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
9185. The method of claim 9093 wherein the agent is a leukotriene inhibitor.
9186. The method of claim 9093 wherein the agent is a MCP-1 antagonist.
9187. The method of claim 9093 wherein the agent is a MMP inhibitor.
9188. The method of claim 9093 wherein the agent is an NF kappa B inhibitor.
9189. The method of claim 9093 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
9190. The method of claim 9093 wherein the agent is an NO antagonist.
9191. The method of claim 9093 wherein the agent is a p38 MAP kinase inhibitor.
9192. The method of claim 9093 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
9193. The method of claim 9093 wherein the agent is a phosphodiesterase inhibitor.
9194. The method of claim 9093 wherein the agent is a TGF beta inhibitor.
9195. The method of claim 9093 wherein the agent is a thromboxane A2 antagonist.
9196. The method of claim 9093 wherein the agent is a TNF alpha antagonist.
9197. The method of claim 9093 wherein the agent is a TACE inhibitor.
9198. The method of claim 9093 wherein the agent is a tyrosine kinase inhibitor.
9199. The method of claim 9093 wherein the agent is a vitronectin inhibitor.
9200. The method of claim 9093 wherein the agent is a fibroblast growth factor inhibitor.
9201. The method of claim 9093 wherein the agent is a protein kinase inhibitor.
9202. The method of claim 9093 wherein the agent is a PDGF receptor kinase inhibitor.
9203. The method of claim 9093 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
9204. The method of claim 9093 wherein the agent is a retinoic acid receptor antagonist.
9205. The method of claim 9093 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
9206. The method of claim 9093 wherein the agent is a fibrinogen antagonist.
9207. The method of claim 9093 wherein the agent is an antimycotic agent.
9208. The method of claim 9093 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
9209. The method of claim 9093 wherein the agent is a bisphosphonate.
9210. The method of claim 9093 wherein the agent is a phospholipase A1 inhibitor.
9211. The method of claim 9093 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
9212. The method of claim 9093 wherein the agent is a macrolide antibiotic.
9213. The method of claim 9093 wherein the agent is a GPIIb/llla receptor antagonist.
9214. The method of claim 9093 wherein the agent is an endothelin receptor antagonist.
9215. The method of claim 9093 wherein the agent is a peroxisome proliferator-activated receptor agonist.
9216. The method of claim 9093 wherein the agent is an estrogen receptor agent.
9217. The method of claim 9093 wherein the agent is a somastostatin analogue.
9218. The method of claim 9093 wherein the agent is a neurokinin 1 antagonist.
9219. The method of claim 9093 wherein the agent is a neurokinin 3 antagonist.
9220. The method of claim 9093 wherein the agent is a VLA-4 antagonist.
9221. The method of claim 9093 wherein the agent is an osteoclast inhibitor.
9222. The method of claim 9093 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
9223. The method of claim 9093 wherein the agent is an angiotensin I converting enzyme inhibitor.
9224. The method of claim 9093 wherein the agent is an angiotensin II antagonist.
9225. The method of claim 9093 wherein the agent is an enkephalinase inhibitor.
9226. The method of claim 9093 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
9227. The method of claim 9093 wherein the agent is a protein kinase C inhibitor.
9228. The method of claim 9093 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
9229. The method of claim 9093 wherein the agent is a CXCR3 inhibitor.
9230. The method of claim 9093 wherein the agent is an Itk inhibitor.
9231. The method of claim 9093 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
9232. The method of claim 9093 wherein the agent is a PPAR agonist.
9233. The method of claim 9093 wherein the agent is an immunosuppressant
9234. The method of claim 9093 wherein the agent is an Erb inhibitor.
9235. The method of claim 9093 wherein the agent is an apoptosis agonist.
9236. The method of claim 9093 wherein the agent is a lipocortin agonist.
9237. The method of claim 9093 wherein the agent is a VCAM-1 antagonist.
9238. The method of claim 9093 wherein the agent is a collagen antagonist
9239. The method of claim 9093 wherein the agent is an alpha 2 integrin antagonist.
9240. The method of claim 9093 wherein the agent is a TNF alpha inhibitor.
9241. The method of claim 9093 wherein the agent is a nitric oxide inhibitor
9242. The method of claim 9093 wherein the agent is a cathepsin inhibitor.
9243. The method of claim 9093 wherein the agent is not an anti- inflammatory agent.
9244. The method of claim 9093 wherein the agent is not a steroid.
9245. The method of claim 9093 wherein the agent is not a glucocorticosteroid.
9246. The method of claim 9093 wherein the agent is not dexamethasone.
9247. The method of claim 9093 wherein the agent is not beclomethasone.
9248. The method of claim 9093 wherein the agent is not dipropionate.
9249. The method of claim 9093 wherein the agent is not an anti- infective agent.
9250. The method of claim 9093 wherein the agent is not an antibiotic.
9251. The method of claim 9093 wherein the agent is not an anti- fungal agent.
9252. The method of claim 9093, wherein the composition comprises a polymer.
9253. The method of claim 9093, wherein the composition comprises a polymeric carrier.
9254. The method of claim 9093 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
9255. The method of claim 9093 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
9256. The method of claim 9093 wherein the device has a coating that comprises the anti-scarring agent.
9257. The method of claim 9093, wherein the device has a coating that comprises the agent and is disposed on a surface of the sensor.
9258. The method of claim 9093, wherein the device has a coating that comprises the agent and directly contacts the sensor.
9259. The method of claim 9093, wherein the device has a coating that comprises the agent and indirectly contacts the sensor.
9260. The method of claim 9093, wherein the device has a coating that comprises the agent and partially covers the sensor.
9261. The method of claim 9093, wherein the device has a coating that comprises the agent and completely covers the sensor.
9262. The method of claim 9093, wherein the device has a uniform coating.
9263. The method of claim 9093, wherein the device has a non- uniform coating.
9264. The method of claim 9093, wherein the device has a discontinuous coating.
9265. The method of claim 9093, wherein the device has a patterned coating.
9266. The method of claim 9093, wherein the device has a coating with a thickness of 100 μm or less.
9267. The method of claim 9093, wherein the device has a coating with a thickness of 10 μm or less.
9268. The method of claim 9093, wherein the device has a coating, and the coating adheres to the surface of the sensor upon deployment of the sensor.
9269. The method of claim 9093, wherein the device has a coating, and wherein the coating is stable at room temperature for a period of 1 year.
9270. The method of claim 9093, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001 % to about 1 % by weight.
9271. The method of claim 9093, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
9272. The method of claim 9093, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
9273. The method of claim 9093, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
9274. The method of claim 9093, wherein the device has a coating, and wherein the coating further comprises a polymer.
9275. The method of claim 9093, wherein the device has a first coating having a first composition and a second coating having a second composition.
9276. The method of claim 9093, wherein the device has a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different. ,
9277. The method of claim 9093, wherein the composition comprises a polymer.
9278. The method of claim 9093, wherein the composition comprises a polymeric carrier.
9279. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a copolymer.
9280. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a block copolymer.
9281. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a random copolymer.
9282. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a biodegradable polymer.
9283. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-biodegradable polymer.
9284. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophilic polymer.
9285. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophobic polymer.
9286. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophilic domains.
9287. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophobic domains.
9288. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-conductive polymer.
9289. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an elastomer.
9290. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrogel.
9291. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a silicone polymer.
9292. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrocarbon polymer.
9293. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a styrene-derived polymer.
9294. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a butadiene polymer.
9295. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a macromer.
9296. The method of claim 9093, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a poly( ethylene glycol) polymer.
9297. The method of claim 9093 wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an amorphous polymer.
9298. The method of claim 9093, wherein the device comprises a lubricious coating.
9299. The method of claim 9093 wherein the anti-scarring agent is located within pores or holes of the device.
9300. The method of claim 9093 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
9301. The method of claim 9093, wherein the device comprises a second pharmaceutically active agent.
9302. The method of claim 9093 wherein the device comprises an anti-inflammatory agent.
9303. The method of claim 9093 wherein the device comprises an agent that inhibits infection.
9304. The method of claim 9093 wherein the device comprises an agent that inhibits infection, and wherein the agent is an anthracycline.
9305. The method of claim 9093 wherein the device comprises an agent that inhibits infection, and wherein the agent is doxorubicin.
9306. The method of claim 9093 wherein the device comprises an agent that inhibits infection, and wherein the agent is mitoxantrone.
9307. The method of claim 9093 wherein the device comprises an agent that inhibits infection, and wherein the agent is a fluoropyrimidine.
9308. The method of claim 9093 wherein the device comprises an agent that inhibits infection, and wherein the agent is 5-fluorouracil (5-FU).
9309. The method of claim 9093 wherein the device comprises an agent that inhibits infection, and wherein the agent is a folic acid antagonist.
9310. The method of claim 9093 wherein the device comprises an agent that inhibits infection, and wherein the agent is methotrexate.
9311. The method of claim 9093 wherein the device comprises an agent that inhibits infection, and wherein the agent is a podophylotoxin.
9312. The method of claim 9093 wherein the device comprises an agent that inhibits infection, and wherein the agent is etoposide.
9313. The method of claim 9093 wherein the device comprises an agent that inhibits infection, and wherein the agent is a camptothecin.
9314. The method of claim 9093 wherein the device comprises an agent that inhibits infection, and wherein the agent is a hydroxyurea.
9315. The method of claim 9093 wherein the device comprises an agent that inhibits infection, and wherein the agent is a platinum complex.
9316. The method of claim 9093 wherein the device comprises an agent that inhibits infection, and wherein the agent is cisplatin.
9317. The method of claim 9093, further comprising an anti- thrombotic agent.
9318. The method of claim 9093 wherein the device comprises a visualization agent.
9319. The method of claim 9093 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
9320. The method of claim 9093 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises barium, tantalum, or technetium.
9321. The method of claim 9093 wherein the device comprises a visualization agent, and wherein the visualization agent is a MRI responsive material.
9322. The method of claim 9093 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a gadolinium chelate.
9323. The method of claim 9093 wherein the device comprises a visualization agent, and wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
9324. The method of claim 9093 wherein the device comprises a visualization agent, and wherein the visualization agent comprises an iron oxide compound.
9325. The method of claim 9093 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a dye, pigment, or colorant.
9326. The method of claim 9093 wherein the device comprises an echogenic material.
9327. The method of claim 9093 wherein the device comprises an echogenic material, and wherein the echogenic material is in the form of a coating.
9328. The method of claim 9093 wherein the device is sterile.
9329. The method of claim 9093 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
9330. The method of claim 9093 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is connective tissue.
9331. The method of claim 9093 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is muscle tissue.
9332. The method of claim 9093 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is nerve tissue.
9333. The method of claim 9093 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is epithelium tissue.
9334. The method of claim 9093 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
9335. The method of claim 9093 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
9336. The method of claim 9093 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
9337. The method of claim 9093 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
9338. The method of claim 9093 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
9339. The method of claim 9093 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
9340. The method of claim 9093 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
9341. The method of claim 9093 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
9342. The method of claim 9093 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
9343. The method of claim 9093 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent
9344. The method of claim 9093 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
9345. The method of claim 9093 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent
9346. The method of claim 9093 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
9347. The method of claim 9093 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9348. The method of claim 9093 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9349. The method of claim 9093 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9350. The method of claim 9093 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9351. The method of claim 9093 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9352. The method of claim 9093 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9353. The method of claim 9093 wherein the combining is performed by direct affixing the agent or the composition to the sensor.
9354. The method of claim 9093 wherein the combining is performed by spraying the agent or the component onto the sensor.
9355. The method of claim 9093 wherein the combining is performed by electrospraying the agent or the composition onto the sensor.
9356. The method of claim 9093 wherein the combining is performed by dipping the sensor into a solution comprising the agent or the composition.
9357. The method of claim 9093 wherein the combining is performed by covalently attaching the agent or the composition to the sensor.
9358. The method of claim 9093 wherein the combining is performed by non-covalently attaching the agent or the composition to the sensor.
9359. The method of claim 9093 wherein the combining is performed by coating the sensor with a substance that contains the agent or the composition.
9360. The method of claim 9093 wherein the combining is performed by coating the sensor with a substance that absorbs the agent.
9361. The method of claim 9093 wherein the combining is performed by interweaving a thread composed of, or coated with, the agent or the composition.
9362. The method of claim 9093 wherein the combining is performed by completely covering the sensor with a sleeve that contains the agent or the composition.
9363. The method of claim 9093 wherein the combining is performed by covering a portion of the sensor with a sleeve that contains the agent or the composition.
9364. The method of claim 9093 wherein the combining is performed by completely covering the sensor with a cover that contains the agent or the composition.
9365. The method of claim 9093 wherein the combining is performed by covering a portion of the sensor with a cover that contains the agent or the composition.
9366. The method of claim 9093 wherein the combining is performed by completely covering the sensor with an electrospun fabric that contains the agent or the composition.
9367. The method of claim 9093 wherein the combining is performed by covering a portion of the sensor with an electrospun fabric that contains the agent or the composition.
9368. The method of claim 9093 wherein the combining is performed by completely covering the sensor with a mesh that contains the agent or the composition.
9369. The method of claim 9093 wherein the combining is performed by covering a portion of the sensor with a mesh that contains the agent or the composition.
9370. The method of claim 9093 wherein the combining is performed by constructing a portion of the sensor with the agent or the composition.
9371. The method of claim 9093 wherein the combining is performed by impregnating the sensor with the agent or the composition.
9372. The method of claim 9093 wherein the combining is performed by constructing a portion of the sensor from a degradable polymer that releases the agent.
9373. The method of claim 9093 wherein the combining is performed by dipping the sensor into a solution that comprise the agent and an inert solvent for the sensor.
9374. The method of claim 9093 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
9375. The method of claim 9093 wherein the combining is performed by dipping the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
9376. The method of claim 9093 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
9377. The method of claim 9093 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
9378. The method of claim 9093 wherein the combining is performed by dipping the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
9379. The method of claim 9093 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and an inert solvent for the sensor.
9380. The method of claim 9093 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will swill the sensor.
9381. The method of claim 9093 wherein the combining is performed by spraying the sensor into a solution that comprises the agent and a solvent that will dissolve the sensor.
9382. The method of claim 9093 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and an inert solvent for the sensor.
9383. The method of claim 9093 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will swill the sensor.
9384. The method of claim 9093 wherein the combining is performed by spraying the sensor into a solution that comprises the agent, a polymer and a solvent that will dissolve the sensor.
9385. The method of any one of claims 9093-9384 wherein the device emits a source of radiation directed towards blood to interact with a plurality of detectors that provide an output signal.
9386. The method of any one of claims 9093-9384 wherein the device is a biosensing transponder composed of a dye that has optical properties that change in response to changes in the environment, a photosensor to sense the optical changes, and a transponder for transmitting data to a remote reader.
9387. The method of any one of claims 9093-9384 wherein the device is a monolithic bioelectronic device for detecting at least one analyte within the host.
9388. A method for making a device comprising: combining a pump and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
9389. The method of claim 9388 wherein the agent inhibits cell regeneration.
9390. The method of claim 9388 wherein the agent inhibits angiogenesis.
9391. The method of claim 9388 wherein the agent inhibits fibroblast migration.
9392. The method of claim 9388 wherein the agent inhibits fibroblast proliferation.
9393. The method of claim 9388 wherein the agent inhibits deposition of extracellular matrix.
9394. The method of claim 9388 wherein the agent inhibits tissue remodeling.
9395. The method of claim 9388 wherein the agent is an angiogenesis inhibitor.
9396. The method of claim 9388 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
9397. The method of claim 9388 wherein the agent is a chemokine receptor antagonist.
9398. The method of claim 9388 wherein the agent is a cell cycle inhibitor.
9399. The method of claim 9388 wherein the agent is a taxane.
9400. The method of claim 9388 wherein the agent is an antimicrotubule agent.
9401. The method of claim 9388 wherein the agent is paclitaxel.
9402. The method of claim 9388 wherein the agent is not paclitaxel.
9403. The method of claim 9388 wherein the agent is an analogue or derivative of paclitaxel.
9404. The method of claim 9388 wherein the agent is a vinca alkaloid.
9405. The method of claim 9388 wherein the agent is camptothecin or an analogue or derivative thereof.
9406. The method of claim 9388 wherein the agent is a podophyllotoxin.
9407. The method of claim 9388 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
9408. The method of claim 9388 wherein the agent is an anthracycline.
9409. The method of claim 9388 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
9410. The method of claim 9388 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
9411. The method of claim 9388 wherein the agent is a platinum compound.
9412. The method of claim 9388 wherein the agent is a nitrosourea.
9413. The method of claim 9388 wherein the agent is a nitroimidazole.
9414. The method of claim 9388 wherein the agent is a folic acid antagonist.
9415. The method of claim 9388 wherein the agent is a cytidine analogue.
9416. The method of claim 9388 wherein the agent is a pyrimidine analogue.
9417. The method of claim 9388 wherein the agent is a fluoropyrimidine analogue.
9418. The method of claim 9388 wherein the agent is a purine analogue.
9419. The method of claim 9388 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
9420. The method of claim 9388 wherein the agent is a hydroxyurea.
9421. The method of claim 9388 wherein the agent is a mytomicin or an analogue or derivative thereof.
9422. The method of claim 9388 wherein the agent is an alkyl sulfonate.
9423. The method of claim 9388 wherein the agent is a benzamide or an analogue or derivative thereof.
9424. The method of claim 9388 wherein the agent is a nicotinamide or an analogue or derivative thereof.
9425. The method of claim 9388 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
9426. The method of claim 9388 wherein the agent is a DNA alkylating agent.
9427. The method of claim 9388 wherein the agent is an antimicrotubule agent.
9428. The method of claim 9388 wherein the agent is a topoisomerase inhibitor.
9429. The method of claim 9388 wherein the agent is a DNA cleaving agent.
9430. The method of claim 9388 wherein the agent is an antimetabolite.
9431. The method of claim 9388 wherein the agent inhibits adenosine deaminase.
9432. The method of claim 9388 wherein the agent inhibits purine ring synthesis.
9433. The method of claim 9388 wherein the agent is a nucleotide interconversion inhibitor.
9434. The method of claim 9388 wherein the agent inhibits dihydrofolate reduction.
9435. The method of claim 9388 wherein the agent blocks thymidine monophosphate.
9436. The method of claim 9388 wherein the agent causes DNA damage.
9437. The method of claim 9388 wherein the agent is a DNA intercalation agent
9438. The method of claim 9388 wherein the agent is a RNA synthesis inhibitor.
9439. The method of claim 9388 wherein the agent is a pyrimidine synthesis inhibitor.
9440. The method of claim 9388 wherein the agent inhibits ribonucleotide synthesis or function.
9441. The method of claim 9388 wherein the agent inhibits thymidine monophosphate synthesis or function.
9442. The method of claim 9388 wherein the agent inhibits DNA synthesis.
9443. The method of claim 9388 wherein the agent causes DNA adduct formation.
9444. The method of claim 9388 wherein the agent inhibits protein synthesis.
9445. The method of claim 9388 wherein the agent inhibits microtubule function.
9446. The method of claim 9388 wherein the agent is a cyclin dependent protein kinase inhibitor.
9447. The method of claim 9388 wherein the agent is an epidermal growth factor kinase inhibitor.
9448. The method of claim 9388 wherein the agent is an elastase inhibitor.
9449. The method of claim 9388 wherein the agent is a factor Xa inhibitor.
9450. The method of claim 9388 wherein the agent is a farnesyltransferase inhibitor.
9451. The method of claim 9388 wherein the agent is a fibrinogen antagonist
9452. The method of claim 9388 wherein the agent is a guanylate cyclase stimulant.
9453. The method of claim 9388 wherein the agent is a heat shock protein 90 antagonist.
9454. The method of claim 9388 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
9455. The method of claim 9388 wherein the agent is a guanylate cyclase stimulant.
9456. The method of claim 9388 wherein the agent is a HMGCoA reductase inhibitor.
9457. The method of claim 9388 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
9458. The method of claim 9388 wherein the agent is a hydroorotate dehydrogenase inhibitor.
9459. The method of claim 9388 wherein the agent is an IKK2 inhibitor.
9460. The method of claim 9388 wherein the agent is an IL-1 antagonist.
9461. The method of claim 9388 wherein the agent is an ICE antagonist.
9462. The method of claim 9388 wherein the agent is an IRAK antagonist.
9463. The method of claim 9388 wherein the agent is an IL-4 agonist.
9464. The method of claim 9388 wherein the agent is an immunomodulatory agent.
9465. The method of claim 9388 wherein the agent is sirolimus or an analogue or derivative thereof.
9466. The method of claim 9388 wherein the agent is not sirolimus.
9467. The method of claim 9388 wherein the agent is everolimus or an analogue or derivative thereof.
9468. The method of claim 9388 wherein the agent is tacrolimus or an analogue or derivative thereof.
9469. The method of claim 9388 wherein the agent is not tacrolimus.
9470. The method of claim 9388 wherein the agent is biolmus or an analogue or derivative thereof.
9471. The method of claim 9388 wherein the agent is tresperimus or an analogue or derivative thereof.
9472. The method of claim 9388 wherein the agent is auranofin or an analogue or derivative thereof.
9473. The method of claim 9388 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
9474. The method of claim 9388 wherein the agent is gusperimus or an analogue or derivative thereof.
9475. The method of claim 9388 wherein the agent is pimecrolimus or an analogue or derivative thereof.
9476. The method of claim 9388 wherein the agent is ABT-578 or an analogue or derivative thereof.
9477. The method of claim 9388 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
9478. The method of claim 9388 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
9479. The method of claim 9388 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
9480. The method of claim 9388 wherein the agent is a leukotriene inhibitor.
9481. The method of claim 9388 wherein the agent is a MCP-1 antagonist
9482. The method of claim 9388 wherein the agent is a MMP inhibitor.
9483. The method of claim 9388 wherein the agent is an NF kappa B inhibitor.
9484. The method of claim 9388 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
9485. The method of claim 9388 wherein the agent is an NO antagonist.
9486. The method of claim 9388 wherein the agent is a p38 MAP kinase inhibitor.
9487. The method of claim 9388 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
9488. The method of claim 9388 wherein the agent is a phosphodiesterase inhibitor.
9489. The method of claim 9388 wherein the agent is a TGF beta inhibitor.
9490. The method of claim 9388 wherein the agent is a thromboxane A2 antagonist.
9491. The method of claim 9388 wherein the agent is a TNF alpha antagonist.
9492. The method of claim 9388 wherein the agent is a TACE inhibitor.
9493. The method of claim 9388 wherein the agent is a tyrosine kinase inhibitor.
9494. The method of claim 9388 wherein the agent is a vitronectin inhibitor.
9495. The method of claim 9388 wherein the agent is a fibroblast growth factor inhibitor.
9496. The method of claim 9388 wherein the agent is a protein kinase inhibitor.
9497. The method of claim 9388 wherein the agent is a PDGF receptor kinase inhibitor.
9498. The method of claim 9388 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
9499. The method of claim 9388 wherein the agent is a retinoic acid receptor antagonist.
9500. The method of claim 9388 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
9501. The method of claim 9388 wherein the agent is a fibrinogen antagonist.
9502. The method of claim 9388 wherein the agent is an antimycotic agent.
9503. The method of claim 9388 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
9504. The method of claim 9388 wherein the agent is a bisphosphonate.
9505. The method of claim 9388 wherein the agent is a phospholipase A1 inhibitor.
9506. The method of claim 9388 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
9507. The method of claim 9388 wherein the agent is a macrolide antibiotic.
9508. The method of claim 9388 wherein the agent is a GPIIb/llla receptor antagonist.
9509. The method of claim 9388 wherein the agent is an endothelin receptor antagonist.
9510. The method of claim 9388 wherein the agent is a peroxisome proliferator-activated receptor agonist.
9511. The method of claim 9388 wherein the agent is an estrogen receptor agent.
9512. The method of claim 9388 wherein the agent is a somastostatin analogue.
9513. The method of claim 9388 wherein the agent is a neurokinin 1 antagonist.
9514. The method of claim 9388 wherein the agent is a neurokinin 3 antagonist.
9515. The method of claim 9388 wherein the agent is a VLA-4 antagonist.
9516. The method of claim 9388 wherein the agent is an osteoclast inhibitor.
9517. The method of claim 9388 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
9518. The method of claim 9388 wherein the agent is an angiotensin I converting enzyme inhibitor.
9519. The method of claim 9388 wherein the agent is an angiotensin II antagonist.
9520. The method of claim 9388 wherein the agent is an enkephalinase inhibitor.
9521. The method of claim 9388 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
9522. The method of claim 9388 wherein the agent is a protein kinase C inhibitor.
9523. The method of claim 9388 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
9524. The method of claim 9388 wherein the agent is a CXCR3 inhibitor.
9525. The method of claim 9388 wherein the agent is an Itk inhibitor.
9526. The method of claim 9388 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
9527. The method of claim 9388 wherein the agent is a PPAR agonist.
9528. The method of claim 9388 wherein the agent is an immunosuppressant.
9529. The method of claim 9388 wherein the agent is an Erb inhibitor.
9530. The method of claim 9388 wherein the agent is an apoptosis agonist.
9531. The method of claim 9388 wherein the agent is a lipocortin agonist.
9532. The method of claim 9388 wherein the agent is a VCAM-1 antagonist.
9533. The method of claim 9388 wherein the agent is a collagen antagonist.
9534. The method of claim 9388 wherein the agent is an alpha 2 integrin antagonist.
9535. The method of claim 9388 wherein the agent is a TNF alpha inhibitor.
9536. The method of claim 9388 wherein the agent is a nitric oxide inhibitor
9537. The method of claim 9388 wherein the agent is a cathepsin inhibitor.
9538. The method of claim 9388 wherein the agent is not an anti- inflammatory agent.
9539. The method of claim 9388 wherein the agent is not a steroid.
9540. The method of claim 9388 wherein the agent is not a glucocorticosteroid.
9541. The method of claim 9388 wherein the agent is not dexamethasone.
9542. The method of claim 9388 wherein the agent is not beclomethasone.
9543. The method of claim 9388 wherein the agent is not dipropionate.
9544. The method of claim 9388 wherein the agent is not an anti- infective agent.
9545. The method of claim 9388 wherein the agent is not an antibiotic.
9546. The method of claim 9388 wherein the agent is not an anti- fungal agent.
9547. The method of claim 9388, wherein the composition comprises a polymer.
9548. The method of claim 9388, wherein the composition comprises a polymeric carrier.
9549. The method of claim 9388 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
9550. The method of claim 9388 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
9551. The method of claim 9388 wherein the device has a coating that comprises the anti-scarring agent.
9552. The method of claim 9388, wherein the device has a coating that comprises the agent and is disposed on a surface of the pump.
9553. The method of claim 9388, wherein the device has a coating that comprises the agent and directly contacts the pump.
9554. The method of claim 9388, wherein the device has a coating that comprises the agent and indirectly contacts the pump.
9555. The method of claim 9388, wherein the device has a coating that comprises the agent and partially covers the pump.
9556. The method of claim 9388, wherein the device has a coating that comprises the agent and completely covers the pump.
9557. The method of claim 9388, wherein the device has a uniform coating.
9558. The method of claim 9388, wherein the device has a non- uniform coating.
9559. The method of claim 9388, wherein the device has a discontinuous coating.
9560. The method of claim 9388, wherein the device has a patterned coating.
9561. The method of claim 9388, wherein the device has a coating with a thickness of 100 μm or less.
9562. The method of claim 9388, wherein the device has a coating with a thickness of 10 μm or less.
9563. The method of claim 9388, wherein the device has a coating, and the coating adheres to the surface of the pump upon deployment of the pump.
9564. The method of claim 9388, wherein the device has a coating, and wherein the coating is stable at room temperature for a period of 1 year.
9565. The method of claim 9388, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1 % by weight.
9566. The method of claim 9388, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
9567. The method of claim 9388, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight
9568. The method of claim 9388, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
9569. The method of claim 9388, wherein the device has a coating, and wherein the coating further comprises a polymer.
9570. The method of claim 9388, wherein the device has a first coating having a first composition and a second coating having a second composition.
9571. The method of claim 9388, wherein the device has a first coating having a first compositiori and a second coating having a second composition, wherein the first composition and the second composition are different.
9572. The method of claim 9388, wherein the composition comprises a polymer.
9573. The method of claim 9388, wherein the composition comprises a polymeric carrier.
9574. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a copolymer.
9575. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a block copolymer.
9576. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a random copolymer.
9577. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a biodegradable polymer.
9578. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-biodegradable polymer.
9579. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophilic polymer.
9580. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophobic polymer.
9581. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophilic domains.
9582. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophobic domains.
9583. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-conductive polymer.
9584. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an elastomer.
9585. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrogel.
9586. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a silicone polymer.
9587. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrocarbon polymer.
9588. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a styrene-derived polymer.
9589. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a butadiene polymer.
9590. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a macromer.
9591. The method of claim 9388, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
9592. The method of claim 9388 wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an amorphous polymer.
9593. The method of claim 9388, wherein the device comprises a lubricious coating.
9594. The method of claim 9388 wherein the anti-scarring agent is located within pores or holes of the device.
9595. The method of claim 9388 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
9596. The method of claim 9388, wherein the device comprises a second pharmaceutically active agent
9597. The method of claim 9388 wherein the device comprises an anti-inflammatory agent.
9598. The method of claim 9388 wherein the device comprises an agent that inhibits infection.
9599. The method of claim 9388 wherein the device comprises an agent that inhibits infection, and wherein the agent is an anthracycline.
9600. The method of claim 9388 wherein the device comprises an agent that inhibits infection, and wherein the agent is doxorubicin.
9601. The method of claim 9388 wherein the device comprises an agent that inhibits infection, and wherein the agent is mitoxantrone.
9602. The method of claim 9388 wherein the device comprises an agent that inhibits infection, and wherein the agent is a fluoropyrimidine.
9603. The method of claim 9388 wherein the device comprises an agent that inhibits infection, and wherein the agent is 5-fluorouracil (5-FU).
9604. The method of claim 9388 wherein the device comprises an agent that inhibits infection, and wherein the agent is a folic acid antagonist.
9605. The method of claim 9388 wherein the device comprises an agent that inhibits infection, and wherein the agent is methotrexate.
9606. The method of claim 9388 wherein the device comprises an agent that inhibits infection, and wherein the agent is a podophylotoxin.
9607. The method of claim 9388 wherein the device comprises an agent that inhibits infection, and wherein the agent is etoposide.
9608. The method of claim 9388 wherein the device comprises an agent that inhibits infection, and wherein the agent is a camptothecin.
9609. The method of claim 9388 wherein the device comprises an agent that inhibits infection, and wherein the agent is a hydroxyurea.
9610. The method of claim 9388 wherein the device comprises an agent that inhibits infection, and wherein the agent is a platinum complex.
9611. The method of claim 9388 wherein the device comprises an agent that inhibits infection, and wherein the agent is cisplatin.
9612. The method of claim 9388, further comprising an anti- thrombotic agent.
9613. The method of claim 9388 wherein the device comprises a visualization agent.
9614. The method of claim 9388 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
9615. The method of claim 9388 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises barium, tantalum, or technetium.
9616. The method of claim 9388 wherein the device comprises a visualization agent, and wherein the visualization agent is a MRI responsive material.
9617. The method of claim 9388 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a gadolinium chelate.
9618. The method of claim 9388 wherein the device comprises a visualization agent, and wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
9619. The method of claim 9388 wherein the device comprises a visualization agent, and wherein the visualization agent comprises an iron oxide compound.
9620. The method of claim 9388 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a dye, pigment, or colorant.
9621. The method of claim 9388 wherein the device comprises an echogenic material.
9622. The method of claim 9388 wherein the device comprises an echogenic material, and wherein the echogenic material is in the form of a coating.
9623. The method of claim 9388 wherein the device is sterile.
9624. The method of claim 9388 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
9625. The method of claim 9388 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is connective tissue.
9626. The method of claim 9388 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is muscle tissue.
9627. The method of claim 9388 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is nerve tissue.
9628. The method of claim 9388 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is epithelium tissue.
9629. The method of claim 9388 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
9630. The method of claim 9388 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
9631. The method of claim 9388 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
9632. The method of claim 9388 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
9633. The method of claim 9388 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
9634. The method of claim 9388 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
9635. The method of claim 9388 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
9636. The method of claim 9388 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
9637. The method of claim 9388 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
9638. The method of claim 9388 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
9639. The method of claim 9388 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
9640. The method of claim 9388 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
9641. The method of claim 9388 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
9642. The method of claim 9388 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9643. The method of claim 9388 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9644. The method of claim 9388 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9645. The method of claim 9388 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9646. The method of claim 9388 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9647. The method of claim 9388 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9648. The method of claim 9388 wherein the combining is performed by direct affixing the agent or the composition to the pump.
9649. The method of claim 9388 wherein the combining is performed by spraying the agent or the component onto the pump.
9650. The method of claim 9388 wherein the combining is performed by electrospraying the agent or the composition onto the pump.
9651. The method of claim 9388 wherein the combining is performed by dipping the pump into a solution comprising the agent or the composition.
9652. The method of claim 9388 wherein the combining is performed by covalently attaching the agent or the composition to the pump.
9653. The method of claim 9388 wherein the combining is performed by non-covalently attaching the agent or the composition to the pump.
9654. The method of claim 9388 wherein the combining is performed by coating the pump with a substance that contains the agent or the composition.
9655. The method of claim 9388 wherein the combining is performed by coating the pump with a substance that absorbs the agent.
9656. The method of claim 9388 wherein the combining is performed by interweaving the pump with a thread composed of, or coated with, the agent or the composition.
9657. The method of claim 9388 wherein the combining is performed by completely covering the pump with a sleeve that contains the agent or the composition.
9658. The method of claim 9388 wherein the combining is performed by covering a portion of the pump with a sleeve that contains the agent or the composition.
9659. The method of claim 9388 wherein the combining is performed by completely covering the pump with a cover that contains the agent or the composition.
9660. The method of claim 9388 wherein the combining is performed by covering a portion of the pump with a cover that contains the agent or the composition.
9661. The method of claim 9388 wherein the combining is performed by completely covering the pump with an electrospun fabric that contains the agent or the composition.
9662. The method of claim 9388 wherein the combining is performed by covering a portion of the pump with an electrospun fabric that contains the agent or the composition.
9663. The method of claim 9388 wherein the combining is performed by completely covering the pump with a mesh that contains the agent or the composition.
9664. The method of claim 9388 wherein the combining is performed by covering a portion of the pump with a mesh that contains the agent or the composition.
9665. The method of claim 9388 wherein the combining is performed by constructing a portion of the pump with the agent or the composition.
9666. The method of claim 9388 wherein the combining is performed by impregnating the pump with the agent or the composition.
9667. The method of claim 9388 wherein the combining is performed by constructing a portion of the pump from a degradable polymer that releases the agent.
9668. The method of claim 9388 wherein the combining is performed by dipping the pump into a solution that comprise the agent and an inert solvent for the pump.
9669. The method of claim 9388 wherein the combining is performed by dipping the pump into a solution that comprises the agent and a solvent that will swill the pump.
9670. The method of claim 9388 wherein the combining is performed by dipping the pump into a solution that comprises the agent and a solvent that will dissolve the pump.
9671. The method of claim 9388 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and an inert solvent for the pump.
9672. The method of claim 9388 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and a solvent that will swill the pump.
9673. The method of claim 9388 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and a solvent that will dissolve the pump.
9674. The method of claim 9388 wherein the combining is performed by spraying the pump into a solution that comprises the agent and an inert solvent for the pump.
9675. The method of claim 9388 wherein the combining is performed by spraying the pump into a solution that comprises the agent and a solvent that will swill the pump.
9676. The method of claim 9388 wherein the combining is performed by spraying the pump into a solution that comprises the agent and a solvent that will dissolve the pump.
9677. The method of claim 9388 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and an inert solvent for the pump.
9678. The method of claim 9388 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and a solvent that will swill the pump.
9679. The method of claim 9388 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and a solvent that will dissolve the pump.
9680. The method of any one of claims 9388-9679 wherein the device is adapted for delivering insulin.
9681. The method of any one of claims 9388-9679 wherein the device is adapted for delivering a narcotic.
9682. The method of any one of claims 9388-9679 wherein the device is adapted for delivering a chemotherapeutic agent.
9683. The method of any one of claims 9388-9679 wherein the device is adapted for delivering an anti-arrhythmic drug.
9684. The method of any one of claims 9388-9679 wherein the device is adapted for delivering an anti-spasmotic drug.
9685. The method of any one of claims 9388-9679 wherein the device is adapted for delivering an anti-spastic agent.
9686. The method of any one of claims 9388-9679 wherein the device is adapted for delivering an antibiotic.
9687. The method of any one of claims 9388-9679 wherein the device is adapted for delivering a drug only when changes in the host are detected.
9688. The method of any one of claims 9388-9679 wherein the device is adapted for delivering a drug as a continuous slow release.
9689. The method of any one of claims 9388-9679 wherein the device is adapted for delivering a drug at prescribed dosages in a pulsatile manner.
9690. The method of any one of claims 9388-9679 wherein the device is a programmable drug delivery pump.
9691. The method of any one of claims 9388-9679 wherein the device is adapted for intraocularly delivering a drug.
9692. The method of any one of claims 9388-9679 wherein the device is adapted for intrathecally delivering a drug.
9693. The method of any one of claims 9388-9679 wherein the device is adapted for intraperitoneally delivering a drug.
9694. The method of any one of claims 9388-9679 wherein the device is adapted for intra-arterially delivering a drug.
9695. The method of any one of claims 9388-9679 wherein the device is adapted for intracardiac delivery of a drug.
9696. The method of any one of claims 9388-9679 wherein the device is an implantable osmotic pump.
9697. The method of any one of claims 9388-9679 wherein the device is an ocular drug delivery pump.
9698. The method of any one of claims 9388-9679 wherein the device is metering system.
9699. The method of any one of claims 9388-9679 wherein the device is a peristaltic (roller) pump.
9700. The method of any one of claims 9388-9679 wherein the device is an electronically driven pump.
9701. The method of any one of claims 9388-9679 wherein the device is an elastomeric pump.
9702. The method of any one of claims 9388-9679 wherein the device is a spring contraction pump.
9703. The method of any one of claims 9388-9679 wherein the device is a gas-driven pump.
9704. The method of any one of claims 9388-9679 wherein the device is a hydraulic pump.
9705. The method of any one of claims 9388-9679 wherein the device is a piston-dependent pump.
9706. The method of any one of claims 9388-9679 wherein the device is a non-piston-dependent pump.
9707. The method of any one of claims 9388-9679 wherein the device is a dispensing chamber.
9708. The method of any one of claims 9388-9679 wherein the device is an infusion pump.
9709. The method of any one of claims 9388-9679 wherein the device is a passive pump.
9710. A method for making a device comprising: combining an implantable insulin pump and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
9711. The method of claim 9710 wherein the agent inhibits cell regeneration.
9712. The method of claim 9710 wherein the agent inhibits angiogenesis.
9713. The method of claim 9710 wherein the agent inhibits fibroblast migration.
9714. The method of claim 9710 wherein the agent inhibits fibroblast proliferation.
9715. The method of claim 9710 wherein the agent inhibits deposition of extracellular matrix.
9716. The method of claim 9710 wherein the agent inhibits tissue remodeling.
9717. The method of claim 9710 wherein the agent is an angiogenesis inhibitor.
9718. The method of claim 9710 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
9719. The method of claim 9710 wherein the agent is a chemokine receptor antagonist.
9720. The method of claim 9710 wherein the agent is a cell cycle inhibitor.
9721. The method of claim 9710 wherein the agent is a taxane.
9722. The method of claim 9710 wherein the agent is an antimicrotubule agent.
9723. The method of claim 9710 wherein the agent is paclitaxel.
9724. The method of claim 9710 wherein the agent is not paclitaxel.
9725. The method of claim 9710 wherein the agent is an analogue or derivative of paclitaxel.
9726. The method of claim 9710 wherein the agent is a vinca alkaloid.
9727. The method of claim 9710 wherein the agent is camptothecin or an analogue or derivative thereof.
9728. The method of claim 9710 wherein the agent is a podophyllotoxin.
9729. The method of claim 9710 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
9730. The method of claim 9710 wherein the agent is an anthracycline.
9731. The method of claim 9710 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
9732. The method of claim 9710 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
9733. The method of claim 9710 wherein the agent is a platinum compound.
9734. The method of claim 9710 wherein the agent is a nitrosourea.
9735. The method of claim 9710 wherein the agent is a nitroimidazole.
9736. The method of claim 9710 wherein the agent is a folic acid antagonist.
9737. The method of claim 9710 wherein the agent is a cytidine analogue.
9738. The method of claim 9710 wherein the agent is a pyrimidine analogue.
9739. The method of claim 9710 wherein the agent is a fluoropyrimidine analogue.
9740. The method of claim 9710 wherein the agent is a purine analogue.
9741. The method of claim 9710 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
9742. The method of claim 9710 wherein the agent is a hydroxyurea.
9743. The method of claim 9710 wherein the agent is a mytomicin or an analogue or derivative thereof.
9744. The method of claim 9710 wherein the agent is an alkyl sulfonate.
9745. The method of claim 9710 wherein the agent is a benzamide or an analogue or derivative thereof.
9746. The method of claim 9710 wherein the agent is a nicotinamide or an analogue or derivative thereof.
9747. The method of claim 9710 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
9748. The method of claim 9710 wherein the agent is a DNA alkylating agent.
9749. The method of claim 9710 wherein the agent is an antimicrotubule agent.
9750. The method of claim 9710 wherein the agent is a topoisomerase inhibitor.
9751. The method of claim 9710 wherein the agent is a DNA cleaving agent.
9752. The method of claim 9710 wherein the agent is an antimetabolite.
9753. The method of claim 9710 wherein the agent inhibits adenosine deaminase.
9754. The method of claim 9710 wherein the agent inhibits purine ring synthesis.
9755. The method of claim 9710 wherein the agent is a nucleotide interconversion inhibitor.
9756. The method of claim 9710 wherein the agent inhibits dihydrofolate reduction.
9757. The method of claim 9710 wherein the agent blocks thymidine monophosphate.
9758. The method of claim 9710 wherein the agent causes DNA damage.
9759. The method of claim 9710 wherein the agent is a DNA intercalation agent.
9760. The method of claim 9710 wherein the agent is a RNA synthesis inhibitor.
9761. The method of claim 9710 wherein the agent is a pyrimidine synthesis inhibitor.
9762. The method of claim 9710 wherein the agent inhibits ribonucleotide synthesis or function.
9763. The method of claim 9710 wherein the agent inhibits thymidine monophosphate synthesis or function.
9764. The method of claim 9710 wherein the agent inhibits DNA synthesis.
9765. The method of claim 9710 wherein the agent causes DNA adduct formation.
9766. The method of claim 9710 wherein the agent inhibits protein synthesis.
9767. The method of claim 9710 wherein the agent inhibits microtubule function.
9768. The method of claim 9710 wherein the agent is a cyclin dependent protein kinase inhibitor.
9769. The method of claim 9710 wherein the agent is an epidermal growth factor kinase inhibitor.
9770. The method of claim 9710 wherein the agent is an elastase inhibitor.
9771. The method of claim 9710 wherein the agent is a factor Xa inhibitor.
9772. The method of claim 9710 wherein the agent is a farnesyltransferase inhibitor.
9773. The method of claim 9710 wherein the agent is a fibrinogen antagonist
9774. The method of claim 9710 wherein the agent is a guanylate cyclase stimulant.
9775. The method of claim 9710 wherein the agent is a heat shock protein 90 antagonist.
9776. The method of claim 9710 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
9777. The method of claim 9710 wherein the agent is a guanylate cyclase stimulant.
9778. The method of claim 9710 wherein the agent is a HMGCoA reductase inhibitor.
9779. The method of claim 9710 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
9780. The method of claim 9710 wherein the agent is a hydroorotate dehydrogenase inhibitor.
9781. The method of claim 9710 wherein the agent is an IKK2 inhibitor.
9782. The method of claim 9710 wherein the agent is an IL-1 antagonist.
9783. The method of claim 9710 wherein the agent is an ICE antagonist.
9784. The method of claim 9710 wherein the agent is an IRAK antagonist
9785. The method of claim 9710 wherein the agent is an IL-4 agonist.
9786. The method of claim 9710 wherein the agent is an immunomodulatory agent.
9787. The method of claim 9710 wherein the agent is sirolimus or an analogue or derivative thereof.
9788. The method of claim 9710 wherein the agent is not sirolimus.
9789. The method of claim 9710 wherein the agent is everolimus or an analogue or derivative thereof.
9790. The method of claim 9710 wherein the agent is tacrolimus or an analogue or derivative thereof.
9791. The method of claim 9710 wherein the agent is not tacrolimus.
9792. The method of claim 9710 wherein the agent is biolmus or an analogue or derivative thereof.
9793. The method of claim 9710 wherein the agent is tresperimus or an analogue or derivative thereof.
9794. The method of claim 9710 wherein the agent is auranofin or an analogue or derivative thereof.
9795. The method of claim 9710 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
9796. The method of claim 9710 wherein the agent is gusperimus or an analogue or derivative thereof.
9797. The method of claim 9710 wherein the agent is pimecrolimus or an analogue or derivative thereof.
9798. The method of claim 9710 wherein the agent is ABT-578 or an analogue or derivative thereof.
9799. The method of claim 9710 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
9800. The method of claim 9710 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
9801. The method of claim 9710 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
9802. The method of claim 9710 wherein the agent is a leukotriene inhibitor.
9803. The method of claim 9710 wherein the agent is a MCP-1 antagonist.
9804. The method of claim 9710 wherein the agent is a MMP inhibitor.
9805. The method of claim 9710 wherein the agent is an NF kappa B inhibitor.
9806. The method of claim 9710 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
9807. The method of claim 9710 wherein the agent is an NO antagonist.
9808. The method of claim 9710 wherein the agent is a p38 MAP kinase inhibitor.
9809. The method of claim 9710 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
9810. The method of claim 9710 wherein the agent is a phosphodiesterase inhibitor.
9811. The method of claim 9710 wherein the agent is a TGF beta inhibitor.
9812. The method of claim 9710 wherein the agent is a thromboxane A2 antagonist.
9813. The method of claim 9710 wherein the agent is a TNF alpha antagonist.
9814. The method of claim 9710 wherein the agent is a TACE inhibitor.
9815. The method of claim 9710 wherein the agent is a tyrosine kinase inhibitor.
9816. The method of claim 9710 wherein the agent is a vitronectin inhibitor.
9817. The method of claim 9710 wherein the agent is a fibroblast growth factor inhibitor.
9818. The method of claim 9710 wherein the agent is a protein kinase inhibitor.
9819. The method of claim 9710 wherein the agent is a PDGF receptor kinase inhibitor.
9820. The method of claim 9710 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
9821. The method of claim 9710 wherein the agent is a retinoic acid receptor antagonist.
9822. The method of claim 9710 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
9823. The method of claim 9710 wherein the agent is a fibrinogen antagonist.
9824. The method of claim 9710 wherein the agent is an antimycotic agent.
9825. The method of claim 9710 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
9826. The method of claim 9710 wherein the agent is a bisphosphonate.
9827. The method of claim 9710 wherein the agent is a phospholipase A1 inhibitor.
9828. The method of claim 9710 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
9829. The method of claim 9710 wherein the agent is a macrolide antibiotic.
9830. The method of claim 9710 wherein the agent is a GPIIb/llla receptor antagonist.
9831. The method of claim 9710 wherein the agent is an endothelin receptor antagonist.
9832. The method of claim 9710 wherein the agent is a peroxisome proliferator-activated receptor agonist.
9833. The method of claim 9710 wherein the agent is an estrogen receptor agent.
9834. The method of claim 9710 wherein the agent is a somastostatin analogue.
9835. The method of claim 9710 wherein the agent is a neurokinin 1 antagonist.
9836. The method of claim 9710 wherein the agent is a neurokinin 3 antagonist.
9837. The method of claim 9710 wherein the agent is a VLA-4 antagonist.
9838. The method of claim 9710 wherein the agent is an osteoclast inhibitor.
9839. The method of claim 9710 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
9840. The method of claim 9710 wherein the agent is an angiotensin I converting enzyme inhibitor.
9841. The method of claim 9710 wherein the agent is an angiotensin II antagonist.
9842. The method of claim 9710 wherein the agent is an enkephalinase inhibitor.
9843. The method of claim 9710 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
9844. The method of claim 9710 wherein the agent is a protein kinase C inhibitor.
9845. The method of claim 9710 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
9846. The method of claim 9710 wherein the agent is a CXCR3 inhibitor.
9847. The method of claim 9710 wherein the agent is an Itk inhibitor.
9848. The method of claim 9710 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
9849. The method of claim 9710 wherein the agent is a PPAR agonist.
9850. The method of claim 9710 wherein the agent is an immunosuppressant
9851. The method of claim 9710 wherein the agent is an Erb inhibitor.
9852. The method of claim 9710 wherein the agent is an apoptosis agonist.
9853. The method of claim 9710 wherein the agent is a lipocortin agonist.
9854. The method of claim 9710 wherein the agent is a VCAM-1 antagonist.
9855. The method of claim 9710 wherein the agent is a collagen antagonist.
9856. The method of claim 9710 wherein the agent is an alpha 2 integrin antagonist.
9857. The method of claim 9710 wherein the agent is a TNF alpha inhibitor.
9858. The method of claim 9710 wherein the agent is a nitric oxide inhibitor
9859. The method of claim 9710 wherein the agent is a cathepsin inhibitor.
9860. The method of claim 9710 wherein the agent is not an anti- inflammatory agent.
9861. The method of claim 9710 wherein the agent is not a steroid.
9862. The method of claim 9710 wherein the agent is not a glucocorticosteroid.
9863. The method of claim 9710 wherein the agent is not dexamethasone.
9864. The method of claim 9710 wherein the agent is not beclomethasone.
9865. The method of claim 9710 wherein the agent is not dipropionate.
9866. The method of claim 9710 wherein the agent is not an anti- infective agent.
9867. The method of claim 9710 wherein the agent is not an antibiotic.
9868. The method of claim 9710 wherein the agent is not an anti- fungal agent.
9869. The method of claim 9710, wherein the composition comprises a polymer.
9870. The method of claim 9710, wherein the composition comprises a polymeric carrier.
9871. The method of claim 9710 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
9872. The method of claim 9710 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
9873. The method of claim 9710 wherein the device has a coating that comprises the anti-scarring agent
9874. The method of claim 9710, wherein the device has a coating that comprises the agent and is disposed on a surface of the pump.
9875. The method of claim 9710, wherein the device has a coating that comprises the agent and directly contacts the pump.
9876. The method of claim 9710, wherein the device has a coating that comprises the agent and indirectly contacts the pump.
9877. The method of claim 9710, wherein the device has a coating that comprises the agent and partially covers the pump.
9878. The method of claim 9710, wherein the device has a coating that comprises the agent and completely covers the pump.
9879. The method of claim 9710, wherein the device has a uniform coating.
9880. The method of claim 9710, wherein the device has a non- uniform coating.
9881. The method of claim 9710, wherein the device has a discontinuous coating.
9882. The method of claim 9710, wherein the device has a patterned coating.
9883. The method of claim 9710, wherein the device has a coating with a thickness of 100 μm or less.
9884. The method of claim 9710, wherein the device has a coating with a thickness of 10 μm or less.
9885. The method of claim 9710, wherein the device has a coating, and the coating adheres to the surface of the pump upon deployment of the pump.
9886. The method of claim 9710, wherein the device has a coating, and wherein the coating is stable at room temperature for a period of 1 year.
9887. The method of claim 9710, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1 % by weight.
9888. The method of claim 9710, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 1 % to about 10% by weight.
9889. The method of claim 9710, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
9890. The method of claim 9710, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
9891. The method of claim 9710, wherein the device has a coating, and wherein the coating further comprises a polymer.
9892. The method of claim 9710, wherein the device has a first coating having a first composition and a second coating having a second composition.
9893. The method of claim 9710, wherein the device has a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
9894. The method of claim 9710, wherein the composition comprises a polymer.
9895. The method of claim 9710, wherein the composition comprises a polymeric carrier.
9896. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a copolymer.
9897. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a block copolymer.
9898. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a random copolymer.
9899. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a biodegradable polymer.
9900. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-biodegradable polymer.
9901. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophilic polymer.
9902. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophobic polymer.
9903. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophilic domains.
9904. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophobic domains.
9905. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-conductive polymer.
9906. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an elastomer.
9907. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrogel.
9908. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a silicone polymer.
9909. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrocarbon polymer.
9910. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a styrene-derived polymer.
9911. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a butadiene polymer.
9912. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a macromer.
9913. The method of claim 9710, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a poly( ethylene glycol) polymer.
9914. The method of claim 9710 wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an amorphous polymer.
9915. The method of claim 9710, wherein the device comprises a lubricious coating.
9916. The method of claim 9710 wherein the anti-scarring agent is located within pores or holes of the device.
9917. The method of claim 9710 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
9918. The method of claim 9710, wherein the device comprises a second pharmaceutically active agent.
9919. The method of claim 9710 wherein the device comprises an anti-inflammatory agent
9920. The method of claim 9710 wherein the device comprises an agent that inhibits infection.
9921. The method of claim 9710 wherein the device comprises an agent that inhibits infection, and wherein the agent is an anthracycline.
9922. The method of claim 9710 wherein the device comprises an agent that inhibits infection, and wherein the agent is doxorubicin.
9923. The method of claim 9710 wherein the device comprises an agent that inhibits infection, and wherein the agent is mitoxantrone.
9924. The method of claim 9710 wherein the device comprises an agent that inhibits infection, and wherein the agent is a fluoropyrimidine.
9925. The method of claim 9710 wherein the device comprises an agent that inhibits infection, and wherein the agent is 5-fluorouracil (5-FU).
9926. The method of claim 9710 wherein the device comprises an agent that inhibits infection, and wherein the agent is a folic acid antagonist.
9927. The method of claim 9710 wherein the device comprises an agent that inhibits infection, and wherein the agent is methotrexate.
9928. The method of claim 9710 wherein the device comprises an agent that inhibits infection, and wherein the agent is a podophylotoxin.
9929. The method of claim 9710 wherein the device comprises an agent that inhibits infection, and wherein the agent is etoposide.
9930. The method of claim 9710 wherein the device comprises an agent that inhibits infection, and wherein the agent is a camptothecin.
9931. The method of claim 9710 wherein the device comprises an agent that inhibits infection, and wherein the agent is a hydroxyurea.
9932. The method of claim 9710 wherein the device comprises an agent that inhibits infection, and wherein the agent is a platinum complex.
9933. The method of claim 9710 wherein the device comprises an agent that inhibits infection, and wherein the agent is cisplatin.
9934. The method of claim 9710, further comprising an anti- thrombotic agent.
9935. The method of claim 9710 wherein the device comprises a visualization agent.
9936. The method of claim 9710 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
9937. The method of claim 9710 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises barium, tantalum, or technetium.
9938. The method of claim 9710 wherein the device comprises a visualization agent, and wherein the visualization agent is a MRI responsive material.
9939. The method of claim 9710 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a gadolinium chelate.
9940. The method of claim 9710 wherein the device comprises a visualization agent, and wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
9941. The method of claim 9710 wherein the device comprises a visualization agent, and wherein the visualization agent comprises an iron oxide compound.
9942. The method of claim 9710 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a dye, pigment, or colorant.
9943. The method of claim 9710 wherein the device comprises an echogenic material.
9944. The method of claim 9710 wherein the device comprises an echogenic material, and wherein the echogenic material is in the form of a coating.
9945. The method of claim 9710 wherein the device is sterile.
9946. The method of claim 9710 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
9947. The method of claim 9710 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is connective tissue.
9948. The method of claim 9710 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is muscle tissue.
9949. The method of claim 9710 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is nerve tissue.
9950. The method of claim 9710 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is epithelium tissue.
9951. The method of claim 9710 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
9952. The method of claim 9710 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
9953. The method of claim 9710 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
9954. The method of claim 9710 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
9955. The method of claim 9710 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
9956. The method of claim 9710 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
9957. The method of claim 9710 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
9958. The method of claim 9710 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
9959. The method of claim 9710 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
9960. The method of claim 9710 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
9961. The method of claim 9710 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
9962. The method of claim 9710 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
9963. The method of claim 9710 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
9964. The method of claim 9710 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9965. The method of claim 9710 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9966. The method of claim 9710 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9967. The method of claim 9710 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9968. The method of claim 9710 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti- scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9969. The method of claim 9710 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
9970. The method of claim 9710 wherein the combining is performed by direct affixing the agent or the composition to the pump.
9971. The method of claim 9710 wherein the combining is performed by spraying the agent or the component onto the pump.
9972. The method of claim 9710 wherein the combining is performed by electrospraying the agent or the composition onto the pump.
9973. The method of claim 9710 wherein the combining is performed by dipping the pump into a solution comprising the agent or the composition.
9974. The method of claim 9710 wherein the combining is performed by covalently attaching the agent or the composition to the pump.
9975. The method of claim 9710 wherein the combining is performed by non-covalently attaching the agent or the composition to the pump.
9976. The method of claim 9710 wherein the combining is performed by coating the pump with a substance that contains the agent or the composition.
9977. The method of claim 9710 wherein the combining is performed by coating the pump with a substance that absorbs the agent.
9978. The method of claim 9710 wherein the combining is performed by interweaving the pump with a thread composed of, or coated with, the agent or the composition.
9979. The method of claim 9710 wherein the combining is performed by completely covering the pump with a sleeve that contains the agent or the composition.
9980. The method of claim 9710 wherein the combining is performed by covering a portion of the pump with a sleeve that contains the agent or the composition.
9981. The method of claim 9710 wherein the combining is performed by completely covering the pump with a cover that contains the agent or the composition.
9982. The method of claim 9710 wherein the combining is performed by covering a portion of the pump with a cover that contains the agent or the composition.
9983. The method of claim 9710 wherein the combining is performed by completely covering the pump with an electrospun fabric that contains the agent or the composition.
9984. The method of claim 9710 wherein the combining is performed by covering a portion of the pump with an electrospun fabric that contains the agent or the composition.
9985. The method of claim 9710 wherein the combining is performed by completely covering the pump with a mesh that contains the agent or the composition.
9986. The method of claim 9710 wherein the combining is performed by covering a portion of the pump with a mesh that contains the agent or the composition.
9987. The method of claim 9710 wherein the combining is performed by constructing a portion of the pump with the agent or the composition.
9988. The method of claim 9710 wherein the combining is performed by impregnating the pump with the agent or the composition.
9989. The method of claim 9710 wherein the combining is performed by constructing a portion of the pump from a degradable polymer that releases the agent.
9990. The method of claim 9710 wherein the combining is performed by dipping the pump into a solution that comprise the agent and an inert solvent for the pump.
9991. The method of claim 9710 wherein the combining is performed by dipping the pump into a solution that comprises the agent and a solvent that will swill the pump.
9992. The method of claim 9710 wherein the combining is performed by dipping the pump into a solution that comprises the agent and a solvent that will dissolve the pump.
9993. The method of claim 9710 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and an inert solvent for the pump.
9994. The method of claim 9710 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and a solvent that will swill the pump.
9995. The method of claim 9710 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and a solvent that will dissolve the pump.
9996. The method of claim 9710 wherein the combining is performed by spraying the pump into a solution that comprises the agent and an inert solvent for the pump.
9997. The method of claim 9710 wherein the combining is performed by spraying the pump into a solution that comprises the agent and a solvent that will swill the pump.
9998. The method of claim 9710 wherein the combining is performed by spraying the pump into a solution that comprises the agent and a solvent that will dissolve the pump.
9999. The method of claim 9710 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and an inert solvent for the pump.
10000. The method of claim 9710 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and a solvent that will swill the pump.
10001. The method of claim 9710 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and a solvent that will dissolve the pump.
10002. A method for making a device comprising: combining an intrathecal drug delivery pump and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
10003. The method of claim 10002 wherein the agent inhibits cell regeneration.
10004. The method of claim 10002 wherein the agent inhibits angiogenesis.
10005. The method of claim 10002 wherein the agent inhibits fibroblast migration.
10006. The method of claim 10002 wherein the agent inhibits fibroblast proliferation.
10007. The method of claim 10002 wherein the agent inhibits deposition of extracellular matrix.
10008. The method of claim 10002 wherein the agent inhibits tissue remodeling.
10009. The method of claim 10002 wherein the agent is an angiogenesis inhibitor.
10010. The method of claim 10002 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
10011. The method of claim 10002 wherein the agent is a chemokine receptor antagonist.
10012. The method of claim 10002 wherein the agent is a cell cycle inhibitor.
10013. The method of claim 10002 wherein the agent is a taxane.
10014. The method of claim 10002 wherein the agent is an antimicrotubule agent.
10015. The method of claim 10002 wherein the agent is paclitaxel.
10016. The method of claim 10002 wherein the agent is not paclitaxel.
10017. The method of claim 10002 wherein the agent is an analogue or derivative of paclitaxel.
10018. The method of claim 10002 wherein the agent is a vinca alkaloid.
10019. The method of claim 10002 wherein the agent is camptothecin or an analogue or derivative thereof.
10020. The method of claim 10002 wherein the agent is a podophyllotoxin.
10021. The method of claim 10002 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
10022. The method of claim 10002 wherein the agent is an anthracycline.
10023. The method of claim 10002 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
10024. The method of claim 10002 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
10025. The method of claim 10002 wherein the agent is a platinum compound.
10026. The method of claim 10002 wherein the agent is a nitrosourea.
10027. The method of claim 10002 wherein the agent is a nitroimidazole.
10028. The method of claim 10002 wherein the agent is a folic acid antagonist.
10029. The method of claim 10002 wherein the agent is a cytidine analogue.
10030. The method of claim 10002 wherein the agent is a pyrimidine analogue.
10031. The method of claim 10002 wherein the agent is a fluoropyrimidine analogue.
10032. The method of claim 10002 wherein the agent is a purine analogue.
10033. The method of claim 10002 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
10034. The method of claim 10002 wherein the agent is a hydroxyurea.
10035. The method of claim 10002 wherein the agent is a mytomicin or an analogue or derivative thereof.
10036. The method of claim 10002 wherein the agent is an alkyl sulfonate.
10037. The method of claim 10002 wherein the agent is a benzamide or an analogue or derivative thereof.
10038. The method of claim 10002 wherein the agent is a nicotinamide or an analogue or derivative thereof.
10039. The method of claim 10002 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
10040. The method of claim 10002 wherein the agent is a DNA alkylating agent.
10041. The method of claim 10002 wherein the agent is an antimicrotubule agent
10042. The method of claim 10002 wherein the agent is a topoisomerase inhibitor.
10043. The method of claim 10002 wherein the agent is a DNA cleaving agent.
10044. The method of claim 10002 wherein the agent is an antimetabolite.
10045. The method of claim 10002 wherein the agent inhibits adenosine deaminase.
10046. The method of claim 10002 wherein the agent inhibits purine ring synthesis^
10047. The method of claim 10002 wherein the agent is a nucleotide interconversion inhibitor.
10048. The method of claim 10002 wherein the agent inhibits dihydrofolate reduction.
10049. The method of claim 10002 wherein the agent blocks thymidine monophosphate.
10050. The method of claim 10002 wherein the agent causes DNA damage.
10051. The method of claim 10002 wherein the agent is a DNA intercalation agent.
10052. The method of claim 10002 wherein the agent is a RNA synthesis inhibitor.
10053. The method of claim 10002 wherein the agent is a pyrimidine synthesis inhibitor.
10054. The method of claim 10002 wherein the agent inhibits ribonucleotide synthesis or function.
10055. The method of claim 10002 wherein the agent inhibits thymidine monophosphate synthesis or function.
10056. The method of claim 10002 wherein the agent inhibits DNA synthesis.
10057. The method of claim 10002 wherein the agent causes DNA adduct formation.
10058. The method of claim 10002 wherein the agent inhibits protein synthesis.
10059. The method of claim 10002 wherein the agent inhibits microtubule function.
10060. The method of claim 10002 wherein the agent is a cyclin dependent protein kinase inhibitor.
10061. The method of claim 10002 wherein the agent is an epidermal growth factor kinase inhibitor.
10062. The method of claim 10002 wherein the agent is an elastase inhibitor.
10063. The method of claim 10002 wherein the agent is a factor Xa inhibitor.
10064. The method of claim 10002 wherein the agent is a farnesyltransferase inhibitor.
10065. The method of claim 10002 wherein the agent is a fibrinogen antagonist.
10066. The method of claim 10002 wherein the agent is a guanylate cyclase stimulant.
10067. The method of claim 10002 wherein the agent is a heat shock protein 90 antagonist.
10068. The method of claim 10002 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
10069. The method of claim 10002 wherein the agent is a guanylate cyclase stimulant.
10070. The method of claim 10002 wherein the agent is a HMGCoA reductase inhibitor.
10071. The method of claim 10002 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
10072. The method of claim 10002 wherein the agent is a hydroorotate dehydrogenase inhibitor.
10073. The method of claim 10002 wherein the agent is an IKK2 inhibitor.
10074. The method of claim 10002 wherein the agent is an IL-1 antagonist.
10075. The method of claim 10002 wherein the agent is an ICE antagonist.
10076. The method of claim 10002 wherein the agent is an IRAK antagonist.
10077. The method of claim 10002 wherein the agent is an IL-4 agonist.
10078. The method of claim 10002 wherein the agent is an immunomodulatory agent
10079. The method of claim 10002 wherein the agent is sirolimus or an analogue or derivative thereof.
10080. The method of claim 10002 wherein the agent is not sirolimus.
10081. The method of claim 10002 wherein the agent is everolimus or an analogue or derivative thereof.
10082. The method of claim 10002 wherein the agent is tacrolimus or an analogue or derivative thereof.
10083. The method of claim 10002 wherein the agent is not tacrolimus.
10084. The method of claim 10002 wherein the agent is biolmus or an analogue or derivative thereof.
10085. The method of claim 10002 wherein the agent is tresperimus or an analogue or derivative thereof.
10086. The method of claim 10002 wherein the agent is auranofin or an analogue or derivative thereof.
10087. The method of claim 10002 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
10088. The method of claim 10002 wherein the agent is gusperimus or an analogue or derivative thereof.
10089. The method of claim 10002 wherein the agent is pimecrolimus or an analogue or derivative thereof.
10090. The method of claim 10002 wherein the agent is ABT-578 or an analogue or derivative thereof.
10091. The method of claim 10002 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
10092. The method of claim 10002 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
10093. The method of claim 10002 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D or an analogue or derivative thereof.
10094. The method of claim 10002 wherein the agent is a leukotriene inhibitor.
10095. The method of claim 10002 wherein the agent is a MCP-1 antagonist.
10096. The method of claim 10002 wherein the agent is a MMP inhibitor.
10097. The method of claim 10002 wherein the agent is an NF kappa B inhibitor.
10098. The method of claim 10002 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
10099. The method of claim 10002 wherein the agent is an NO antagonist.
10100. The method of claim 10002 wherein the agent is a p38 MAP kinase inhibitor.
10101. The method of claim 10002 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
10102. The method of claim 10002 wherein the agent is a phosphodiesterase inhibitor.
10103. The method of claim 10002 wherein the agent is a TGF beta inhibitor.
10104. The method of claim 10002 wherein the agent is a thromboxane A2 antagonist.
10105. The method of claim 10002 wherein the agent is a TNF alpha antagonist.
10106. The method of claim 10002 wherein the agent is a TACE inhibitor.
10107. The method of claim 10002 wherein the agent is a tyrosine kinase inhibitor.
10108. The method of claim 10002 wherein the agent is a vitronectin inhibitor.
10109. The method of claim 10002 wherein the agent is a fibroblast growth factor inhibitor.
10110. The method of claim 10002 wherein the agent is a protein kinase inhibitor.
10111. The method of claim 10002 wherein the agent is a PDGF receptor kinase inhibitor.
10112. The method of claim 10002 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
10113. The method of claim 10002 wherein the agent is a retinoic acid receptor antagonist.
10114. The method of claim 10002 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
10115. The method of claim 10002 wherein the agent is a fibrinogen antagonist
10116. The method of claim 10002 wherein the agent is an antimycotic agent.
10117. The method of claim 10002 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
10118. The method of claim 10002 wherein the agent is a bisphosphonate.
10119. The method of claim 10002 wherein the agent is a phospholipase A1 inhibitor.
10120. The method of claim 10002 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
10121. The method of claim 10002 wherein the agent is a macrolide antibiotic.
10122. The method of claim 10002 wherein the agent is a GPIIb/llla receptor antagonist
10123. The method of claim 10002 wherein the agent is an endothelin receptor antagonist.
10124. The method of claim 10002 wherein the agent is a peroxisome proliferator-activated receptor agonist.
10125. The method of claim 10002 wherein the agent is an estrogen receptor agent.
10126. The method of claim 10002 wherein the agent is a somastostatin analogue.
10127. The method of claim 10002 wherein the agent is a neurokinin 1 antagonist.
10128. The method of claim 10002 wherein the agent is a neurokinin 3 antagonist.
10129. The method of claim 10002 wherein the agent is a VLA-4 antagonist.
10130. The method of claim 10002 wherein the agent is an osteoclast inhibitor.
10131. The method of claim 10002 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
10132. The method of claim 10002 wherein the agent is an angiotensin I converting enzyme inhibitor.
10133. The method of claim 10002 wherein the agent is an angiotensin II antagonist.
10134. The method of claim 10002 wherein the agent is an enkephalinase inhibitor.
10135. The method of claim 10002 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
10136. The method of claim 10002 wherein the agent is a protein kinase C inhibitor.
10137. The method of claim 10002 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
10138. The method of claim 10002 wherein the agent is a CXCR3 inhibitor.
10139. The method of claim 10002 wherein the agent is an Itk inhibitor.
10140. The method of claim 10002 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
10141. The method of claim 10002 wherein the agent is a PPAR agonist.
10142. The method of claim 10002 wherein the agent is an immunosuppressant
10143. The method of claim 10002 wherein the agent is an Erb inhibitor.
10144. The method of claim 10002 wherein the agent is an apoptosis agonist.
10145. The method of claim 10002 wherein the agent is a lipocortin agonist.
10146. The method of claim 10002 wherein the agent is a VCAM-1 antagonist
10147. The method of claim 10002 wherein the agent is a collagen antagonist.
10148. The method of claim 10002 wherein the agent is an alpha 2 integrin antagonist.
10149. The method of claim 10002 wherein the agent is a TNF alpha inhibitor.
10150. The method of claim 10002 wherein the agent is a nitric oxide inhibitor
10151. The method of claim 10002 wherein the agent is a cathepsin inhibitor.
10152. The method of claim 10002 wherein the agent is not an anti-inflammatory agent.
10153. The method of claim 10002 wherein the agent is not a steroid.
10154. The method of claim 10002 wherein the agent is not a glucocorticosteroid.
10155. The method of claim 10002 wherein the agent is not dexamethasone.
10156. The method of claim 10002 wherein the agent is not beclomethasone.
10157. The method of claim 10002 wherein the agent is not dipropionate.
10158. The method of claim 10002 wherein the agent is not an anti-infective agent
10159. The method of claim 10002 wherein the agent is not an antibiotic.
10160. The method of claim 10002 wherein the agent is not an anti-fungal agent.
10161. The method of claim 10002, wherein the composition comprises a polymer.
10162. The method of claim 10002, wherein the composition comprises a polymeric carrier.
10163. The method of claim 10002 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
10164. The method of claim 10002 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
10165. The method of claim 10002 wherein the device has a coating that comprises the anti-scarring agent.
10166. The method of claim 10002, wherein the device has a coating that comprises the agent and is disposed on a surface of the pump.
10167. The method of claim 10002, wherein the device has a coating that comprises the agent and directly contacts the pump.
10168. The method of claim 10002, wherein the device has a coating that comprises the agent and indirectly contacts the pump.
10169. The method of claim 10002, wherein the device has a coating that comprises the agent and partially covers the pump.
10170. The method of claim 10002, wherein the device has a coating that comprises the agent and completely covers the pump.
10171. The method of claim 10002, wherein the device has a uniform coating.
10172. The method of claim 10002, wherein the device has a non-uniform coating.
10173. The method of claim 10002, wherein the device has a discontinuous coating.
10174. The method of claim 10002, wherein the device has a patterned coating.
10175. The method of claim 10002, wherein the device has a coating with a thickness of 100 μm or less.
10176. The method of claim 10002, wherein the device has a coating with a thickness of 10 μm or less.
10177. The method of claim 10002, wherein the device has a coating, and the coating adheres to the surface of the pump upon deployment of the pump.
10178. The method of claim 10002, wherein the device has a coating, and wherein the coating is stable at room temperature for a period of 1 year.
10179. The method of claim 10002, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1 % by weight.
10180. The method of claim 10002, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
10181. The method of claim 10002, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
10182. The method of claim 10002, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
10183. The method of claim 10002, wherein the device has a coating, and wherein the coating further comprises a polymer.
10184. The method of claim 10002, wherein the device has a first coating having a first composition and a second coating having a second composition.
10185. The method of claim 10002, wherein the device has a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
10186. The method of claim 10002, wherein the composition comprises a polymer.
10187. The method of claim 10002, wherein the composition comprises a polymeric carrier.
10188. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a copolymer.
10189. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a block copolymer.
10190. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a random copolymer.
10191. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a biodegradable polymer.
10192. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-biodegradable polymer.
10193. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophilic polymer.
10194. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophobic polymer.
10195. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophilic domains.
10196. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophobic domains.
10197. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-conductive polymer.
10198. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an elastomer.
10199. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrogel.
10200. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a silicone polymer.
10201. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrocarbon polymer.
10202. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a styrene-derived polymer.
10203. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a butadiene polymer.
10204. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a macromer.
10205. The method of claim 10002, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a poly( ethylene glycol) polymer.
10206. The method of claim 10002 wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an amorphous polymer.
10207. The method of claim 10002, wherein the device comprises a lubricious coating.
10208. The method of claim 10002 wherein the anti-scarring agent is located within pores or holes of the device.
10209. The method of claim 10002 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
10210. The method of claim 10002, wherein the device comprises a second pharmaceutically active agent.
10211. The method of claim 10002 wherein the device comprises an anti-inflammatory agent.
10212. The method of claim 10002 wherein the device comprises an agent that inhibits infection.
10213. The method of claim 10002 wherein the device comprises an agent that inhibits infection, and wherein the agent is an anthracycline.
10214. The method of claim 10002 wherein the device comprises an agent that inhibits infection, and wherein the agent is doxorubicin.
10215. The method of claim 10002 wherein the device comprises an agent that inhibits infection, and wherein the agent is mitoxantrone.
10216. The method of claim 10002 wherein the device comprises an agent that inhibits infection, and wherein the agent is a fluoropyrimidine.
10217. The method of claim 10002 wherein the device comprises an agent that inhibits infection, and wherein the agent is 5-fluorouracil (5-FU).
10218. The method of claim 10002 wherein the device comprises an agent that inhibits infection, and wherein the agent is a folic acid antagonist
10219. The method of claim 10002 wherein the device comprises an agent that inhibits infection, and wherein the agent is methotrexate.
10220. The method of claim 10002 wherein the device comprises an agent that inhibits infection, and wherein the agent is a podophylotoxin.
10221. The method of claim 10002 wherein the device comprises an agent that inhibits infection, and wherein the agent is etoposide.
10222. The method of claim 10002 wherein the device comprises an agent that inhibits infection, and wherein the agent is a camptothecin.
10223. The method of claim 10002 wherein the device comprises an agent that inhibits infection, and wherein the agent is a hydroxyurea.
10224. The method of claim 10002 wherein the device comprises an agent that inhibits infection, and wherein the agent is a platinum complex.
10225. The method of claim 10002 wherein the device comprises an agent that inhibits infection, and wherein the agent is cisplatin.
10226. The method of claim 10002, further comprising an anti- thrombotic agent
10227. The method of claim 10002 wherein the device comprises a visualization agent.
10228. The method of claim 10002 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
10229. The method of claim 10002 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises barium, tantalum, or technetium.
10230. The method of claim 10002 wherein the device comprises a visualization agent, and wherein the visualization agent is a MRI responsive material.
10231. The method of claim 10002 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a gadolinium chelate.
10232. The method of claim 10002 wherein the device comprises a visualization agent, and wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
10233. The method of claim 10002 wherein the device comprises a visualization agent, and wherein the visualization agent comprises an iron oxide compound.
10234. The method of claim 10002 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a dye, pigment, or colorant
10235. The method of claim 10002 wherein the device comprises an echogenic material.
10236. The method of claim 10002 wherein the device comprises an echogenic material, and wherein the echogenic material is in the form of a coating.
10237. The method of claim 10002 wherein the device is sterile.
10238. The method of claim 10002 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
10239. The method of claim 10002 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is connective tissue.
10240. The method of claim 10002 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is muscle tissue.
10241. The method of claim 10002 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is nerve tissue.
10242. The method of claim 10002 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is epithelium tissue.
10243. The method of claim 10002 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to- about 1 year.
10244. The method of claim 10002 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
10245. The method of claim 10002 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
10246. The method of claim 10002 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
10247. The method of claim 10002 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
10248. The method of claim 10002 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
10249. The method of claim 10002 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
10250. The method of claim 10002 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
10251. The method of claim 10002 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
10252. The method of claim 10002 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
10253. The method of claim 10002 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
10254. The method of claim 10002 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
10255. The method of claim 10002 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
10256. The method of claim 10002 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10257. The method of claim 10002 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10258. The method of claim 10002 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm 2 of device surface to which the anti-scarring agent is applied.
10259. The method of claim 10002 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10260. The method of claim 10002 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10261. The method of claim 10002 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10262. The method of claim 10002 wherein the combining is performed by direct affixing the agent or the composition to the pump.
10263. The method of claim 10002 wherein the combining is performed by spraying the agent or the component onto the pump.
10264. The method of claim 10002 wherein the combining is performed by electrospraying the agent or the composition onto the pump.
10265. The method of claim 10002 wherein the combining is performed by dipping the pump into a solution comprising the agent or the composition.
10266. The method of claim 10002 wherein the combining is performed by covalently attaching the agent or the composition to the pump.
10267. The method of claim 10002 wherein the combining is performed by non-covalently attaching the agent or the composition to the pump.
10268. The method of claim 10002 wherein the combining is performed by coating the pump with a substance that contains the agent or the composition.
10269. The method of claim 10002 wherein the combining is performed by coating the pump with a substance that absorbs the agent.
10270. The method of claim 10002 wherein the combining is performed by interweaving the pump with a thread composed of, or coated with, the agent or the composition.
10271. The method of claim 10002 wherein the combining is performed by completely covering the pump with a sleeve that contains the agent or the composition.
10272. The method of claim 10002 wherein the combining is performed by covering a portion of the pump with a sleeve that contains the agent or the composition.
10273. The method of claim 10002 wherein the combining is performed by completely covering the pump with a cover that contains the agent or the composition.
10274. The method of claim 10002 wherein the combining is performed by covering a portion of the pump with a cover that contains the agent or the composition.
10275. The method of claim 10002 wherein the combining is performed by completely covering the pump with an electrospun fabric that contains the agent or the composition.
10276. The method of claim 10002 wherein the combining is performed by covering a portion of the pump with an electrospun fabric that contains the agent or the composition.
10277. The method of claim 10002 wherein the combining is performed by completely covering the pump with a mesh that contains the agent or the composition.
10278. The method of claim 10002 wherein the combining is performed by covering a portion of the pump with a mesh that contains the agent or the composition.
10279. The method of claim 10002 wherein the combining is performed by constructing a portion of the pump with the agent or the composition.
10280. The method of claim 10002 wherein the combining is performed by impregnating the pump with the agent or the composition.
10281. The method of claim 10002 wherein the combining is performed by constructing a portion of the pump from a degradable polymer that releases the agent.
10282. The method of claim 10002 wherein the combining is performed by dipping the pump into a solution that comprise the agent and an inert solvent for the pump.
10283. The method of claim 10002 wherein the combining is performed by dipping the pump into a solution that comprises the agent and a solvent that will swill the pump.
10284. The method of claim 10002 wherein the combining is performed by dipping the pump into a solution that comprises the agent and a solvent that will dissolve the pump.
10285. The method of claim 10002 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and an inert solvent for the pump.
10286. The method of claim 10002 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and a solvent that will swill the pump.
10287. The method of claim 10002 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and a solvent that will dissolve the pump.
10288. The method of claim 10002 wherein the combining is performed by spraying the pump into a solution that comprises the agent and an inert solvent for the pump.
10289. The method of claim 10002 wherein the combining is performed by spraying the pump into a solution that comprises the agent and a solvent that will swill the pump.
10290. The method of claim 10002 wherein the combining is performed by spraying the pump into a solution that comprises the agent and a solvent that will dissolve the pump.
10291. The method of claim 10002 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and an inert solvent for the pump.
10292. The method of claim 10002 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and a solvent that will swill the pump.
10293. The method of claim 10002 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and a solvent that will dissolve the pump.
10294. The method of any one of claims 10002-10293 wherein the device is adapted for delivering pain medication directly into the cerebrospinal fluid of the intrathecal space surrounding the spinal cord.
10295. The method of any one of claims 10002-10293 wherein the device is adapted for delivering a drug to the brain.
10296. The method of any one of claims 10002-10293 wherein the device is adapted for intrathecal delivering baclofen.
10297. The method of any one of claims 10002-10293 wherein the device further comprises an intraspinal catheter.
10298. The method of any one of claims 10002-10293 wherein the device further comprises a second intrathecal drug delivery pump.
10299. The method of any one of claims 10002-10293 wherein the device further comprises a catheter and an electrode.
10300. A method for making a device comprising: combining an implantable drug delivery pump for chemotherapy and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
10301. The method of claim 10300 wherein the agent inhibits cell regeneration.
10302. The method of claim 10300 wherein the agent inhibits angiogenesis.
10303. The method of claim 10300 wherein the agent inhibits fibroblast migration.
10304. The method of claim 10300 wherein the agent inhibits fibroblast proliferation.
10305. The method of claim 10300 wherein the agent inhibits deposition of extracellular matrix.
10306. The method of claim 10300 wherein the agent inhibits tissue remodeling.
10307. The method of claim 10300 wherein the agent is an angiogenesis inhibitor.
10308. The method of claim 10300 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
10309. The method of claim 10300 wherein the agent is a chemokine receptor antagonist.
10310. The method of claim 10300 wherein the agent is a cell cycle inhibitor.
10311. The method of claim 10300 wherein the agent is a taxane.
10312. The method of claim 10300 wherein the agent is an antimicrotubule agent.
10313. The method of claim 10300 wherein the agent is paclitaxel.
10314. The method of claim 10300 wherein the agent is not paclitaxel.
10315. The method of claim 10300 wherein the agent is an analogue or derivative of paclitaxel.
10316. The method of claim 10300 wherein the agent is a vinca alkaloid.
10317. The method of claim 10300 wherein the agent is camptothecin or an analogue or derivative thereof.
10318. The method of claim 10300 wherein the agent is a podophyllotoxin.
10319. The method of claim 10300 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
10320. The method of claim 10300 wherein the agent is an anthracycline.
10321. The method of claim 10300 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
10322. The method of claim 10300 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
10323. The method of claim 10300 wherein the agent is a platinum compound.
10324. The method of claim 10300 wherein the agent is a nitrosourea.
10325. The method of claim 10300 wherein the agent is a nitroimidazole.
10326. The method of claim 10300 wherein the agent is a folic acid antagonist.
10327. The method of claim 10300 wherein the agent is a cytidine analogue.
10328. The method of claim 10300 wherein the agent is a pyrimidine analogue.
10329. The method of claim 10300 wherein the agent is a fluoropyrimidine analogue.
10330. The method of claim 10300 wherein the agent is a purine analogue.
10331. The method of claim 10300 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
10332. The method of claim 10300 wherein the agent is a hydroxyurea.
10333. The method of claim 10300 wherein the agent is a mytomicin or an analogue or derivative thereof.
10334. The method of claim 10300 wherein the agent is an alkyl sulfonate.
10335. The method of claim 10300 wherein the agent is a benzamide or an analogue or derivative thereof.
10336. The method of claim 10300 wherein the agent is a nicotinamide or an analogue or derivative thereof.
10337. The method of claim 10300 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
10338. The method of claim 10300 wherein the agent is a DNA alkylating agent.
10339. The method of claim 10300 wherein the agent is an antimicrotubule agent.
10340. The method of claim 10300 wherein the agent is a topoisomerase inhibitor.
10341. The method of claim 10300 wherein the agent is a DNA cleaving agent.
10342. The method of claim 10300 wherein the agent is an antimetabolite.
10343. The method of claim 10300 wherein the agent inhibits adenosine deaminase.
10344. The method of claim 10300 wherein the agent inhibits purine ring synthesis.
10345. The method of claim 10300 wherein the agent is a nucleotide interconversion inhibitor.
10346. The method of claim 10300 wherein the agent inhibits dihydrofolate reduction.
10347. The method of claim 10300 wherein the agent blocks thymidine monophosphate.
10348. The method of claim 10300 wherein the agent causes DNA damage.
10349. The method of claim 10300 wherein the agent is a DNA intercalation agent.
10350. The method of claim 10300 wherein the agent is a RNA synthesis inhibitor.
10351. The method of claim 10300 wherein the agent is a pyrimidine synthesis inhibitor.
10352. The method of claim 10300 wherein the agent inhibits ribonucleotide synthesis or function.
10353. The method of claim 10300 wherein the agent inhibits thymidine monophosphate synthesis or function.
10354. The method of claim 10300 wherein the agent inhibits DNA synthesis.
10355. The method of claim 10300 wherein the agent causes DNA adduct formation.
10356. The method of claim 10300 wherein the agent inhibits protein synthesis.
10357. The method of claim 10300 wherein the agent inhibits microtubule function.
10358. The method of claim 10300 wherein the agent is a cyclin dependent protein kinase inhibitor.
10359. The method of claim 10300 wherein the agent is an epidermal growth factor kinase inhibitor.
10360. The method of claim 10300 wherein the agent is an elastase inhibitor.
10361. The method of claim 10300 wherein the agent is a factor Xa inhibitor.
10362. The method of claim 10300 wherein the agent is a farnesyltransferase inhibitor.
10363. The method of claim 10300 wherein the agent is a fibrinogen antagonist.
10364. The method of claim 10300 wherein the agent is a guanylate cyclase stimulant.
10365. The method of claim 10300 wherein the agent is a heat shock protein 90 antagonist.
10366. The method of claim 10300 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
10367. The method of claim 10300 wherein the agent is a guanylate cyclase stimulant.
10368. The method of claim 10300 wherein the agent is a HMGCoA reductase inhibitor.
10369. The method of claim 10300 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
10370. The method of claim 10300 wherein the agent is a hydroorotate dehydrogenase inhibitor.
10371. The method of claim 10300 wherein the agent is an IKK2 inhibitor.
10372. The method of claim 10300 wherein the agent is an IL-1 antagonist.
10373. The method of claim 10300 wherein the agent is an ICE antagonist.
10374. The method of claim 10300 wherein the agent is an IRAK antagonist
10375. The method of claim 10300 wherein the agent is an IL-4 agonist.
10376. The method of claim 10300 wherein the agent is an immunomodulatory agent.
10377. The method of claim 10300 wherein the agent is sirolimus or an analogue or derivative thereof.
10378. The method of claim 10300 wherein the agent is not sirolimus.
10379. The method of claim 10300 wherein the agent is everolimus or an analogue or derivative thereof.
10380. The method of claim 10300 wherein the agent is tacrolimus or an analogue or derivative thereof.
10381. The method of claim 10300 wherein the agent is not tacrolimus.
10382. The method of claim 10300 wherein the agent is biolmus or an analogue or derivative thereof.
10383. The method of claim 10300 wherein the agent is tresperimus or an analogue or derivative thereof.
10384. The method of claim 10300 wherein the agent is auranofin or an analogue or derivative thereof.
10385. The method of claim 10300 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
10386. The method of claim 10300 wherein the agent is gusperimus or an analogue or derivative thereof.
10387. The method of claim 10300 wherein the agent is pimecrolimus or an analogue or derivative thereof.
10388. The method of claim 10300 wherein the agent is ABT-578 or an analogue or derivative thereof.
10389. The method of claim 10300 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
10390. The method of claim 10300 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
10391. The method of claim 10300 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
10392. The method of claim 10300 wherein the agent is a leukotriene inhibitor.
10393. The method of claim 10300 wherein the agent is a MCP-1 antagonist.
10394. The method of claim 10300 wherein the agent is a MMP inhibitor.
10395. The method of claim 10300 wherein the agent is an NF kappa B inhibitor.
10396. The method of claim 10300 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
10397. The method of claim 10300 wherein the agent is an NO antagonist.
10398. The method of claim 10300 wherein the agent is a p38 MAP kinase inhibitor.
10399. The method of claim 10300 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
10400. The method of claim 10300 wherein the agent is a phosphodiesterase inhibitor.
10401. The method of claim 10300 wherein the agent is a TGF beta inhibitor.
10402. The method of claim 10300 wherein the agent is a thromboxane A2 antagonist.
10403. The method of claim 10300 wherein the agent is a TNF alpha antagonist.
10404. The method of claim 10300 wherein the agent is a TACE inhibitor.
10405. The method of claim 10300 wherein the agent is a tyrosine kinase inhibitor.
10406. The method of claim 10300 wherein the agent is a vitronectin inhibitor.
10407. The method of claim 10300 wherein the agent is a fibroblast growth factor inhibitor.
10408. The method of claim 10300 wherein the agent is a protein kinase inhibitor.
10409. The method of claim 10300 wherein the agent is a PDGF receptor kinase inhibitor.
10410. The method of claim 10300 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
10411. The method of claim 10300 wherein the agent is a retinoic acid receptor antagonist.
10412. The method of claim 10300 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
10413. The method of claim 10300 wherein the agent is a fibrinogen antagonist.
10414. The method of claim 10300 wherein the agent is an antimycotic agent.
10415. The method of claim 10300 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
10416. The method of claim 10300 wherein the agent is a bisphosphonate.
10417. The method of claim 10300 wherein the agent is a phospholipase A1 inhibitor.
10418. The method of claim 10300 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
10419. The method of claim 10300 wherein the agent is a macrolide antibiotic.
10420. The method of claim 10300 wherein the agent is a GPIIb/llla receptor antagonist.
10421. The method of claim 10300 wherein the agent is an endothelin receptor antagonist.
10422. The method of claim 10300 wherein the agent is a peroxisome proliferator-activated receptor agonist.
10423. The method of claim 10300 wherein the agent is an estrogen receptor agent.
10424. The method of claim 10300 wherein the agent is a somastostatin analogue.
10425. The method of claim 10300 wherein the agent is a neurokinin 1 antagonist.
10426. The method of claim 10300 wherein the agent is a neurokinin 3 antagonist.
10427. The method of claim 10300 wherein the agent is a VLA-4 antagonist.
10428. The method of claim 10300 wherein the agent is an osteoclast inhibitor.
10429. The method of claim 10300 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
10430. The method of claim 10300 wherein the agent is an angiotensin I converting enzyme inhibitor.
10431. The method of claim 10300 wherein the agent is an angiotensin II antagonist.
10432. The method of claim 10300 wherein the agent is an enkephalinase inhibitor.
10433. The method of claim 10300 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
10434. The method of claim 10300 wherein the agent is a protein kinase C inhibitor.
10435. The method of claim 10300 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
10436. The method of claim 10300 wherein the agent is a CXCR3 inhibitor.
10437. The method of claim 10300 wherein the agent is an Itk inhibitor.
10438. The method of claim 10300 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
10439. The method of claim 10300 wherein the agent is a PPAR agonist
10440. The method of claim 10300 wherein the agent is an immunosuppressant.
10441. The method of claim 10300 wherein the agent is an Erb inhibitor.
10442. The method of claim 10300 wherein the agent is an apoptosis agonist.
10443. The method of claim 10300 wherein the agent is a lipocortin agonist.
10444. The method of claim 10300 wherein the agent is a VCAM-1 antagonist.
10445. The method of claim 10300 wherein the agent is a collagen antagonist.
10446. The method of claim 10300 wherein the agent is an alpha 2 integrin antagonist
10447. The method of claim 10300 wherein the agent is a TNF alpha inhibitor.
10448. The method of claim 10300 wherein the agent is a nitric oxide inhibitor
10449. The method of claim 10300 wherein the agent is a cathepsin inhibitor.
10450. The method of claim 10300 wherein the agent is not an anti-inflammatory agent.
10451. The method of claim 10300 wherein the agent is not a steroid.
10452. The method of claim 10300 wherein the agent is not a glucocorticosteroid.
10453. The method of claim 10300 wherein the agent is not dexamethasone.
10454. The method of claim 10300 wherein the agent is not beclomethasone.
10455. The method of claim 10300 wherein the agent is not dipropionate.
10456. The method of claim 10300 wherein the agent is not an anti-infective agent.
10457. The method of claim 10300 wherein the agent is not an antibiotic.
10458. The method of claim 10300 wherein the agent is not an anti-fungal agent.
10459. The method of claim 10300, wherein the composition comprises a polymer.
10460. The method of claim 10300, wherein the composition comprises a polymeric carrier.
10461. The method of claim 10300 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
10462. The method of claim 10300 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
10463. The method of claim 10300 wherein the device has a coating that comprises the anti-scarring agent.
10464. The method of claim 10300, wherein the device has a coating that comprises the agent and is disposed on a surface of the pump.
10465. The method of claim 10300, wherein the device has a coating that comprises the agent and directly contacts the pump.
10466. The method of claim 10300, wherein the device has a coating that comprises the agent and indirectly contacts the pump.
10467. The method of claim 10300, wherein the device has a coating that comprises the agent and partially covers the pump.
10468. The method of claim 10300, wherein the device has a coating that comprises the agent and completely covers the pump.
10469. The method of claim 10300, wherein the device has a uniform coating.
10470. The method of claim 10300, wherein the device has a non-uniform coating. 2005/051871
10471. The method of claim 10300, wherein the device has a discontinuous coating.
10472. The method of claim 10300, wherein the device has a patterned coating.
10473. The method of claim 10300, wherein the device has a coating with a thickness of 100 μm or less.
10474. The method of claim 10300, wherein the device has a coating with a thickness of 10 μm or less.
10475. The method of claim 10300, wherein the device has a coating, and the coating adheres to the surface of the pump upon deployment of the pump.
10476. The method of claim 10300, wherein the device has a coating, and wherein the coating is stable at room temperature for a period of 1 year.
10477. The method of claim 10300, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001% to about 1% by weight.
10478. The method of claim 10300, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
10479. The method of claim 10300, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
10480. The method of claim 10300, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
10481. The method of claim 10300, wherein the device has a coating, and wherein the coating further comprises a polymer.
10482. The method of claim 10300, wherein the device has a first coating having a first composition and a second coating having a second composition.
10483. The method of claim 10300, wherein the device has a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different
10484. The method of claim 10300, wherein the composition comprises a polymer.
10485. The method of claim 10300, wherein the composition comprises a polymeric carrier.
10486. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a copolymer.
10487. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a block copolymer.
10488. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a random copolymer.
10489. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a biodegradable polymer.
10490. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-biodegradable polymer.
10491. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophilic polymer.
10492. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophobic polymer.
10493. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophilic domains.
10494. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophobic domains.
10495. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-conductive polymer.
10496. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an elastomer.
10497. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrogel.
10498. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a silicone polymer.
10499. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrocarbon polymer.
10500. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a styrene-derived polymer.
10501. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a butadiene polymer.
10502. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a macromer.
10503. The method of claim 10300, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
10504. The method of claim 10300 wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an amorphous polymer.
10505. The method of claim 10300, wherein the device comprises a lubricious coating.
10506. The method of claim 10300 wherein the anti-scarring agent is located within pores or holes of the device.
10507. The method of claim 10300 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
10508. The method of claim 10300, wherein the device comprises a second pharmaceutically active agent.
10509. The method of claim 10300 wherein the device comprises an anti-inflammatory agent
10510. The method of claim 10300 wherein the device comprises an agent that inhibits infection.
10511. The method of claim 10300 wherein the device comprises an agent that inhibits infection, and wherein the agent is an anthracycline.
10512. The method of claim 10300 wherein the device comprises an agent that inhibits infection, and wherein the agent is doxorubicin.
10513. The method of claim 10300 wherein the device comprises an agent that inhibits infection, and wherein the agent is mitoxantrone.
10514. The method of claim 10300 wherein the device comprises an agent that inhibits infection, and wherein the agent is a fluoropyrimidine.
10515. The method of claim 10300 wherein the device comprises an agent that inhibits infection, and wherein the agent is 5-fluorouracil (5-FU).
10516. The method of claim 10300 wherein the device comprises an agent that inhibits infection, and wherein the agent is a folic acid antagonist.
10517. The method of claim 10300 wherein the device comprises an agent that inhibits infection, and wherein the agent is methotrexate.
10518. The method of claim 10300 wherein the device comprises an agent that inhibits infection, and wherein the agent is a podophylotoxin.
10519. The method of claim 10300 wherein the device comprises an agent that inhibits infection, and wherein the agent is etoposide.
10520. The method of claim 10300 wherein the device comprises an agent that inhibits infection, and wherein the agent is a camptothecin.
10521. The method of claim 10300 wherein the device comprises an agent that inhibits infection, and wherein the agent is a hydroxyurea.
10522. The method of claim 10300 wherein the device comprises an agent that inhibits infection, and wherein the agent is a platinum complex.
10523. The method of claim 10300 wherein the device comprises an agent that inhibits infection, and wherein the agent is cisplatin.
10524. The method of claim 10300, further comprising an anti- thrombotic agent.
10525. The method of claim 10300 wherein the device comprises a visualization agent.
10526. The method of claim 10300 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
10527. The method of claim 10300 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises barium, tantalum, or technetium.
10528. The method of claim 10300 wherein the device comprises a visualization agent, and wherein the visualization agent is a MRI responsive material.
10529. The method of claim 10300 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a gadolinium chelate.
10530. The method of claim 10300 wherein the device comprises a visualization agent, and wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
10531. The method of claim 10300 wherein the device comprises a visualization agent, and wherein the visualization agent comprises an iron oxide compound.
10532. The method of claim 10300 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a dye, pigment, or colorant.
10533. The method of claim 10300 wherein the device comprises an echogenic material.
10534. The method of claim 10300 wherein the device comprises an echogenic material, and wherein the echogenic material is in the form of a coating.
10535. The method of claim 10300 wherein the device is sterile.
10536. The method of claim 10300 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
10537. The method of claim 10300 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is connective tissue.
10538. The method of claim 10300 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is muscle tissue.
10539. The method of claim 10300 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is nerve tissue.
10540. The method of claim 10300 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is epithelium tissue.
10541. The method of claim 10300 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
10542. The method of claim 10300 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
10543. The method of claim 10300 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
10544. The method of claim 10300 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
10545. The method of claim 10300 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
10546. The method of claim 10300 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
10547. The method of claim 10300 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
10548. The method of claim 10300 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
10549. The method of claim 10300 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
10550. The method of claim 10300 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
10551. The method of claim 10300 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
10552. The method of claim 10300 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
10553. The method of claim 10300 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
10554. The method of claim 10300 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10555. The method of claim 10300 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10556. The method of claim 10300 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm 2 of device surface to which the anti-scarring agent is applied.
10557. The method of claim 10300 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10558. The method of claim 10300 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10559. The method of claim 10300 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10560. The method of claim 10300 wherein the combining is performed by direct affixing the agent or the composition to the pump.
10561. The method of claim 10300 wherein the combining is performed by spraying the agent or the component onto the pump.
10562. The method of claim 10300 wherein the combining is performed by electrospraying the agent or the composition onto the pump.
10563. The method of claim 10300 wherein the combining is performed by dipping the pump into a solution comprising the agent or the composition.
10564. The method of claim 10300 wherein the combining is performed by covalently attaching the agent or the composition to the pump.
10565. The method of claim 10300 wherein the combining is performed by non-covalently attaching the agent or the composition to the pump.
10566. The method of claim 10300 wherein the combining is performed by coating the pump with a substance that contains the agent or the composition.
10567. The method of claim 10300 wherein the combining is performed by coating the pump with a substance that absorbs the agent.
10568. The method of claim 10300 wherein the combining is performed by interweaving the pump with a thread composed of, or coated with, the agent or the composition.
10569. The method of claim 10300 wherein the combining is performed by completely covering the pump with a sleeve that contains the agent or the composition.
10570. The method of claim 10300 wherein the combining is performed by covering a portion of the pump with a sleeve that contains the agent or the composition.
10571. The method of claim 10300 wherein the combining is performed by completely covering the pump with a cover that contains the agent or the composition.
10572. The method of claim 10300 wherein the combining is performed by covering a portion of the pump with a cover that contains the agent or the composition.
10573. The method of claim 10300 wherein the combining is performed by completely covering the pump with an electrospun fabric that contains the agent or the composition.
10574. The method of claim 10300 wherein the combining is performed by covering a portion of the pump with an electrospun fabric that contains the agent or the composition.
10575. The method of claim 10300 wherein the combining is performed by completely covering the pump with a mesh that contains the agent or the composition.
10576. The method of claim 10300 wherein the combining is performed by covering a portion of the pump with a mesh that contains the agent or the composition.
10577. The method of claim 10300 wherein the combining is performed by constructing a portion of the pump with the agent or the composition.
10578. The method of claim 10300 wherein the combining is performed by impregnating the pump with the agent or the composition.
10579. The method of claim 10300 wherein the combining is performed by constructing a portion of the pump from a degradable polymer that releases the agent.
10580. The method of claim 10300 wherein the combining is performed by dipping the pump into a solution that comprise the agent and an inert solvent for the pump.
10581. The method of claim 10300 wherein the combining is performed by dipping the pump into a solution that comprises the agent and a solvent that will swill the pump.
10582. The method of claim 10300 wherein the combining is performed by dipping the pump into a solution that comprises the agent and a solvent that will dissolve the pump.
10583. The method of claim 10300 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and an inert solvent for the pump.
10584. The method of claim 10300 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and a solvent that will swill the pump.
10585. The method of claim 10300 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and a solvent that will dissolve the pump.
10586. The method of claim 10300 wherein the combining is performed by spraying the pump into a solution that comprises the agent and an inert solvent for the pump.
10587. The method of claim 10300 wherein the combining is performed by spraying the pump into a solution that comprises the agent and a solvent that will swill the pump.
10588. The method of claim 10300 wherein the combining is performed by spraying the pump into a solution that comprises the agent and a solvent that will dissolve the pump.
10589. The method of claim 10300 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and an inert solvent for the pump.
10590. The method of claim 10300 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and a solvent that will swill the pump.
10591. The method of claim 10300 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and a solvent that will dissolve the pump.
10592. The method of any one of claims 10300-10591 wherein the device is adapted for delivering 2'-deoxy 5-fluorouridine.
10593. The method of any one of claims 10300-10591 wherein the device is adapted for infusing a chemotherapeutic agent to a solid tumor.
10594. The method of any one of claims 10300-10591 wherein the device is adapted for infusing a chemotherapeutic agent to blood vessels that supply a tumor.
10595. The method of any one of claims 10300-10591 wherein the device is adapted for delivering a chemotherapeutic agent to the artery that provides blood supply to the liver of the host.
10596. A method for making a device comprising: combining a drug delivery pump for treating heart disease and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
10597. The method of claim 10596 wherein the agent inhibits cell regeneration.
10598. The method of claim 10596 wherein the agent inhibits angiogenesis.
10599. The method of claim 10596 wherein the agent inhibits fibroblast migration.
10600. The method of claim 10596 wherein the agent inhibits fibroblast proliferation.
10601. The method of claim 10596 wherein the agent inhibits deposition of extracellular matrix.
10602. The method of claim 10596 wherein the agent inhibits tissue remodeling.
10603. The method of claim 10596 wherein the agent is an angiogenesis inhibitor.
10604. The method of claim 10596 wherein the agent is a 5- lipoxygenase inhibitor or antagonist.
10605. The method of claim 10596 wherein the agent is a chemokine receptor antagonist.
10606. The method of claim 10596 wherein the agent is a cell cycle inhibitor.
10607. The method of claim 10596 wherein the agent is a taxane.
10608. The method of claim 10596 wherein the agent is an antimicrotubule agent.
10609. The method of claim 10596 wherein the agent is paclitaxel.
10610. The method of claim 10596 wherein the agent is not paclitaxel.
10611. The method of claim 10596 wherein the agent is an analogue or derivative of paclitaxel.
10612. The method of claim 10596 wherein the agent is a vinca alkaloid.
10613. The method of claim 10596 wherein the agent is camptothecin or an analogue or derivative thereof.
10614. The method of claim 10596 wherein the agent is a podophyllotoxin.
10615. The method of claim 10596 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
10616. The method of claim 10596 wherein the agent is an anthracycline.
10617. The method of claim 10596 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
10618. The method of claim 10596 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
10619. The method of claim 10596 wherein the agent is a platinum compound.
10620. The method of claim 10596 wherein the agent is a nitrosourea.
10621. The method of claim 10596 wherein the agent is a nitroimidazole.
10622. The method of claim 10596 wherein the agent is a folic acid antagonist.
10623. The method of claim 10596 wherein the agent is a cytidine analogue.
10624. The method of claim 10596 wherein the agent is a pyrimidine analogue.
10625. The method of claim 10596 wherein the agent is a fluoropyrimidine analogue.
10626. The method of claim 10596 wherein the agent is a purine analogue.
10627. The method of claim 10596 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
10628. The method of claim 10596 wherein the agent is a hydroxyurea.
10629. The method of claim 10596 wherein the agent is a mytomicin or an analogue or derivative thereof.
10630. The method of claim 10596 wherein the agent is an alkyl sulfonate.
10631. The method of claim 10596 wherein the agent is a benzamide or an analogue or derivative thereof.
10632. The method of claim 10596 wherein the agent is a nicotinamide or an analogue or derivative thereof.
10633. The method of claim 10596 wherein the agent is a . halogenated sugar or an analogue or derivative thereof.
10634. The method of claim 10596 wherein the agent is a DNA alkylating agent.
10635. The method of claim 10596 wherein the agent is an antimicrotubule agent.
10636. The method of claim 10596 wherein the agent is a topoisomerase inhibitor.
10637. The method of claim 10596 wherein the agent is a DNA cleaving agent.
10638. The method of claim 10596 wherein the agent is an antimetabolite.
10639. The method of claim 10596 wherein the agent inhibits adenosine deaminase.
10640. The method of claim 10596 wherein the agent inhibits purine ring synthesis.
10641. The method of claim 10596 wherein the agent is a nucleotide interconversion inhibitor.
10642. The method of claim 10596 wherein the agent inhibits dihydrofolate reduction.
10643. The method of claim 10596 wherein the agent blocks thymidine monophosphate.
10644. The method of claim 10596 wherein the agent causes DNA damage.
10645. The method of claim 10596 wherein the agent is a DNA intercalation agent.
10646. The method of claim 10596 wherein the agent is a RNA synthesis inhibitor.
10647. The method of claim 10596 wherein the agent is a pyrimidine synthesis inhibitor.
10648. The method of claim 10596 wherein the agent inhibits ribonucleotide synthesis or function.
10649. The method of claim 10596 wherein the agent inhibits thymidine monophosphate synthesis or function.
10650. The method of claim 10596 wherein the agent inhibits DNA synthesis.
10651. The method of claim 10596 wherein the agent causes DNA adduct formation.
10652. The method of claim 10596 wherein the agent inhibits protein synthesis.
10653. The method of claim 10596 wherein the agent inhibits microtubule function.
10654. The method of claim 10596 wherein the agent is a cyclin dependent protein kinase inhibitor.
10655. The method of claim 10596 wherein the agent is an epidermal growth factor kinase inhibitor.
10656. The method of claim 10596 wherein the agent is an elastase inhibitor.
10657. The method of claim 10596 wherein the agent is a factor Xa inhibitor.
10658. The method of claim 10596 wherein the agent is a farnesyltransferase inhibitor.
10659. The method of claim 10596 wherein the agent is a fibrinogen antagonist.
10660. The method of claim 10596 wherein the agent is a guanylate cyclase stimulant.
10661. The method of claim 10596 wherein the agent is a heat shock protein 90 antagonist.
10662. The method of claim 10596 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
10663. The method of claim 10596 wherein the agent is a guanylate cyclase stimulant.
10664. The method of claim 10596 wherein the agent is a HMGCoA reductase inhibitor.
10665. The method of claim 10596 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
10666. The method of claim 10596 wherein the agent is a hydroorotate dehydrogenase inhibitor.
10667. The method of claim 10596 wherein the agent is an IKK2 inhibitor.
10668. The method of claim 10596 wherein the agent is an IL-1 antagonist.
10669. The method of claim 10596 wherein the agent is an ICE antagonist
10670. The method of claim 10596 wherein the agent is an IRAK antagonist.
10671. The method of claim 10596 wherein the agent is an IL-4 agonist.
10672. The method of claim 10596 wherein the agent is an immunomodulatory agent.
10673. The method of claim 10596 wherein the agent is sirolimus or an analogue or derivative thereof.
10674. The method of claim 10596 wherein the agent is not sirolimus.
10675. The method of claim 10596 wherein the agent is everolimus or an analogue or derivative thereof.
10676. The method of claim 10596 wherein the agent is tacrolimus or an analogue or derivative thereof.
10677. The method of claim 10596 wherein the agent is not tacrolimus. 0678. The method of claim 10596 wherein the agent is biolmus or an analogue or derivative thereof.
10679. The method of claim 10596 wherein the agent is tresperimus or an analogue or derivative thereof.
10680. The method of claim 10596 wherein the agent is auranofin or an analogue or derivative thereof.
10681. The method of claim 10596 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
10682. The method of claim 10596 wherein the agent is gusperimus or an analogue or derivative thereof.
10683. The method of claim 10596 wherein the agent is pimecrolimus or an analogue or derivative thereof.
10684. The method of claim 10596 wherein the agent is ABT-578 or an analogue or derivative thereof.
10685. The method of claim 10596 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
10686. The method of claim 10596 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
10687. The method of claim 10596 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
10688. The method of claim 10596 wherein the agent is a leukotriene inhibitor.
10689. The method of claim 10596 wherein the agent is a MCP-1 antagonist.
10690. The method of claim 10596 wherein the agent is a MMP inhibitor.
10691. The method of claim 10596 wherein the agent is an NF kappa B inhibitor.
10692. The method of claim 10596 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
10693. The method of claim 10596 wherein the agent is an NO antagonist.
10694. The method of claim 10596 wherein the agent is a p38 MAP kinase inhibitor.
10695. The method of claim 10596 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
10696. The method of claim 10596 wherein the agent is a phosphodiesterase inhibitor.
10697. The method of claim 10596 wherein the agent is a TGF beta inhibitor.
10698. The method of claim 10596 wherein the agent is a thromboxane A2 antagonist.
10699. The method of claim 10596 wherein the agent is a TNF alpha antagonist
10700. The method of claim 10596 wherein the agent is a TACE inhibitor.
10701. The method of claim 10596 wherein the agent is a tyrosine kinase inhibitor.
10702. The method of claim 10596 wherein the agent is a vitronectin inhibitor.
10703. The method of claim 10596 wherein the agent is a fibroblast growth factor inhibitor.
10704. The method of claim 10596 wherein the agent is a protein kinase inhibitor.
10705. The method of claim 10596 wherein the agent is a PDGF receptor kinase inhibitor.
10706. The method of claim 10596 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
10707. The method of claim 10596 wherein the agent is a retinoic acid receptor antagonist.
10708. The method of claim 10596 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
10709. The method of claim 10596 wherein the agent is a fibrinogen antagonist
10710. The method of claim 10596 wherein the agent is an antimycotic agent.
10711. The method of claim 10596 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
10712. The method of claim 10596 wherein the agent is a bisphosphonate.
10713. The method of claim 10596 wherein the agent is a phospholipase A1 inhibitor.
10714. The method of claim 10596 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
10715. The method of claim 10596 wherein the agent is a macrolide antibiotic. 2005/051871
10716. The method of claim 10596 wherein the agent is a GPIIb/llla receptor antagonist.
10717. The method of claim 10596 wherein the agent is an endothelin receptor antagonist.
10718. The method of claim 10596 wherein the agent is a peroxisome proliferator-activated receptor agonist.
10719. The method of claim 10596 wherein the agent is an estrogen receptor agent.
10720. The method of claim 10596 wherein the agent is a somastostatin analogue.
10721. The method of claim 10596 wherein the agent is a neurokinin 1 antagonist.
10722. The method of claim 10596 wherein the agent is a neurokinin 3 antagonist.
10723. The method of claim 10596 wherein the agent is a VLA-4 antagonist.
10724. The method of claim 10596 wherein the agent is an osteoclast inhibitor.
10725. The method of claim 10596 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
10726. The method of claim 10596 wherein the agent is an angiotensin I converting enzyme inhibitor. 2005/051871
10727. The method of claim 10596 wherein the agent is an angiotensin II antagonist.
10728. The method of claim 10596 wherein the agent is an enkephalinase inhibitor.
10729. The method of claim 10596 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
10730. The method of claim 10596 wherein the agent is a protein kinase C inhibitor.
10731. The method of claim 10596 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
10732. The method of claim 10596 wherein the agent is a CXCR3 inhibitor.
10733. The method of claim 10596 wherein the agent is an Itk inhibitor.
10734. The method of claim 10596 wherein the agent is a cytosolic phospholipase A -alpha inhibitor.
10735. The method of claim 10596 wherein the agent is a PPAR agonist
10736. The method of claim 10596 wherein the agent is an immunosuppressant.
10737. The method of claim 10596 wherein the agent is an Erb inhibitor.
10738. The method of claim 10596 wherein the agent is an apoptosis agonist.
10739. The method of claim 10596 wherein the agent is a lipocortin agonist.
10740. The method of claim 10596 wherein the agent is a VCAM-1 antagonist.
10741. The method of claim 10596 wherein the agent is a collagen antagonist.
10742. The method of claim 10596 wherein the agent is an alpha 2 integrin antagonist.
10743. The method of claim 10596 wherein the agent is a TNF alpha inhibitor.
10744. The method of claim 10596 wherein the agent is a nitric oxide inhibitor
10745. The method of claim 10596 wherein the agent is a cathepsin inhibitor.
10746. The method of claim 10596 wherein the agent is not an anti-inflammatory agent.
10747. The method of claim 10596 wherein the agent is not a steroid.
10748. The method of claim 10596 wherein the agent is not a glucocorticosteroid.
10749. The method of claim 10596 wherein the agent is not dexamethasone.
10750. The method of claim 10596 wherein the agent is not beclomethasone.
10751. The method of claim 10596 wherein the agent is not dipropionate.
10752. The method of claim 10596 wherein the agent is not an anti-infective agent.
10753. The method of claim 10596 wherein the agent is not an antibiotic.
10754. The method of claim 10596 wherein the agent is not an anti-fungal agent.
10755. The method of claim 10596, wherein the composition comprises a polymer.
10756. The method of claim 10596, wherein the composition comprises a polymeric carrier.
10757. The method of claim 10596 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
10758. The method of claim 10596 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
10759. The method of claim 10596 wherein the device has a coating that comprises the anti-scarring agent.
10760. The method of claim 10596, wherein the device has a coating that comprises the agent and is disposed on a surface of the pump.
10761. The method of claim 10596, wherein the device has a coating that comprises the agent and directly contacts the pump.
10762. The method of claim 10596, wherein the device has a coating that comprises the agent and indirectly contacts the pump.
10763. The method of claim 10596, wherein the device has a coating that comprises the agent and partially covers the pump.
10764. The method of claim 10596, wherein the device has a coating that comprises the agent and completely covers the pump.
10765. The method of claim 10596, wherein the device has a uniform coating.
10766. The method of claim 10596, wherein the device has a non-uniform coating.
10767. The method of claim 10596, wherein the device has a discontinuous coating.
10768. The method of claim 10596, wherein the device has a patterned coating.
10769. The method of claim 10596, wherein the device has a coating with a thickness of 100 μm or less.
10770. The method of claim 10596, wherein the device has a coating with a thickness of 10 μm or less.
10771. The method of claim 10596, wherein the device has a coating, and the coating adheres to the surface of the pump upon deployment of the pump.
10772. The method of claim 10596, wherein the device has a coating, and wherein the coating is stable at room temperature for a period of 1 year.
10773. The method of claim 10596, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001 % to about 1 % by weight.
10774. The method of claim 10596, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
10775. The method of claim 10596, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
10776. The method of claim 10596, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
10777. The method of claim 10596, wherein the device has a coating, and wherein the coating further comprises a polymer.
10778. The method of claim 10596, wherein the device has a first coating having a first composition and a second coating having a second composition.
10779. The method of claim 10596, wherein the device has a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
10780. The method of claim 10596, wherein the composition comprises a polymer.
10781. The method of claim 10596, wherein the composition comprises a polymeric carrier.
10782. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a copolymer.
10783. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a block copolymer.
10784. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a random copolymer.
10785. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a biodegradable polymer.
10786. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-biodegradable polymer.
10787. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophilic polymer.
10788. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophobic polymer.
10789. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophilic domains.
10790. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophobic domains.
10791. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-conductive polymer.
10792. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an elastomer.
10793. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrogel.
10794. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a silicone polymer.
10795. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrocarbon polymer.
10796. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a styrene-derived polymer.
10797. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a butadiene polymer.
10798. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a macromer.
10799. The method of claim 10596, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a poly(ethylene glycol) polymer.
10800. The method of claim 10596 wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an amorphous polymer.
10801. The method of claim 10596, wherein the device comprises a lubricious coating.
10802. The method of claim 10596 wherein the anti-scarring agent is located within pores or holes of the device.
10803. The method of claim 10596 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
10804. The method of claim 10596, wherein the device comprises a second pharmaceutically active agent.
10805. The method of claim 10596 wherein the device comprises an anti-inflammatory agent.
10806. The method of claim 10596 wherein the device comprises an agent that inhibits infection.
10807. The method of claim 10596 wherein the device comprises an agent that inhibits infection, and wherein the agent is an anthracycline.
10808. The method of claim 10596 wherein the device comprises an agent that inhibits infection, and wherein the agent is doxorubicin.
10809. The method of claim 10596 wherein the device comprises an agent that inhibits infection, and wherein the agent is mitoxantrone.
10810. The method of claim 10596 wherein the device comprises an agent that inhibits infection, and wherein the agent is a fluoropyrimidine.
10811. The method of claim 10596 wherein the device comprises an agent that inhibits infection, and wherein the agent is 5-fluorouracil (5-FU).
10812. The method of claim 10596 wherein the device comprises an agent that inhibits infection, and wherein the agent is a folic acid antagonist.
10813. The method of claim 10596 wherein the device comprises an agent that inhibits infection, and wherein the agent is methotrexate.
10814. The method of claim 10596 wherein the device comprises an agent that inhibits infection, and wherein the agent is a podophylotoxin.
10815. The method of claim 10596 wherein the device comprises an agent that inhibits infection, and wherein the agent is etoposide.
10816. The method of claim 10596 wherein the device comprises an agent that inhibits infection, and wherein the agent is a camptothecin.
10817. The method of claim 10596 wherein the device comprises an agent that inhibits infection, and wherein the agent is a hydroxyurea.
10818. The method of claim 10596 wherein the device comprises an agent that inhibits infection, and wherein the agent is a platinum complex.
10819. The method of claim 10596 wherein the device comprises an agent that inhibits infection, and wherein the agent is cisplatin.
10820. The method of claim 10596, further comprising an anti- thrombotic agent.
10821. The method of claim 10596 wherein the device comprises a visualization agent.
10822. The method of claim 10596 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
10823. The method of claim 10596 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises barium, tantalum, or technetium.
10824. The method of claim 10596 wherein the device comprises a visualization agent, and wherein the visualization agent is a MRI responsive material.
10825. The method of claim 10596 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a gadolinium chelate.
10826. The method of claim 10596 wherein the device comprises a visualization agent, and wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
10827. The method of claim 10596 wherein the device comprises a visualization agent, and wherein the visualization agent comprises an iron oxide compound.
10828. The method of claim 10596 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a dye, pigment, or colorant.
10829. The method of claim 10596 wherein the device comprises an echogenic material.
10830. The method of claim 10596 wherein the device comprises an echogenic material, and wherein the echogenic material is in the form of a coating.
10831. The method of claim 10596 wherein the device is sterile.
10832. The method of claim 10596 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
10833. The method of claim 10596 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is connective tissue.
10834. The method of claim 10596 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is muscle tissue.
10835. The method of claim 10596 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is nerve tissue.
10836. The method of claim 10596 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is epithelium tissue.
10837. The method of claim 10596 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
10838. The method of claim 10596 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
10839. The method of claim 10596 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
10840. The method of claim 10596 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
10841. The method of claim 10596 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
10842. The method of claim 10596 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
10843. The method of claim 10596 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
10844. The method of claim 10596 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
10845. The method of claim 10596 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
10846. The method of claim 10596 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
10847. The method of claim 10596 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
10848. The method of claim 10596 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
10849. The method of claim 10596 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
10850. The method of claim 10596 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10851. The method of claim 10596 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10852. The method of claim 10596 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm 2 of device surface to which the anti-scarring agent is applied.
10853. The method of claim 10596 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10854. The method of claim 10596 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10855. The method of claim 10596 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
10856. The method of claim 10596 wherein the combining is performed by direct affixing the agent or the composition to the pump.
10857. The method of claim 10596 wherein the combining is performed by spraying the agent or the component onto the pump.
10858. The method of claim 10596 wherein the combining is performed by electrospraying the agent or the composition onto the pump.
10859. The method of claim 10596 wherein the combining is performed by dipping the pump into a solution comprising the agent or the composition.
10860. The method of claim 10596 wherein the combining is performed by covalently attaching the agent or the composition to the pump.
10861. The method of claim 10596 wherein the combining is performed by non-covalently attaching the agent or the composition to the pump.
10862. The method of claim 10596 wherein the combining is performed by coating the pump with a substance that contains the agent or the composition.
10863. The method of claim 10596 wherein the combining is performed by coating the pump with a substance that absorbs the agent.
10864. The method of claim 10596 wherein the combining is performed by interweaving the pump with a thread composed of, or coated with, the agent or the composition.
10865. The method of claim 10596 wherein the combining is performed by completely covering the pump with a sleeve that contains the agent or the composition.
10866. The method of claim 10596 wherein the combining is performed by covering a portion of the pump with a sleeve that contains the agent or the composition.
10867. The method of claim 10596 wherein the combining is performed by completely covering the pump with a cover that contains the agent or the composition.
10868. The method of claim 10596 wherein the combining is performed by covering a portion of the pump with a cover that contains the agent or the composition.
10869. The method of claim 10596 wherein the combining is performed by completely covering the pump with an electrospun fabric that contains the agent or the composition.
10870. The method of claim 10596 wherein the combining is performed by covering a portion of the pump with an electrospun fabric that contains the agent or the composition.
10871. The method of claim 10596 wherein the combining is performed by completely covering the pump with a mesh that contains the agent or the composition.
10872. The method of claim 10596 wherein the combining is performed by covering a portion of the pump with a mesh that contains the agent or the composition.
10873. The method of claim 10596 wherein the combining is performed by constructing a portion of the pump with the agent or the composition.
10874. The method of claim 10596 wherein the combining is performed by impregnating the pump with the agent or the composition.
10875. The method of claim 10596 wherein the combining is performed by constructing a portion of the pump from a degradable polymer that releases the agent.
10876. The method of claim 10596 wherein the combining is performed by dipping the pump into a solution that comprise the agent and an inert solvent for the pump.
10877. The method of claim 10596 wherein the combining is performed by dipping the pump into a solution that comprises the agent and a solvent that will swill the pump.
10878. The method of claim 10596 wherein the combining is performed by dipping the pump into a solution that comprises the agent and a solvent that will dissolve the pump.
10879. The method of claim 10596 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and an inert solvent for the pump.
10880. The method of claim 10596 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and a solvent that will swill the pump.
10881. The method of claim 10596 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and a solvent that will dissolve the pump.
10882. The method of claim 10596 wherein the combining is performed by spraying the pump into a solution that comprises the agent and an inert solvent for the pump.
10883. The method of claim 10596 wherein the combining is performed by spraying the pump into a solution that comprises the agent and a solvent that will swill the pump.
10884. The method of claim 10596 wherein the combining is performed by spraying the pump into a solution that comprises the agent and a solvent that will dissolve the pump.
10885. The method of claim 10596 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and an inert solvent for the pump.
10886. The method of claim 10596 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a I polymer and a solvent that will swill the pump.
10887. The method of claim 10596 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and a solvent that will dissolve the pump.
10888. The method of any one of claims 10596-10887 wherein the device is an implantable cardiac electrode that delivers stimulation energy and dispenses drug adjacent to the stimulation site.
10889. A method for making a device comprising: combining a drug delivery pump and an anti-scarring agent or a composition comprising an anti-scarring agent, wherein the agent inhibits scarring between the device and a host into which the device is implanted.
10890. The method of claim 10889 wherein the agent inhibits cell regeneration.
10891. The method of claim 10889 wherein the agent inhibits angiogenesis.
10892. The method of claim 10889 wherein the agent inhibits fibroblast migration.
10893. The method of claim 10889 wherein the agent inhibits fibroblast proliferation.
10894. The method of claim 10889 wherein the agent inhibits deposition of extracellular matrix.
10895. The method of claim 10889 wherein the agent inhibits tissue remodeling.
10896. The method of claim 10889 wherein the agent is an angiogenesis inhibitor.
10897. The method of claim 10889 wherein the agent is a 5- lipoxygenase inhibitor or antagonist
10898. The method of claim 10889 wherein the agent is a chemokine receptor antagonist.
10899. The method of claim 10889 wherein the agent is a cell cycle inhibitor.
10900. The method of claim 10889 wherein the agent is a taxane.
10901. The method of claim 10889 wherein the agent is an antimicrotubule agent.
10902. The method of claim 10889 wherein the agent is paclitaxel.
10903. The method of claim 10889 wherein the agent is not paclitaxel.
10904. The method of claim 10889 wherein the agent is an analogue or derivative of paclitaxel.
10905. The method of claim 10889 wherein the agent is a vinca alkaloid.
10906. The method of claim 10889 wherein the agent is camptothecin or an analogue or derivative thereof.
10907. The method of claim 10889 wherein the agent is a podophyllotoxin.
10908. The method of claim 10889 wherein the agent is a podophyllotoxin, wherein the podophyllotoxin is etoposide or an analogue or derivative thereof.
10909. The method of claim 10889 wherein the agent is an anthracycline.
10910. The method of claim 10889 wherein the agent is an anthracycline, wherein the anthracycline is doxorubicin or an analogue or derivative thereof.
10911. The method of claim 10889 wherein the agent is an anthracycline, wherein the anthracycline is mitoxantrone or an analogue or derivative thereof.
10912. The method of claim 10889 wherein the agent is a platinum compound.
10913. The method of claim 10889 wherein the agent is a nitrosourea.
10914. The method of claim 10889 wherein the agent is a nitroimidazole.
10915. The method of claim 10889 wherein the agent is a folic acid antagonist.
10916. The method of claim 10889 wherein the agent is a cytidine analogue.
10917. The method of claim 10889 wherein the agent is a pyrimidine analogue.
10918. The method of claim 10889 wherein the agent is a fluoropyrimidine analogue.
10919. The method of claim 10889 wherein the agent is a purine analogue.
10920. The method of claim 10889 wherein the agent is a nitrogen mustard or an analogue or derivative thereof.
10921. The method of claim 10889 wherein the agent is a hydroxyurea.
10922. The method of claim 10889 wherein the agent is a mytomicin or an analogue or derivative thereof.
10923. The method of claim 10889 wherein the agent is an alkyl sulfonate.
10924. The method of claim 10889 wherein the agent is a benzamide or an analogue or derivative thereof.
10925. The method of claim 10889 wherein the agent is a nicotinamide or an analogue or derivative thereof.,
10926. The method of claim 10889 wherein the agent is a halogenated sugar or an analogue or derivative thereof.
10927. The method of claim 10889 wherein the agent is a DNA alkylating agent.
10928. The method of claim 10889 wherein the agent is an antimicrotubule agent.
10929. The method of claim 10889 wherein the agent is a topoisomerase inhibitor.
10930. The method of claim 10889 wherein the agent is a DNA cleaving agent.
10931. The method of claim 10889 wherein the agent is an antimetabolite.
10932. The method of claim 10889 wherein the agent inhibits adenosine deaminase.
10933. The method of claim 10889 wherein the agent inhibits purine ring synthesis.
10934. The method of claim 10889 wherein the agent is a nucleotide interconversion inhibitor.
10935. The method of claim 10889 wherein the agent inhibits dihydrofolate reduction.
10936. The method of claim 10889 wherein the agent blocks thymidine monophosphate.
10937. The method of claim 10889 wherein the agent causes DNA damage.
10938. The method of claim 10889 wherein the agent is a DNA intercalation agent.
10939. The method of claim 10889 wherein the agent is a RNA synthesis inhibitor.
10940. The method of claim 10889 wherein the agent is a pyrimidine synthesis inhibitor.
10941. The method of claim 10889 wherein the agent inhibits ribonucleotide synthesis or function.
10942. The method of claim 10889 wherein the agent inhibits thymidine monophosphate synthesis or function.
10943. The method of claim 10889 wherein the agent inhibits DNA synthesis.
10944. The method of claim 10889 wherein the agent causes DNA adduct formation.
10945. The method of claim 10889 wherein the agent inhibits protein synthesis.
10946. The method of claim 10889 wherein the agent inhibits microtubule function.
10947. The method of claim 10889 wherein the agent is a cyclin dependent protein kinase inhibitor.
10948. The method of claim 10889 wherein the agent is an epidermal growth factor kinase inhibitor.
10949. The method of claim 10889 wherein the agent is an elastase inhibitor.
10950. The method of claim 10889 wherein the agent is a factor Xa inhibitor.
10951. The method of claim 10889 wherein the agent is a farnesyltransferase inhibitor.
10952. The method of claim 10889 wherein the agent is a fibrinogen antagonist
10953. The method of claim 10889 wherein the agent is a guanylate cyclase stimulant.
10954. The method of claim 10889 wherein the agent is a heat shock protein 90 antagonist.
10955. The method of claim 10889 wherein the agent is a heat shock protein 90 antagonist, wherein the heat shock protein 90 antagonist is geldanamycin or an analogue or derivative thereof.
10956. The method of claim 10889 wherein the agent is a guanylate cyclase stimulant.
10957. The method of claim 10889 wherein the agent is a HMGCoA reductase inhibitor.
10958. The method of claim 10889 wherein the agent is a HMGCoA reductase inhibitor, wherein the HMGCoA reductase inhibitor is simvastatin or an analogue or derivative thereof.
10959. The method of claim 10889 wherein the agent is a hydroorotate dehydrogenase inhibitor.
10960. The method of claim 10889 wherein the agent is an IKK2 inhibitor.
10961. The method of claim 10889 wherein the agent is an IL-1 antagonist.
10962. The method of claim 10889 wherein the agent is an ICE antagonist.
10963. The method of claim 10889 wherein the agent is an IRAK antagonist.
10964. The method of claim 10889 wherein the agent is an IL-4 agonist.
10965. The method of claim 10889 wherein the agent is an immunomodulatory agent.
10966. The method of claim 10889 wherein the agent is sirolimus or an analogue or derivative thereof.
10967. The method of claim 10889 wherein the agent is not sirolimus.
10968. The method of claim 10889 wherein the agent is everolimus or an analogue or derivative thereof.
10969. The method of claim 10889 wherein the agent is tacrolimus or an analogue or derivative thereof.
10970. The method of claim 10889 wherein the agent is not tacrolimus.
10971. The method of claim 10889 wherein the agent is biolmus or an analogue or derivative thereof.
10972. The method of claim 10889 wherein the agent is tresperimus or an analogue or derivative thereof.
10973. The method of claim 10889 wherein the agent is auranofin or an analogue or derivative thereof.
10974. The method of claim 10889 wherein the agent is 27-0- demethylrapamycin or an analogue or derivative thereof.
10975. The method of claim 10889 wherein the agent is gusperimus or an analogue or derivative thereof.
10976. The method of claim 10889 wherein the agent is pimecrolimus or an analogue or derivative thereof.
10977. The method of claim 10889 wherein the agent is ABT-578 or an analogue or derivative thereof.
10978. The method of claim 10889 wherein the agent is an inosine monophosphate dehydrogenase (IMPDH) inhibitor.
10979. The method of claim 10889 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is mycophenolic acid or an analogue or derivative thereof.
10980. The method of claim 10889 wherein the agent is an IMPDH inhibitor, wherein the IMPDH inhibitor is 1-alpha-25 dihydroxy vitamin D3 or an analogue or derivative thereof.
10981. The method of claim 10889 wherein the agent is a leukotriene inhibitor.
10982. The method of claim 10889 wherein the agent is a MCP-1 antagonist
10983. The method of claim 10889 wherein the agent is a MMP inhibitor.
10984. The method of claim 10889 wherein the agent is an NF kappa B inhibitor.
10985. The method of claim 10889 wherein the agent is an NF kappa B inhibitor, wherein the NF kappa B inhibitor is Bay 11-7082.
10986. The method of claim 10889 wherein the agent is an NO antagonist.
10987. The method of claim 10889 wherein the agent is a p38 MAP kinase inhibitor.
10988. The method of claim 10889 wherein the agent is a p38 MAP kinase inhibitor, wherein the p38 MAP kinase inhibitor is SB 202190.
10989. The method of claim 10889 wherein the agent is a phosphodiesterase inhibitor.
10990. The method of claim 10889 wherein the agent is a TGF beta inhibitor.
10991. The method of claim 10889 wherein the agent is a thromboxane A2 antagonist.
10992. The method of claim 10889 wherein the agent is a TNF alpha antagonist.
10993. The method of claim 10889 wherein the agent is a TACE inhibitor.
10994. The method of claim 10889 wherein the agent is a tyrosine kinase inhibitor.
10995. The method of claim 10889 wherein the agent is a vitronectin inhibitor.
10996. The method of claim 10889 wherein the agent is a fibroblast growth factor inhibitor.
10997. The method of claim 10889 wherein the agent is a protein kinase inhibitor.
10998. The method of claim 10889 wherein the agent is a PDGF receptor kinase inhibitor.
10999. The method of claim 10889 wherein the agent is an endothelial growth factor receptor kinase inhibitor.
11000. The method of claim 10889 wherein the agent is a retinoic acid receptor antagonist.
11001. The method of claim 10889 wherein the agent is a platelet derived growth factor receptor kinase inhibitor.
11002. The method of claim 10889 wherein the agent is a fibrinogen antagonist.
11003. The method of claim 10889 wherein the agent is an antimycotic agent.
11004. The method of claim 10889 wherein the agent is an antimycotic agent, wherein the antimycotic agent is sulconizole.
11005. The method of claim 10889 wherein the agent is a bisphosphonate.
11006. The method of claim 10889 wherein the agent is a phospholipase A1 inhibitor.
11007. The method of claim 10889 wherein the agent is a histamine H1/H2/H3 receptor antagonist.
11008. The method of claim 10889 wherein the agent is a macrolide antibiotic.
11009. The method of claim 10889 wherein the agent is a GPIIb/llla receptor antagonist.
11010. The method of claim 10889 wherein the agent is an endothelin receptor antagonist.
11011. The method of claim 10889 wherein the agent is a peroxisome proliferator-activated receptor agonist.
11012. The method of claim 10889 wherein the agent is an estrogen receptor agent.
11013. The method of claim 10889 wherein the agent is a somastostatin analogue.
11014. The method of claim 10889 wherein the agent is a neurokinin 1 antagonist.
11015. The method of claim 10889 wherein the agent is a neurokinin 3 antagonist.
11016. The method of claim 10889 wherein the agent is a VLA-4 antagonist.
11017. The method of claim 10889 wherein the agent is an osteoclast inhibitor.
11018. The method of claim 10889 wherein the agent is a DNA topoisomerase ATP hydrolyzing inhibitor.
11019. The method of claim 10889 wherein the agent is an angiotensin I converting enzyme inhibitor.
11020. The method of claim 10889 wherein the agent is an angiotensin II antagonist.
11021. The method of claim 10889 wherein the agent is an enkephalinase inhibitor.
11022. The method of claim 10889 wherein the agent is a peroxisome proliferator-activated receptor gamma agonist insulin sensitizer.
11023. The method of claim 10889 wherein the agent is a protein kinase C inhibitor.
11024. The method of claim 10889 wherein the agent is a ROCK (rho-associated kinase) inhibitor.
11025. The method of claim 10889 wherein the agent is a CXCR3 inhibitor.
11026. The method of claim 10889 wherein the agent is an Itk inhibitor.
11027. The method of claim 10889 wherein the agent is a cytosolic phospholipase A2-alpha inhibitor.
11028. The method of claim 10889 wherein the agent is a PPAR agonist.
11029. The method of claim 10889 wherein the agent is an immunosuppressant.
11030. The method of claim 10889 wherein the agent is an Erb inhibitor.
11031. The method of claim 10889 wherein the agent is an apoptosis agonist.
11032. The method of claim 10889 wherein the agent is a lipocortin agonist.
11033. The method of claim 10889 wherein the agent is a VCAM-1 antagonist.
11034. The method of claim 10889 wherein the agent is a collagen antagonist.
11035. The method of claim 10889 wherein the agent is an alpha 2 integrin antagonist.
11036. The method of claim 10889 wherein the agent is a TNF alpha inhibitor.
11037. The method of claim 10889 wherein the agent is a nitric oxide inhibitor
11038. The method of claim 10889 wherein the agent is a cathepsin inhibitor.
11039. The method of claim 10889 wherein the agent is not an anti-inflammatory agent.
11040. The method of claim 10889 wherein the agent is not a steroid.
11041. The method of claim 10889 wherein the agent is not a glucocorticosteroid.
11042. The method of claim 10889 wherein the agent is not dexamethasone.
11043. The method of claim 10889 wherein the agent is not beclomethasone.
11044. The method of claim 10889 wherein the agent is not dipropionate.
11045. The method of claim 10889 wherein the agent is not an anti-infective agent.
11046. The method of claim 10889 wherein the agent is not an antibiotic.
11047. The method of claim 10889 wherein the agent is not an anti-fungal agent.
11048. The method of claim 10889, wherein the composition comprises a polymer.
11049. The method of claim 10889, wherein the composition comprises a polymeric carrier.
11050. The method of claim 10889 wherein the anti-scarring agent inhibits adhesion between the device and a host into which the device is implanted.
11051. The method of claim 10889 wherein the device delivers the anti-scarring agent locally to tissue proximate to the device.
11052. The method of claim 10889 wherein the device has a coating that comprises the anti-scarring agent.
11053. The method of claim 10889, wherein the device has a coating that comprises the agent and is disposed on a surface of the pump.
11054. The method of claim 10889, wherein the device has a coating that comprises the agent and directly contacts the pump.
11055. The method of claim 10889, wherein the device has a coating that comprises the agent and indirectly contacts the pump.
11056. The method of claim 10889, wherein the device has a coating that comprises the agent and partially covers the pump.
11057. The method of claim 10889, wherein the device has a coating that comprises the agent and completely covers the pump.
11058. The method of claim 10889, wherein the device has a uniform coating.
11059. The method of claim 10889, wherein the device has a non-uniform coating.
11060. The method of claim 10889, wherein the device has a discontinuous coating.
11061. The method of claim 10889, wherein the device has a patterned coating.
11062. The method of claim 10889, wherein the device has a coating with a thickness of 100 μm or less.
11063. The method of claim 10889, wherein the device has a coating with a thickness of 10 μm or less.
11064. The method of claim 10889, wherein the device has a coating, and the coating adheres to the surface of the pump upon deployment of the pump.
11065. The method of claim 10889, wherein the device has a coating, and wherein the coating is stable at room temperature for a period of 1 year.
11066. The method of claim 10889, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 0.0001 % to about 1 % by weight
11067. The method of claim 10889, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 1% to about 10% by weight.
11068. The method of claim 10889, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 10% to about 25% by weight.
11069. The method of claim 10889, wherein the device has a coating, and wherein the anti-scarring agent is present in the coating in an amount ranging between about 25% to about 70% by weight.
11070. The method of claim 10889, wherein the device has a coating, and wherein the coating further comprises a polymer.
11071. The method of claim 10889, wherein the device has a first coating having a first composition and a second coating having a second composition.
11072. The method of claim 10889, wherein the device has a first coating having a first composition and a second coating having a second composition, wherein the first composition and the second composition are different.
11073. The method of claim 10889, wherein the composition comprises a polymer.
11074. The method of claim 10889, wherein the composition comprises a polymeric carrier.
11075. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a copolymer.
11076. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a block copolymer.
11077. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a random copolymer.
11078. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a biodegradable polymer.
11079. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-biodegradable polymer.
11080. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophilic polymer.
11081. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrophobic polymer.
11082. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophilic domains.
11083. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a polymer having hydrophobic domains.
11084. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a non-conductive polymer.
11085. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an elastomer.
11086. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrogel.
11087. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a silicone polymer.
11088. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a hydrocarbon polymer.
11089. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a styrene-derived polymer.
11090. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a butadiene polymer.
11091. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a macromer.
11092. The method of claim 10889, wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises a poly( ethylene glycol) polymer.
11093. The method of claim 10889 wherein the composition comprises a polymeric carrier, and wherein the polymeric carrier comprises an amorphous polymer.
11094. The method of claim 10889, wherein the device comprises a lubricious coating.
11095. The method of claim 10889 wherein the anti-scarring agent is located within pores or holes of the device.
11096. The method of claim 10889 wherein the anti-scarring agent is located within a channel, lumen, or divet of the device.
11097. The method of claim 10889, wherein the device comprises a second pharmaceutically active agent.
11098. The method of claim 10889 wherein the device comprises an anti-inflammatory agent.
11099. The method of claim 10889 wherein the device comprises an agent that inhibits infection.
11100. The method of claim 10889 wherein the device comprises an agent that inhibits infection, and wherein the agent is an anthracycline.
11101. The method of claim 10889 wherein the device comprises an agent that inhibits infection, and wherein the agent is doxorubicin.
11102. The method of claim 10889 wherein the device comprises an agent that inhibits infection, and wherein the agent is mitoxantrone.
11103. The method of claim 10889 wherein the device comprises an agent that inhibits infection, and wherein the agent is a fluoropyrimidine.
11104. The method of claim 10889 wherein the device comprises an agent that inhibits infection, and wherein the agent is 5-fluorouracil (5-FU).
11105. The method of claim 10889 wherein the device comprises an agent that inhibits infection, and wherein the agent is a folic acid antagonist.
11106. The method of claim 10889 wherein the device comprises an agent that inhibits infection, and wherein the agent is methotrexate.
11107. The method of claim 10889 wherein the device comprises an agent that inhibits infection, and wherein the agent is a podophylotoxin.
11108. The method of claim 10889 wherein the device comprises an agent that inhibits infection, and wherein the agent is etoposide.
11109. The method of claim 10889 wherein the device comprises an agent that inhibits infection, and wherein the agent is a camptothecin.
11110. The method of claim 10889 wherein the device comprises an agent that inhibits infection, and wherein the agent is a hydroxyurea.
11111. The method of claim 10889 wherein the device comprises an agent that inhibits infection, and wherein the agent is a platinum complex.
11112. The method of claim 10889 wherein the device comprises an agent that inhibits infection, and wherein the agent is cisplatin.
11113. The method of claim 10889, further comprising an anti- thrombotic agent.
11114. The method of claim 10889 wherein the device comprises I a visualization agent.
11115. The method of claim 10889 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises a metal, a halogenated compound, or a barium containing compound.
11116. The method of claim 10889 wherein the device comprises a visualization agent, wherein the visualization agent is a radiopaque material, and wherein the radiopaque material comprises barium, tantalum, or technetium.
11117. The method of claim 10889 wherein the device comprises a visualization agent, and wherein the visualization agent is a MRI responsive material.
11118. The method of claim 10889 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a gadolinium chelate.
11119. The method of claim 10889 wherein the device comprises a visualization agent, and wherein the visualization agent comprises iron, magnesium, manganese, copper, or chromium.
11120. The method of claim 10889 wherein the device comprises a visualization agent, and wherein the visualization agent comprises an iron oxide compound.
11121. The method of claim 10889 wherein the device comprises a visualization agent, and wherein the visualization agent comprises a dye, pigment, or colorant.
11122. The method of claim 10889 wherein the device comprises an echogenic material.
11123. The method of claim 10889 wherein the device comprises an echogenic material, and wherein the echogenic material is in the form of a coating.
11124. The method of claim 10889 wherein the device is sterile.
11125. The method of claim 10889 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device.
11126. The method of claim 10889 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is connective tissue.
11127. The method of claim 10889 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is muscle tissue.
11128. The method of claim 10889 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is nerve tissue.
11129. The method of claim 10889 wherein the anti-scarring agent is released into tissue in the vicinity of the device after deployment of the device, and wherein the tissue is epithelium tissue.
11130. The method of claim 10889 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from the time of deployment of the device to about 1 year.
11131. The method of claim 10889 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 month to 6 months.
11132. The method of claim 10889 wherein the anti-scarring agent is released in effective concentrations from the device over a period ranging from about 1 - 90 days.
11133. The method of claim 10889 wherein the anti-scarring agent is released in effective concentrations from the device at a constant rate.
11134. The method of claim 10889 wherein the anti-scarring agent is released in effective concentrations from the device at an increasing rate.
11135. The method of claim 10889 wherein the anti-scarring agent is released in effective concentrations from the device at a decreasing rate.
11136. The method of claim 10889 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by diffusion over a period ranging from the time of deployment of the device to about 90 days.
11137. The method of claim 10889 wherein the anti-scarring agent is released in effective concentrations from the composition comprising the anti-scarring agent by erosion of the composition over a period ranging from the time of deployment of the device to about 90 days.
11138. The method of claim 10889 wherein the device comprises about 0.01 μg to about 10 μg of the anti-scarring agent.
11139. The method of claim 10889 wherein the device comprises about 10 μg to about 10 mg of the anti-scarring agent.
11140. The method of claim 10889 wherein the device comprises about 10 mg to about 250 mg of the anti-scarring agent.
11141. The method of claim 10889 wherein the device comprises about 250 mg to about 1000 mg of the anti-scarring agent.
11142. The method of claim 10889 wherein the device comprises about 1000 mg to about 2500 mg of the anti-scarring agent.
11143. The method of claim 10889 wherein a surface of the device comprises less than 0.01 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
11144. The method of claim 10889 wherein a surface of the device comprises about 0.01 μg to about 1 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
11145. The method of claim 10889 wherein a surface of the device comprises about 1 μg to about 10 μg of the anti-scarring agent per mm of device surface to which the anti-scarring agent is applied.
11146. The method of claim 10889 wherein a surface of the device comprises about 10 μg to about 250 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
11147. The method of claim 10889 wherein a surface of the device comprises about 250 μg to about 1000 μg of the anti-scarring agent of anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
11148. The method of claim 10889 wherein a surface of the device comprises about 1000 μg to about 2500 μg of the anti-scarring agent per mm2 of device surface to which the anti-scarring agent is applied.
11149. The method of claim 10889 wherein the combining is performed by direct affixing the agent or the composition to the pump.
11150. The method of claim 10889 wherein the combining is performed by spraying the agent or the component onto the pump.
11151. The method of claim 10889 wherein the combining is performed by electrospraying the agent or the composition onto the pump.
11152. The method of claim 10889 wherein the combining is performed by dipping the pump into a solution comprising the agent or the composition.
11153. The method of claim 10889 wherein the combining is performed by covalently attaching the agent or the composition to the pump.
11154. The method of claim 10889 wherein the combining is performed by non-covalently attaching the agent or the composition to the pump.
11155. The method of claim 10889 wherein the combining is performed by coating the pump with a substance that contains the agent or the composition.
11156. The method of claim 10889 wherein the combining is performed by coating the pump with a substance that absorbs the agent.
11157. The method of claim 10889 wherein the combining is performed by interweaving the pump with a thread composed of, oi" coated with, the agent or the composition.
11158. The method of claim 10889 wherein the combining is performed by completely covering the pump with a sleeve that contains the agent or the composition.
11159. The method of claim 10889 wherein the combining is performed by covering a portion of the pump with a sleeve that contains the agent or the composition.
11160. The method of claim 10889 wherein the combining is performed by completely covering the pump with a cover that contains the agent or the composition.
11161. The method of claim 10889 wherein the combining is performed by covering a portion of the pump with a cover that contains the agent or the composition.
11162. The method of claim 10889 wherein the combining is performed by completely covering the pump with an electrospun fabric that contains the agent or the composition.
11163. The method of claim 10889 wherein the combining is performed by covering a portion of the pump with an electrospun fabric that contains the agent or the composition.
11164. The method of claim 10889 wherein the combining is performed by completely covering the pump with a mesh that contains the agent or the composition.
11165. The method of claim 10889 wherein the combining is performed by covering a portion of the pump with a mesh that contains the agent or the composition.
11166. The method of claim 10889 wherein the combining is performed by constructing a portion of the pump with the agent or the composition.
11167. The method of claim 10889 wherein the combining is performed by impregnating the pump with the agent or the composition.
11168. The method of claim 10889 wherein the combining is performed by constructing a portion of the pump from a degradable polymer that releases the agent.
11169. The method of claim 10889 wherein the combining is performed by dipping the pump into a solution that comprise the agent and an inert solvent for the pump.
11170. The method of claim 10889 wherein the combining is performed by dipping the pump into a solution that comprises the agent and a solvent that will swill the pump.
11171. The method of claim 10889 wherein the combining is performed by dipping the pump into a solution that comprises the agent and a solvent that will dissolve the pump.
11172. The method of claim 10889 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and an inert solvent for the pump.
11173. The method of claim 10889 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and a solvent that will swill the pump.
11174. The method of claim 10889 wherein the combining is performed by dipping the pump into a solution that comprises the agent, a polymer and a solvent that will dissolve the pump.
11175. The method of claim 10889 wherein the combining is performed by spraying the pump into a solution that comprises the agent and an inert solvent for the pump.
11176. The method of claim 10889 wherein the combining is performed by spraying the pump into a solution that comprises the agent and a solvent that will swill the pump.
11177. The method of claim 10889 wherein the combining is performed by spraying the pump into a solution that comprises the agent and a solvent that will dissolve the pump.
11178. The method of claim 10889 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and an inert solvent for the pump.
11179. The method of claim 10889 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and a solvent that will swill the pump.
11180. The method of claim 10889 wherein the combining is performed by spraying the pump into a solution that comprises the agent, a polymer and a solvent that will dissolve the pump.